Cu-Al spinel oxide as a sustained release catalyst for methanol steam reforming: Enhancing the catalytic performance via surface reconstruction
-
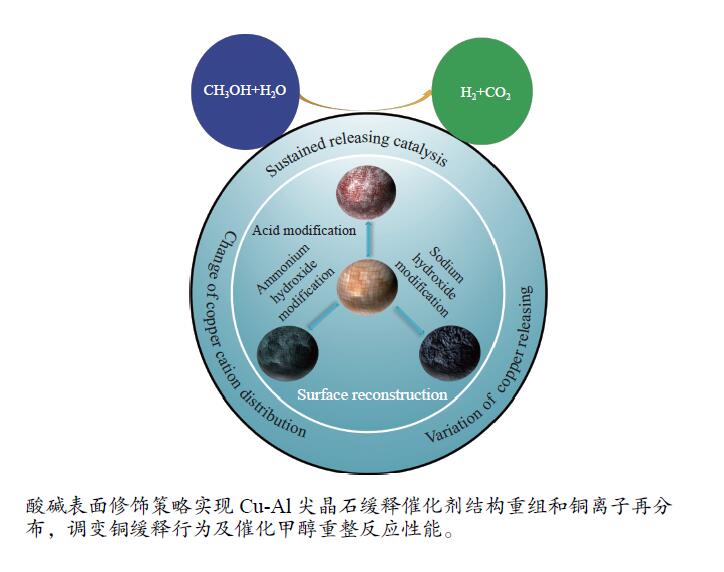
摘要: Cu-Al尖晶石氧化物作为甲醇水蒸气重整制氢缓释催化剂,在反应过程中逐渐释放活性Cu,其催化行为与催化剂的表面结构密切相关。本研究采用酸碱溶液对固相球磨法合成的Cu-Al尖晶石催化剂进行表面处理,以期通过表面组成及结构的修饰来提高其催化性能。研究结果表明,硝酸、氨水、氢氧化钠溶液与催化剂的作用差别极大,酸处理去除了催化剂表面的Cu和Al物种,氢氧化钠溶液处理则主要去除了Al物种,氨水溶液处理作用最弱,去除了极少量的Cu和Al。伴随着Cu、Al物种的流失,催化剂表面结构重组和铜物种再分布改变了铜的缓释行为。反应性能评价结果表明,硝酸和氨水处理改善了缓释催化性能,其中,硝酸处理后的催化剂表现出更优异的催化稳定性;而氢氧化钠溶液处理不利于缓释催化性能的提高。结合反应后催化剂表征结果,阐明缓释Cu粒径大小和Cu晶粒微观结构应变对催化性能发挥着重要作用。当前工作为提高缓释催化提供了一种切实可行的方法。Abstract: Cu-Al spinel oxide as a sustained release catalyst gradually releases active metal Cu during the methanol steam reforming reaction, whose catalytic behavior depends strongly on the surface structure of the catalyst. In this context, Cu-Al spinel solid solution is synthesized by a solid phase ball milling method, followed by treating with acidic and basic solutions in order to modulate the surface composition and structure, thereby to further improve the catalytic performance. Nitric acid is effective for the removal of both surface dispersed Cu and Al oxide species, whereas sodium hydroxide is only effective for the removal of Al oxide species, and ammonium hydroxide shows the weakest effect, removing a very small amount of Cu and Al species. Accompanying with the loss of Cu and Al species, the catalyst surface undergoes structural reconstruction, showing a redistribution of Cu species. Consequently, the copper releasing behavior varies drastically. The catalytic testing results show that the nitric acid and ammonium hydroxide treated catalysts present improved activity, where in the former also shows better stability. Sodium hydroxide treatment has a negative effect on the sustained releasing catalytic performance. In combination with the characterization results of the tested catalysts, it is found that both the copper particle dimension and the microstructure strain of sustained released copper play important roles in the catalytic performance. The findings of this report provide a practical method for the improvement of the sustained releasing catalysis.
-
Key words:
- Cu-Al spinel /
- surface modification /
- surface reconstruction /
- methanol steam reforming
-
Table 1 Results of the element extraction analysis and textural structural properties of the untreated and treated catalysts
Sample CA CA-HNO3 CA-NH3 CA-NaOH Loss of Cua w/% 0 2.40 0.10 0.20 Loss of Ala w/% 0 1.60 0.36 6.90 SBET/(m2·g−1) 40.84 47.08 31.49 41.97 dpore/nm 26.04 33.18 24.21 32.05 vpore/(cm3·g−1) 0.266 0.39 0.191 0.336 a: The molar percentage of metal in catalyst extracted with the chemical leaching solution is analyzed by ICP. Table 2 Structural properties of the catalysts
Sample CA CA-HNO3 CA-NH3 CA-NaOH a/A 7.992 8.0748 8.0507 8.0360 dspinel/nm 14.5 16.2 15.4 15.1 dCu/nm 27.9 11.2 13.5 12.6 Microstrain/% 0.0879 0.0837 0.0850 0.0957 Note: a represents the lattice parameter of the spinel phase obtained by the XRD pattern; dspinel represents average spinel particle size of the spinel (sp) calculated by using XRD data;
dCu represents the copper particle sizes of the tested catalyst calculated by the Scherrer equation from the XRD patterns; Microstrain of the copper crystals in tested catalysts is also calculated by XRD data.Table 3 The component content in the catalyst
Samples CA CA-HNO3 CA-NH3 CA-NaOH Xnon spinela/% 13.1 12.3 13.0 19.6 Xeasily reduced spinela/% 70.0 74.6 74.3 71.4 Xhardly reduced spinela/% 16.9 13.1 12.7 9.0 Cu/Alb (%) 5.7 6.3 6.5 7.0 CuO/Sp CuOb (%) 54/46 13/87 56/44 28/72 a: The molar fraction of non spinel copper species, easily reducible spinel Cu (II) and hardly reducible spinel Cu (II) are denoted as Xnon spinel, Xeasily reduced spinel and Xhardly reduced spinel, respectively according to H2-TPR area integration;
b: Based on XPS data, Cu/Al denotes the molar fraction of surface Cu to Al; CuO/Sp CuO denotes the molar fraction of non spinel CuO with respect to the spinel CuO species. -
[1] TIAN N, ZHOU Z Y, SUN S G, et al. Synthesis of tetrahexahedral platinum nanocrystals with high-index facets and high electro-oxidation activity[J]. Science,2007,316(5825):732−735. doi: 10.1126/science.1140484 [2] ZHANG L, ROLING L T, WANG X, et al. Platinum-based nanocages with subnanometer-thick walls and well-defined, controllable facets[J]. Science,2015,349(6246):412−416. doi: 10.1126/science.aab0801 [3] SANCHES S G, FLORES H, SILVA M I P. Cu/ZnO and Cu/ZnO/ZrO2 catalysts used for methanol steam reforming[J]. Mol Catal,2018,454:55−62. doi: 10.1016/j.mcat.2018.05.012 [4] LI G, GU C, ZHU W, et al. Hydrogen production from methanol decomposition using Cu-Al spinel catalysts[J]. J Clean Prod,2018,183:415−423. doi: 10.1016/j.jclepro.2018.02.088 [5] LI G. Study of copper spinel catalyst for hydrogen generation by methanol[D]. Taiyuan: Institute of Coal Chemistry, Chinese Academy of Sciences, 2012. [6] XI H, HOU X, LIU Y, et al. Cu-Al spinel oxide as an efficient catalyst for methanol steam reforming[J]. Angew Chem Int Ed,2014,53(44):11886−11889. doi: 10.1002/anie.201405213 [7] LIU Y, QING S, HOU X, et al. Temperature dependence of Cu-Al spinel formation and its catalytic performance in methanol steam reforming[J]. Catal Sci Technol,2017,7(21):5069−5078. [8] LIU Y, QING S, HOU X, et al. Cu-Ni-Al spinel oxide as an efficient durable catalyst for methanol steam reforming[J]. ChemCatChem,2018,10(24):5698−5706. doi: 10.1002/cctc.201801472 [9] HOU X, QING S, LIU Y, et al. Enhancing effect of MgO modification of Cu-Al spinel oxide catalyst for methanol steam reforming[J]. Int J Hydrogen Energy,2020,45(1):477−489. doi: 10.1016/j.ijhydene.2019.10.164 [10] SHI L, WANG D, YU X, et al. Adsorption of Cun (n = 1–4) clusters on CuAl2O4 spinel surface: A DFT study[J]. Mol Catal,2019,468:29−35. doi: 10.1016/j.mcat.2019.02.009 [11] KANGO S, KALIA S, CELLI A, et al. The Surface modification of inorganic nanoparticles for development of organic-inorganic nanocomposites-A review[J]. Prog Polym Sci,2013,38:1232−1261. doi: 10.1016/j.progpolymsci.2013.02.003 [12] HONG R, PAN T, QIAN J, et al. Synthesis and surface modification of ZnO nanoparticles[J]. Chem Eng J,2006,119:71−81. doi: 10.1016/j.cej.2006.03.003 [13] PALMA V, RUOCCO C, CORTESE M, et al. Bioalcohol reforming: an overview of the recent advances for the enhancement of catalyst stability[J]. Catalysts,2020,10:665. doi: 10.3390/catal10060665 [14] KUSCHE M, ENZENBERGER F, BAJUS S, et al. Enhanced activity and selectivity in catalytic methanol steam reforming by basic alkali metal salt coatings[J]. Angew Chem,2013,125:5132−5136. doi: 10.1002/ange.201209758 [15] WANG Z, LIU P, HAN J, et al. Engineering the internal surfaces of three-dimensional nanoporous catalysts by surfactant-modified dealloying[J]. Nat Commun,2017,8(1):1066. doi: 10.1038/s41467-017-01085-3 [16] TANG W, XIAO W, WANG S, et al. Boosting catalytic propane oxidation over PGM-free Co3O4 nanocrystal aggregates through chemical leaching: A comparative study with Pt and Pd based catalysts[J]. Appl Catal B: Environ,2018,226:585−595. doi: 10.1016/j.apcatb.2017.12.075 [17] SEVERINO F, BRITO J L, LAINE J, et al. Nature of copper active sites in the carbon monoxide oxidation on CuAl2O4 and CuCr2O4 spinel type catalysts[J]. J Catal,1998,177(1):82−95. doi: 10.1006/jcat.1998.2094 [18] LIN X C, LI X, DING X W, et al. Application of Na/K modified ZSM-5 zeolite in direct catalytic synthesis of light olefins from syngas[J]. Mod Chem Ind,2020,40(3):126−130. [19] RODENBOUGH P P, ZHENG C, LIU Y, et al. Lattice expansion in metal oxide nanoparticles: MgO, Co3O4, & Fe3O4[J]. J Am Ceram Soc,2017,100(1):384−392. doi: 10.1111/jace.14478 [20] LOU Y, MA J, CAS X, et al. Promoting effects of In2O3 on Co3O4 for CO oxidation: Tuning O2 activation and CO adsorption strength simultaneously[J]. ACS Catal,2014,4(11):4143−4152. doi: 10.1021/cs501049r [21] IGBARI O, XIE Y, JIN Z, et al. Microstructural and electrical properties of CuAlO2 ceramic prepared by a novel solvent-free ester elimination process[J]. J Alloy Compd,2015,653:219−227. doi: 10.1016/j.jallcom.2015.08.268 -





 下载:
下载: