Hydrogenation of lignin-derived phenolic compounds over Co@C catalysts
-
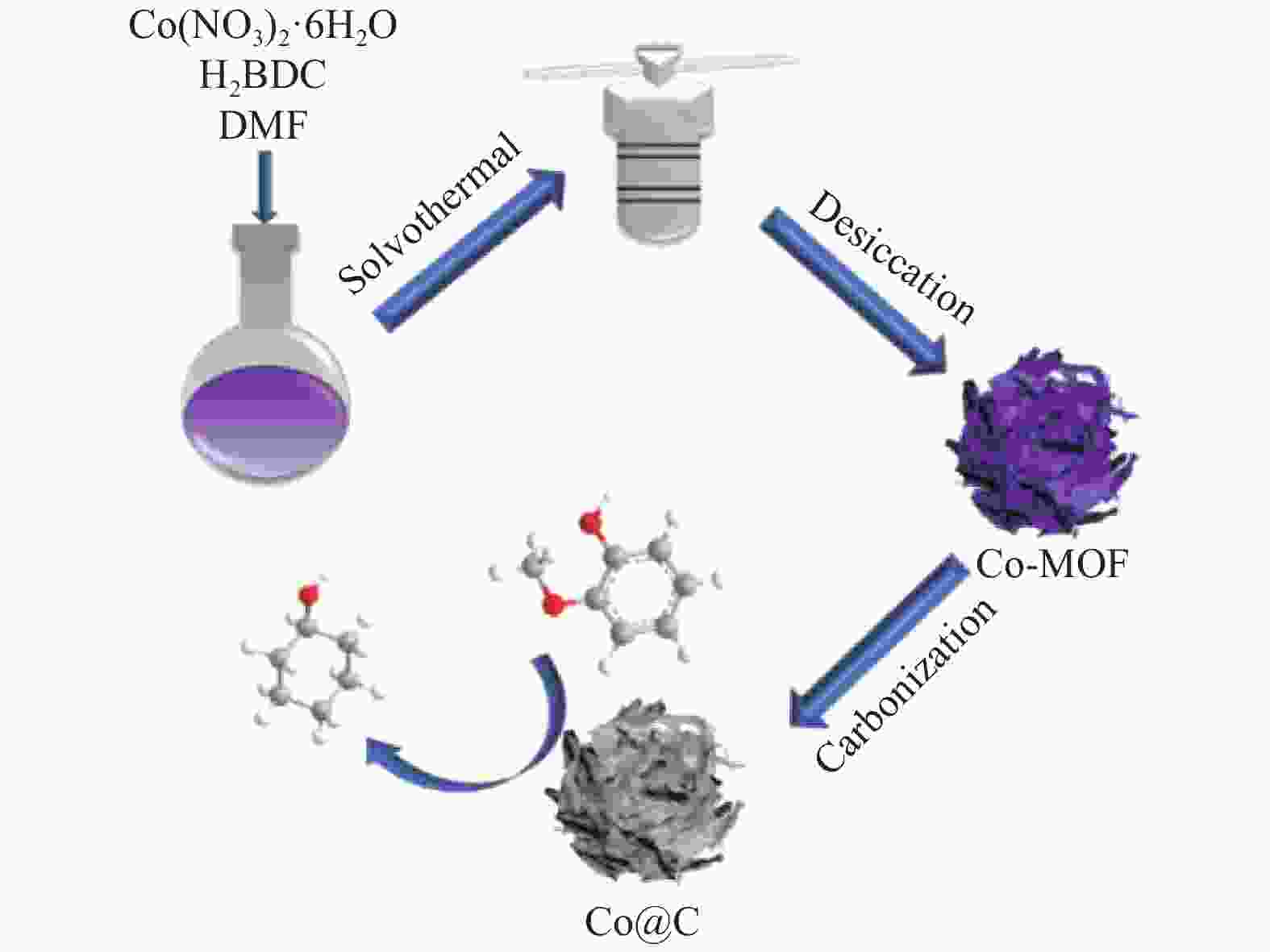
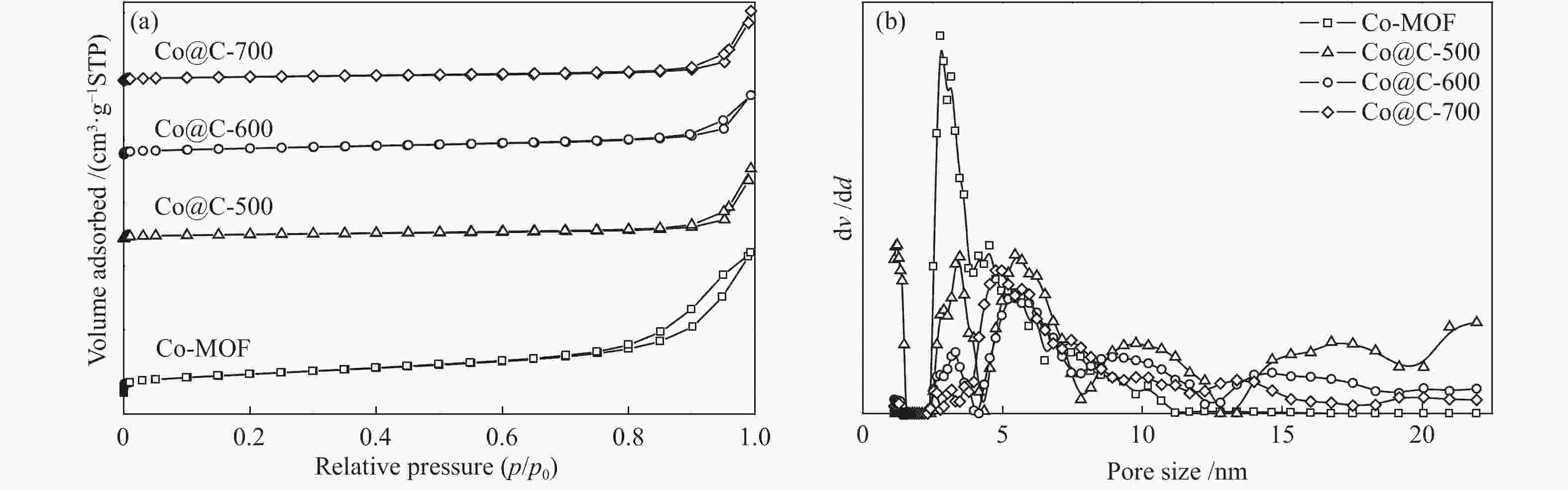
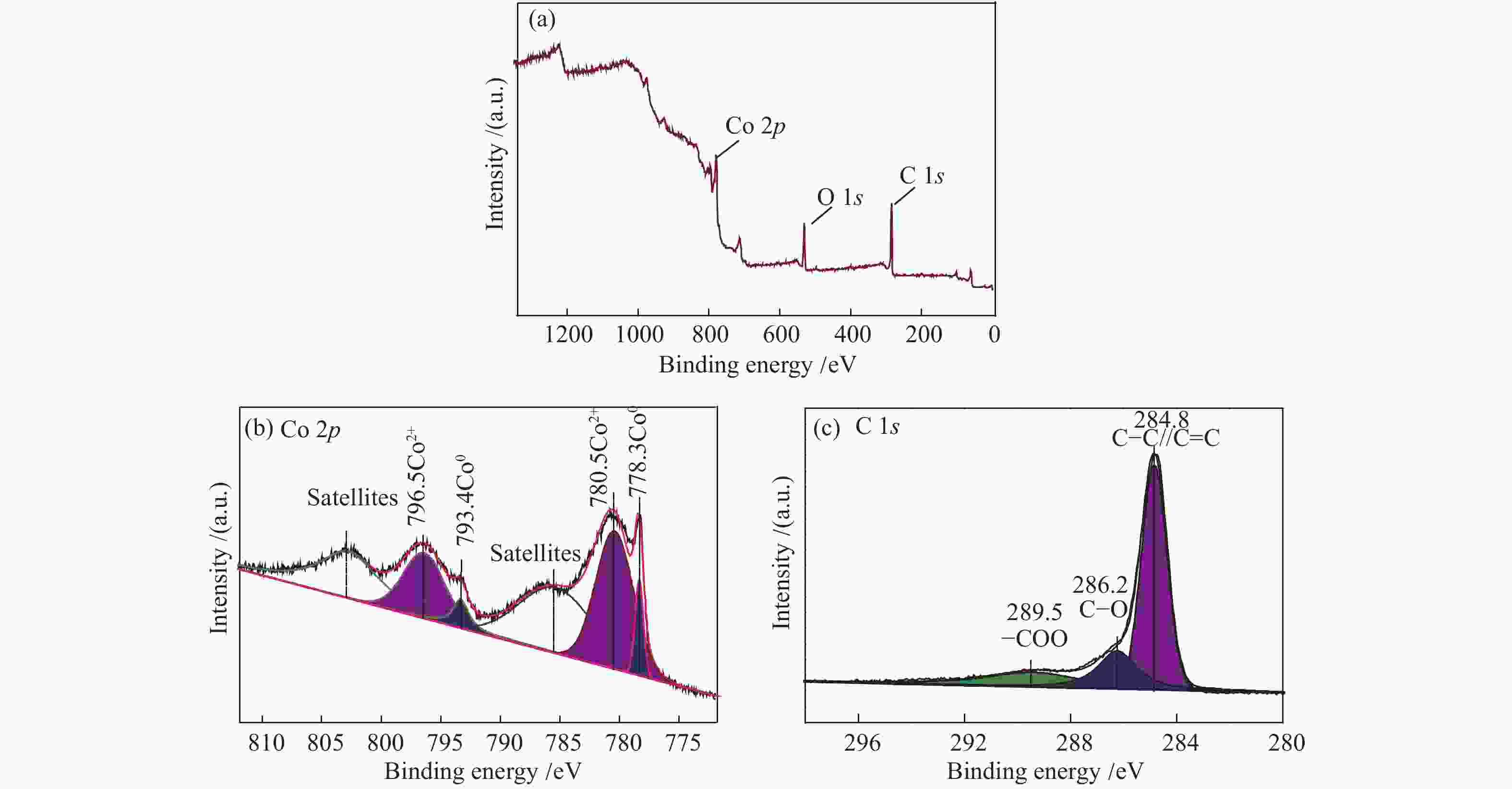
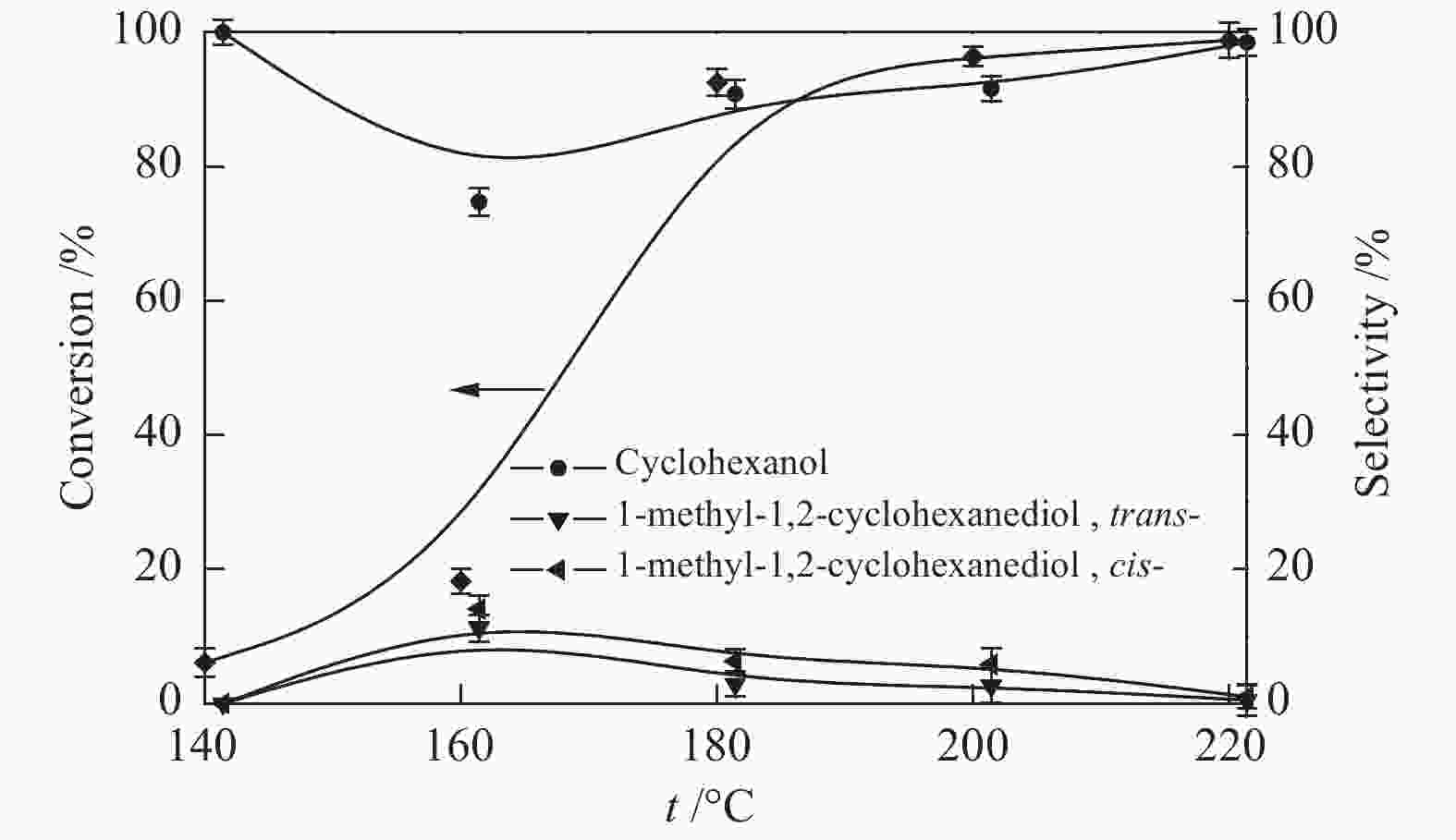
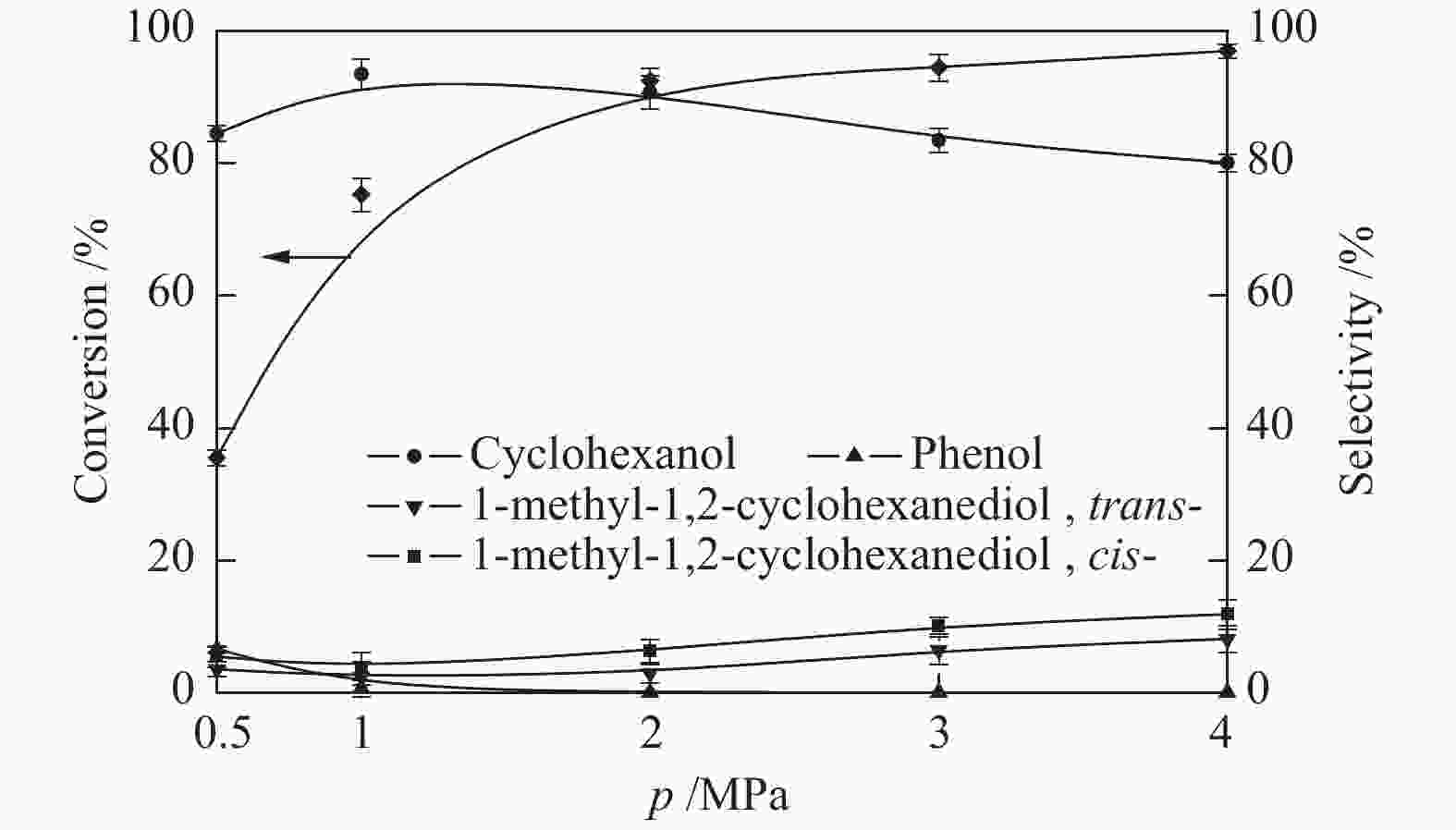
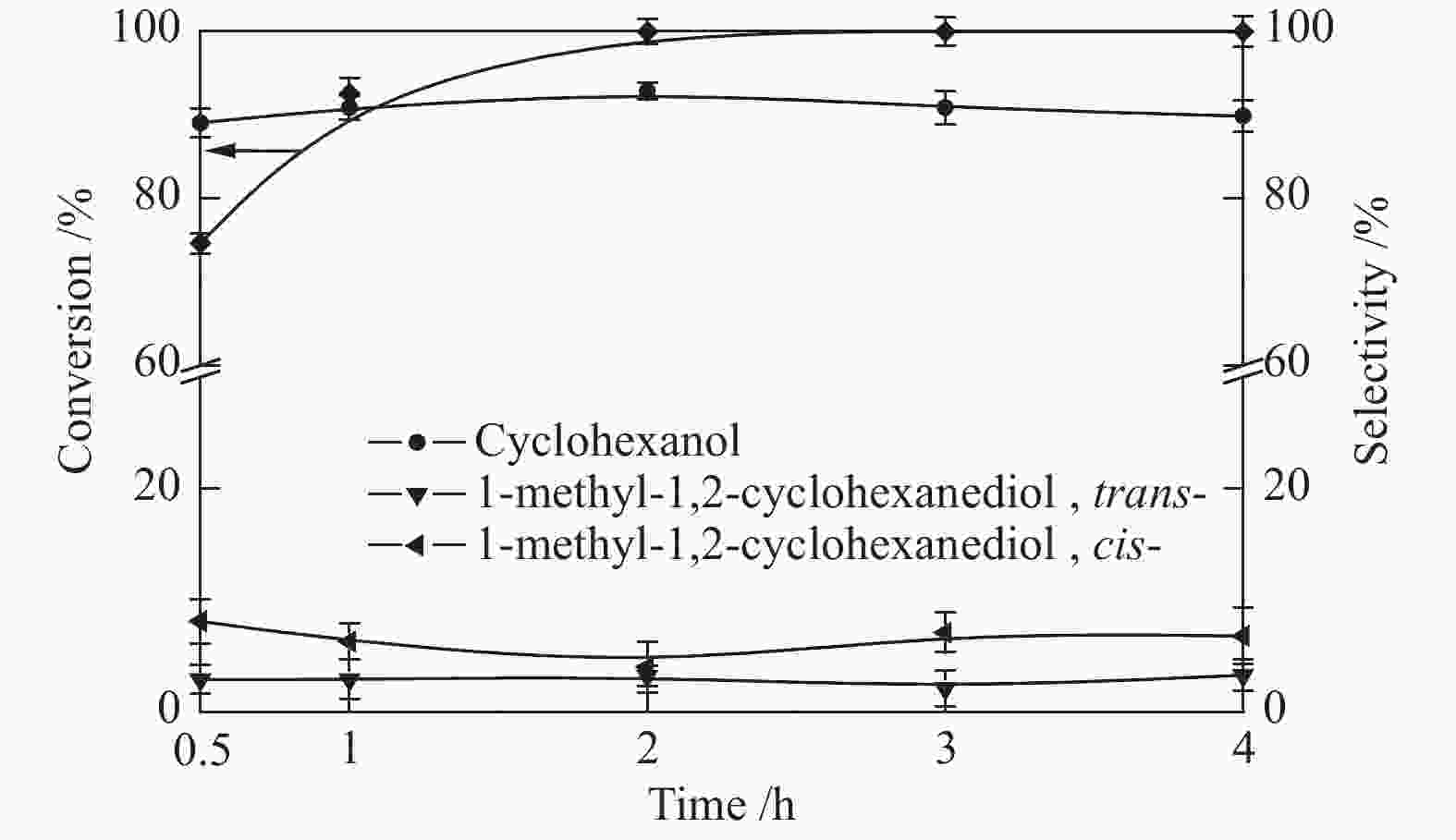
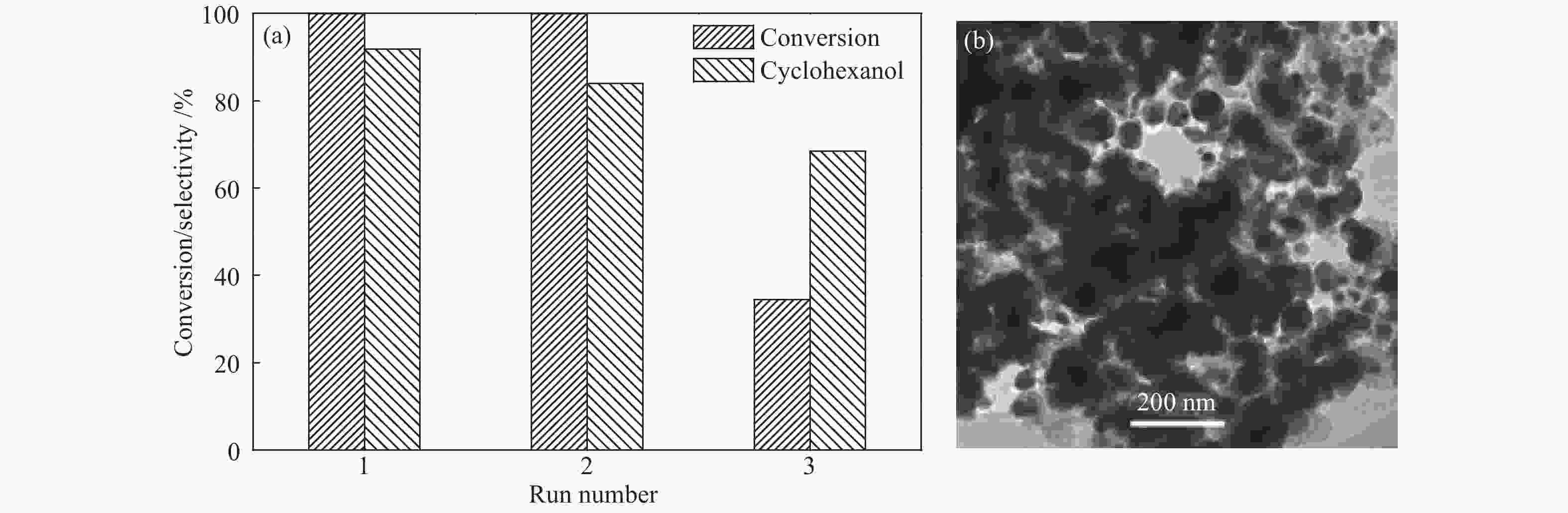
摘要: 采用溶剂热法合成Co-MOF,然后通过一步热解法制备了Co@C催化剂。通过N2物理吸附-脱附(BET)、X射线衍射(XRD)、X射线光电子能谱(XPS)、扫描电子显微镜(SEM)和透射电子显微镜(TEM)等分析手段对Co@C催化剂的结构进行了表征。探讨了Co-MOF热解温度、反应温度、初始氢压以及反应时间对Co@C催化愈创木酚加氢转化的影响。结果表明,Co-MOF和Co@C中均以介孔为主;片层结构的Co-MOF热解后变成不规则的球状,并且随着热解温度升高,Co@C的比表面积不断减小。以Co@C-600为催化剂,在反应温度180 ℃、初始氢压2 MPa、反应时间2 h的条件下,愈创木酚完全转化,环己醇的选择性为92.8%。Co@C催化愈创木酚加氢转化的主要反应路径为先通过脱甲氧基生成苯酚,进一步加氢生成环己醇。此外,Co@C-600对苯酚、对甲氧基苯酚和4-甲基愈创木酚等其他衍生酚单体也具有较好的催化活性。Abstract: Co-MOF was firstly prepared by solvothermal method, and then Co@C catalyst was prepared by one-step pyrolysis method from Co-MOF. The structure of Co@C catalyst was characterized by N2 physical adsorption-desorption (BET), X-ray diffraction (XRD), X-ray photoelectron spectroscopy (XPS), scanning electron microscopy (SEM) and transmission electron microscopy (TEM). Effects of Co-MOF pyrolysis temperature, reaction temperature, initial hydrogen pressure and reaction time on catalytic hydrogenation of guaiacol were investigated. The results show that both Co-MOF and Co@C are dominated by mesoporous. After pyrolysis, lamellar structure of Co-MOF changes into irregular sphericity. As raising pyrolysis temperature, specific surface area of Co@C decreases continuously. Under the conditions of reaction temperature 180 ℃, initial hydrogen pressure 2 MPa and reaction time 2 h, the guaiacol was completely transformed and selectivity of cyclohexanol was 92.8% using Co@C-600 as catalyst. The main reaction pathway of guaiacol hydrogenation catalyzed by Co@C is that guaiacol firstly forms phenol through removal of methoxyl group, and further is hydrogenated to cyclohexanol. In addition, Co@C-600 also has good catalytic activity for other phenolic monomers derived from lignin, such as phenol, p-methoxyphenol and 4-methyl guaiacol.
-
Key words:
- lignin /
- guaiacol /
- cyclohexanol /
- hydrodeoxygenation /
- Co@C catalyst
-
表 1 Co-MOF及Co@C-X的理化性质
Table 1 Physicochemical properties of Co-MOF and Co@C-X
Sample Surface
area/
(m2·g−1)aPore
volume/
(cm3·g−1)bPore diameter/
nmbCobalt content
w/%cCo-MOF 56.30 0.03 3.41 37.3 Co@C-500 35.51 0.03 3.83 50.6 Co@C-600 31.93 0.04 3.81 56.2 Co@C-700 17.77 0.04 3.82 64.8 acalculated by BET method; bcalculated by BJH method; cmeasured with LA-ICP-MS 表 2 愈创木酚在不同催化剂上的转化及产物分布
Table 2 Guaiacol conversion and product distribution over different catalysts
Sample Conversion/% Selectivity/% 

(trans-, cis-)Co@C-500 6.4 77.6 22.4 Co@C-600 92.5 90.8 9.2 Co@C-700 trace − − 表 3 Co@C-600对木质素衍生酚类单体的催化
Table 3 Catalysis of Co@C-600 to lignin-derived phenolic monomers
Substrate Conversion/% Selectivity/% 
99.9 
100
99.9 
75.1
2.8
22.1
99.9 
2.6
97.4
99.9
100.0
99.9 
100.0
99.9 
100.0 -
[1] SHERWOOD J. The significance of biomass in a circular economy[J]. Bioresour Technol,2020,300:122755. doi: 10.1016/j.biortech.2020.122755 [2] 余强, 庄新姝, 袁振宏, 亓伟, 王琼, 谭雪松, 许敬亮, 张宇, 徐慧娟, 马隆龙. 木质纤维素类生物质制取燃料及化学品的研究进展[J]. 化工进展,2012,31(4):784−791.YU Qiang, ZHUANG Xin-shu, YUAN Zhen-hong, QI Wei, WANG Qiong, TAN Xue-song, XU Jin-liang, ZHANG Yu, XU Hui-juan, MA Long-long. Research progress on fuel and chemicals production from lignocellulose biomass[J]. Chem Ind Eng Prog,2012,31(4):784−791. [3] 朱晨杰, 张会岩, 肖睿, 陈勇, 柳东, 杜风光, 应汉杰, 欧阳平凯. 木质纤维素高值化利用的研究进展[J]. 中国科学: 化学,2015,45(5):454−478. doi: 10.1360/N032014-00280ZHU Chen-jie, ZHANG Hui-yan, XIAO Rui, CHEN Yong, LIU Dong, DU Feng-guang, YING Han-jie, OUYANG Ping-kai. Research progress of high value utilization of lignocellulose[J]. Sci Sin Chim,2015,45(5):454−478. doi: 10.1360/N032014-00280 [4] 袁正求, 龙金星, 张兴华, 夏莹, 王铁军, 马隆龙. 木质纤维素催化转化制备能源平台化合物[J]. 化学进展,2016,28(1):103−110.YUAN Zhen-qiu, LONG Jin-xing, ZHANG Xing-hua, XIA Ying, WANG Tie-jun, MA Long-long. Catalytic conversion of lignocellulose into energy platform chemicals[J]. Prog Chem,2016,28(1):103−110. [5] UPTON B M, KASKO A M. Strategies for the conversion of lignin to high-value polymeric materials: review and perspective[J]. Chem Rev,2015,116(4):2275−2306. [6] PEREZ J, MUNOZ-DORADO J, DE LA RUBIA T, MARTINEZ J. Biodegradation and biological treatments of cellulose, hemicellulose and lignin: An overview[J]. Int Microbiol,2002,5(2):53−63. doi: 10.1007/s10123-002-0062-3 [7] 袁亮. 木质素磺酸盐在混凝土外加剂方面的应用[J]. 特钢技术,2012,18(2):58−61. doi: 10.3969/j.issn.1674-0971.2012.02.018YUAN Liang. Application of lignosulfonate in concrete admixture[J]. Spec Steel Techonl,2012,18(2):58−61. doi: 10.3969/j.issn.1674-0971.2012.02.018 [8] TYMCHYSHYN M, YUAN Z, ZHANG Y, XU C C. Catalytic hydrodeoxygenation of guaiacol for organosolv lignin depolymerization-catalyst screening and experimental validation[J]. Fuel,2019,254:115664. doi: 10.1016/j.fuel.2019.115664 [9] VERMA S, NADAGOUDA MN, VARMA RS. Visible light-mediated and water-assisted selective hydrodeoxygenation of lignin-derived guaiacol to cyclohexanol[J]. Green Chem,2019,21(6):1253−1257. doi: 10.1039/C8GC03951H [10] 于玉肖, 徐莹, 王铁军, 马隆龙, 张琦, 张兴华, 张雪. 木质素降解模型化合物愈创木酚及苯酚原位加氢制备环己醇[J]. 燃料化学学报,2013,41(4):443−448. doi: 10.3969/j.issn.0253-2409.2013.04.009YU Yu-xiao, XU Ying, WANG Tie-jun, MA Long-long, ZHANG Qi, ZHANG Xing-hua, ZHANG Xue. In-situ hydrogenation of lignin depolymerization model compounds to cyclohexanol[J]. J Fuel Chem Technol,2013,41(4):443−448. doi: 10.3969/j.issn.0253-2409.2013.04.009 [11] LUO Z C, ZHENG Z X, WANG Y C, SUN G, JIANG H, ZHAO C. Hydrothermally stable Ru/HZSM-5-catalyzed selective hydrogenolysis of lignin-derived substituted phenols to bio-arenes in water[J]. Green Chem,2016,18(21):5845−5858. doi: 10.1039/C6GC01971D [12] WANG X, ZHU S, WANG S, HE Y, LIU Y, WANG J G, FAN W B, LV Y K. Low temperature hydrodeoxygenation of guaiacol into cyclohexane over Ni/SiO2 catalyst combined with Hβ zeolite[J]. RSC Adv,2019,9(7):3868−3876. doi: 10.1039/C8RA09972C [13] WANG X, ZHU S, WANG S, WANG J G, FAN W B, LV Y K. Ni nanoparticles entrapped in nickel phyllosilicate for selective hydrogenation of guaiacol to 2-methoxycyclohexanol[J]. Appl Catal A: Gen,2018,568:231−241. doi: 10.1016/j.apcata.2018.10.009 [14] LU J Q, LIU X, YU G Q, LV J K, RONG Z M, WANG M, WANG Y. Selective Hydrodeoxygenation of guaiacol to cyclohexanol catalyzed by nanoporous nickel[J]. Catal Lett,2019,150(3):837−848. [15] GUO M, PENG J, YANG Q H, LI C. Highly active and selective RuPd bmetallic NPs for the cleavage of the diphenyl ether C–O bond[J]. ACS Catal,2018,8(12):11174−11183. doi: 10.1021/acscatal.8b03253 [16] HUA M L, SONG J L, XIE C, WU H R, HU Y, HUANG X, HAN B X. Ru/hydroxyapatite as a dual-functional catalyst for efficient transfer hydrogenolytic cleavage of aromatic ether bonds without additional bases[J]. Green Chem,2019,21(18):5073−5079. doi: 10.1039/C9GC02336D [17] 严龙, 庞欢, 黄耀兵, 傅尧. Pd催化木质素醚类二聚体分子内氢转移断裂C-O键研究[J]. 化学学报,2014,72(9):1005−1011. doi: 10.6023/A14050397YAN Long, PANG Huan, HUANG Yao-bing, FU Yao. Supported Pd catalysts for the C-O cleavage of the lignin derived model dimers through intramolecular hydrogenolysis reaction[J]. Acta Chim Sin,2014,72(9):1005−1011. doi: 10.6023/A14050397 [18] 邱泽刚, 尹婵娟, 李志勤, 冯跃阔. 酚类加氢脱氧催化剂研究进展[J]. 化工进展,2019,38(8):3658−3669.QIU Ze-gang, YIN Chan-juan, LI Zhi-qin, FENG Kuo-yue. Recent advances in hydrodeoxygenation catalysts for phenols[J]. Chem Ind Eng Prog,2019,38(8):3658−3669. [19] YUAN S, FENG L, WANG K C, PANG J D, BOSCH M, LOLLAR C, SUN Y J, QIN J S, YANG X Y, ZHANG P, WANG Q, ZOU L F, ZHANG Y M, ZHANG L L, FANG Y, LI J L, ZHOU H C. Stable metal-organic frameworks: design, synthesis, and applications[J]. Adv Mater,2018,30(37):1704303. doi: 10.1002/adma.201704303 [20] 黄刚, 陈玉贞, 江海龙. 金属有机骨架材料在催化中的应用[J]. 化学学报,2016,74(2):113−129. doi: 10.6023/A15080547HUANG Gang, CHEN Yu-zhen, JIANG Hai-long. Metal-organic frameworks for catalysis[J]. Acta Chim Sin,2016,74(2):113−129. doi: 10.6023/A15080547 [21] SHEN K, CHEN X D, CHEN J Y, LI Y W. Development of MOF-derived carbon-based nanomaterials for efficient catalysis[J]. ACS Catal,2016,6(9):5887−5903. doi: 10.1021/acscatal.6b01222 [22] WANG J, ZHONG Q, XIONG Y H, CHENG D Y, ZENG Y Q, BU Y F. Fabrication of 3D Co-doped Ni-based MOF hierarchical micro-flowers as a high-performance electrode material for supercapacitors[J]. Appl Surf Sci,2019,483:1158−1165. doi: 10.1016/j.apsusc.2019.03.340 [23] REZAEE S, SHAHROKHIAN S. Facile synthesis of petal-like NiCo/NiO-CoO/nanoporous carbon composite based on mixed-metallic MOFs and their application for electrocatalytic oxidation of methanol[J]. Appl Catal B: Environ,2019,244:802−813. doi: 10.1016/j.apcatb.2018.12.013 [24] CAI J Y, CHEN Y, SONG H T, HOU L X, LI Z H. MOF derived C/Co@C with a “one-way-valve”-like graphitic carbon layer for selective semi-hydrogenation of aromatic alkynes[J]. Carbon,2020,160:64−70. doi: 10.1016/j.carbon.2020.01.006 [25] LIU X H, XU L J, XU G Y, JIA W D, MA Y F, ZHANG Y. Selective hydrodeoxygenation of lignin-derived phenols to cyclohexanols or cyclohexanes over magnetic CoNx@NC catalysts under mild conditions[J]. ACS Catal,2016,6(11):7611−7620. doi: 10.1021/acscatal.6b01785 [26] ZHU J, CHEN F Q, ZHANG Z G, LI M, YANG Q W, YANG Y W, BAO Z B, REN Q L. M-gallate (M = Ni, Co) metal-organic framework-derived Ni/C and bimetallic Ni-Co/C catalysts for lignin conversion into monophenols[J]. ACS Sustainable Chem Eng,2019,7(15). [27] DONG L, YIN L L, XIA Q E, LIU X H, GONG X Q, WANG Y Q. Size-dependent catalytic performance of ruthenium nanoparticles in the hydrogenolysis of a β-O-4 lignin model compound[J]. Catal Sci Technol,2018,8(3):735−745. doi: 10.1039/C7CY02014G [28] SCHUTYSER W, VAN DEN BOSSCHE G, RAAFFELS A, VAN DEN BOSCH S, KOELEWIJN S F, RENDERS T, SELS B F. Selective conversion of lignin-derivable 4-alkylguaiacols to 4-alkylcyclohexanols over noble and non-noble-metal catalysts[J]. ACS Sustainable Chem Eng,2016,4(10):5336−5346. doi: 10.1021/acssuschemeng.6b01580 [29] NAKAGAWA Y, ISHIKAWA M, TAMURA M, TOMISHIGE K. Selective production of cyclohexanol and methanol from guaiacol over Ru catalyst combined with MgO[J]. Green Chem,2014,16(4):2197−2203. doi: 10.1039/C3GC42322K [30] LONG J X, SHU S Y, WU Q Y, YUAN Z Q, WANG T J, XU Y, ZHANG X H, ZHANG Q, MA L L. Selective cyclohexanol production from the renewable lignin derived phenolic chemicals catalyzed by Ni/MgO[J]. Energy Convers Manage,2015,105:570−577. doi: 10.1016/j.enconman.2015.08.016 [31] ZHOU M H, WANG Y, WANG Y B, XIAO G M. Catalytic conversion of guaiacol to alcohols for bio-oil upgrading[J]. J Energy Chem,2015,24(4):425−431. doi: 10.1016/j.jechem.2015.06.012 [32] 郭荷民. 利用电子效应解释取代基的位置及种类对芳香化合物化学性质的影响[J]. 大学化学,2008,(5):54−57.GUO He-min. The effects of substituent position and type on the chemical properties of aromatic compounds were explained by electron effect[J]. Univ Chem,2008,(5):54−57. -





 下载:
下载: