Effect of solvent swelling on structure and pyrolysis product distribution of Ordos lignite
-
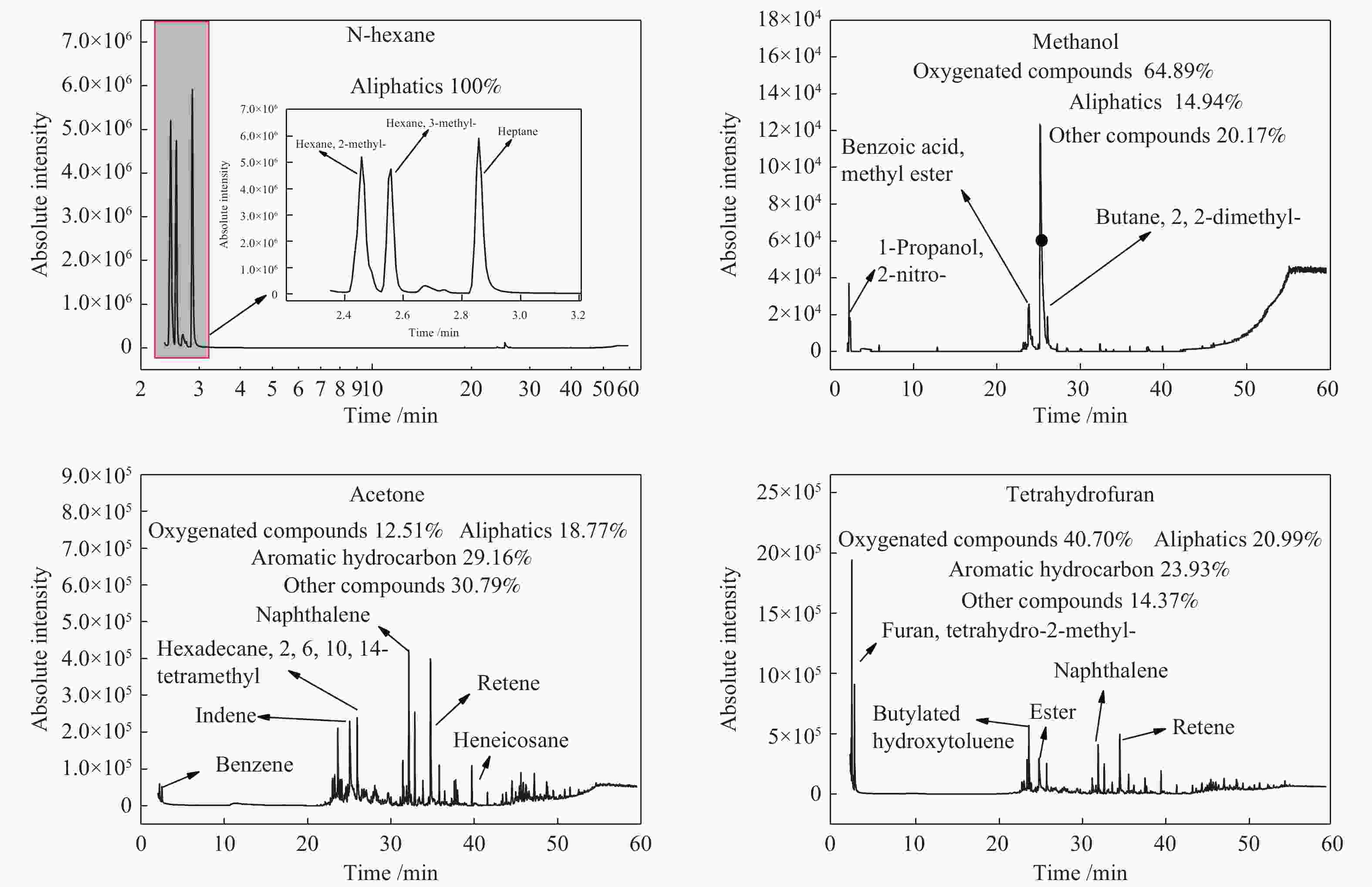
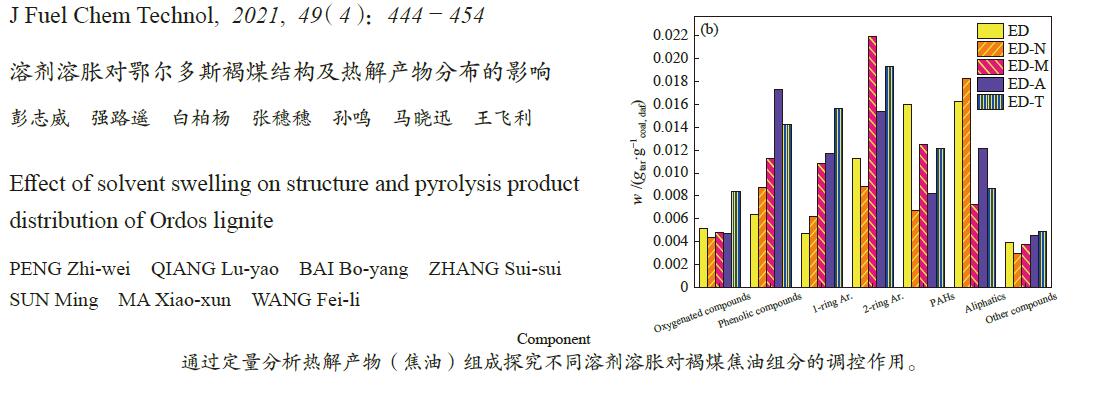
摘要: 采用四种有机溶剂对鄂尔多斯褐煤进行溶胀预处理并在粉-粒流化床上进行快速热解,探究溶胀对褐煤结构及热解特性的影响。采用傅里叶变换红外光谱仪、热重分析仪、气相色谱-质谱联用仪、固体核磁、X射线衍射、全自动化学吸附仪等仪器对溶胀煤及热解产物进行表征。结果表明,非极性溶剂对煤结构影响较小。极性溶剂可以降低褐煤含氧官能团的氢键交联,增加小分子的流动性,增加煤的平均孔径。溶胀后的煤热解焦油和气相产率提高,热解水产率降低。与原煤相比,极性溶剂甲醇、丙酮、四氢呋喃处理煤样的热解焦油产率分别提高了18.88%、26.72%、33.58%,焦油的轻质组分中酚类、单环和双环类芳香烃组分含量明显增加。Abstract: Four kinds of organic solvents were used to pretreat Ordos lignite to obtain swelling coal. The effect of swelling on the structure and pyrolysis characteristics of Ordos lignite was investigated by fast pyrolysis of swelling coal in a powder-particle fluidized bed. The swelling coal and pyrolysis products were characterized by FT-IR, TGA, GC-MS, 13C NMR, XRD. The results show that non-polar solvent has little effect on coal structure. The polar solvents can reduce the hydrogen bond crosslinking of oxygen-containing functional groups of lignite, increase the fluidity of small molecules and the average pore diameter of coal. After swelling, The yield of coal tar and gas phase yield increases, the pyrolysis water yield decreases. After pretreatment with methanol, acetone, and tetrahydrofuran, the yield of pyrolysis tar increased by 18.88%, 26.72%, 33.58% respectively. Compared with raw coal, and the contents of phenolic, monocyclic and bicyclic aromatic hydrocarbons components in the light components of tar were significantly increased.
-
Key words:
- swelling /
- lignite /
- fast pyrolysis /
- powder-particle fluidized bed
-
表 1 粉-粒流化床实验参数
Table 1 Experimental parameters of powder particle fluidized bed
Gas Feed rate/(gcoal·min−1) Mass/g Gas flow/(L·min−1) Temperature/℃ ED SiO2 feed gas fluidized gas reactor cyclone separator condenser tube N2 0.2 5.0 56.9−57.0 1.2 0.6 600 350 −40 ± 2 表 2 实验煤样的溶胀度、工业分析和元素分析
Table 2 Swelling degree and proximate and ultimate analyses of coal samples used in the experiment
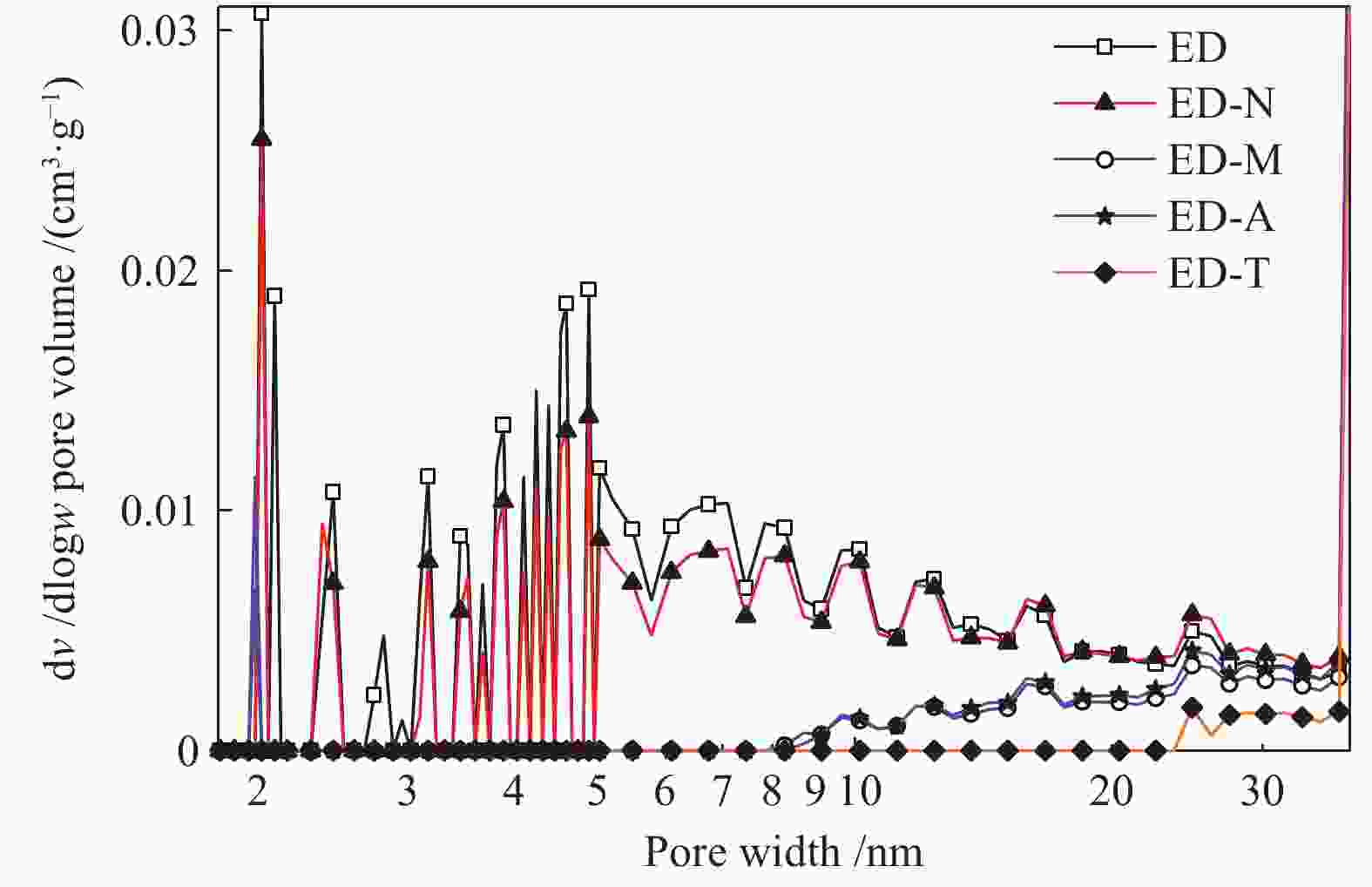
Sample Q Proximate analysis w/% Ultimate analysis wdaf/% Atomic ratio Md Ad Vdaf FCdaf* N C H S O* O/C H/C ED — 2.59 6.21 37.27 62.73 1.24 79.45 4.52 0.40 14.40 0.225 0.683 ED-N 1.07 3.01 6.25 36.54 63.46 1.22 80.54 4.28 0.33 13.63 0.215 0.637 ED-M 1.27 3.29 6.12 37.36 62.64 1.18 81.17 4.27 0.30 14.17 0.221 0.639 ED-A 1.36 3.89 6.16 36.91 63.09 1.21 79.70 4.14 0.26 14.69 0.227 0.624 ED-T 1.50 3.45 6.29 37.84 62.16 1.16 79.85 4.23 0.25 14.52 0.225 0.635 note: * means the data obtained by subtraction method; the data of proximate and ultimate analyses are obtained after the coal samples were dried at 105 ℃ for 12 h 表 3 煤样孔结构参数
Table 3 Pore structure parameters of coal sample
Samples Surface
area /(m2·g−1)Pore volume/
(mL·g−1)Pore size/
nmED 7.86 0.0153 7.78 ED-N 6.51 0.0156 9.61 ED-M 2.23 0.0102 18.30 ED-A 2.70 0.0124 18.42 ED-T 0.77 0.0062 32.40 表 4 TG参数
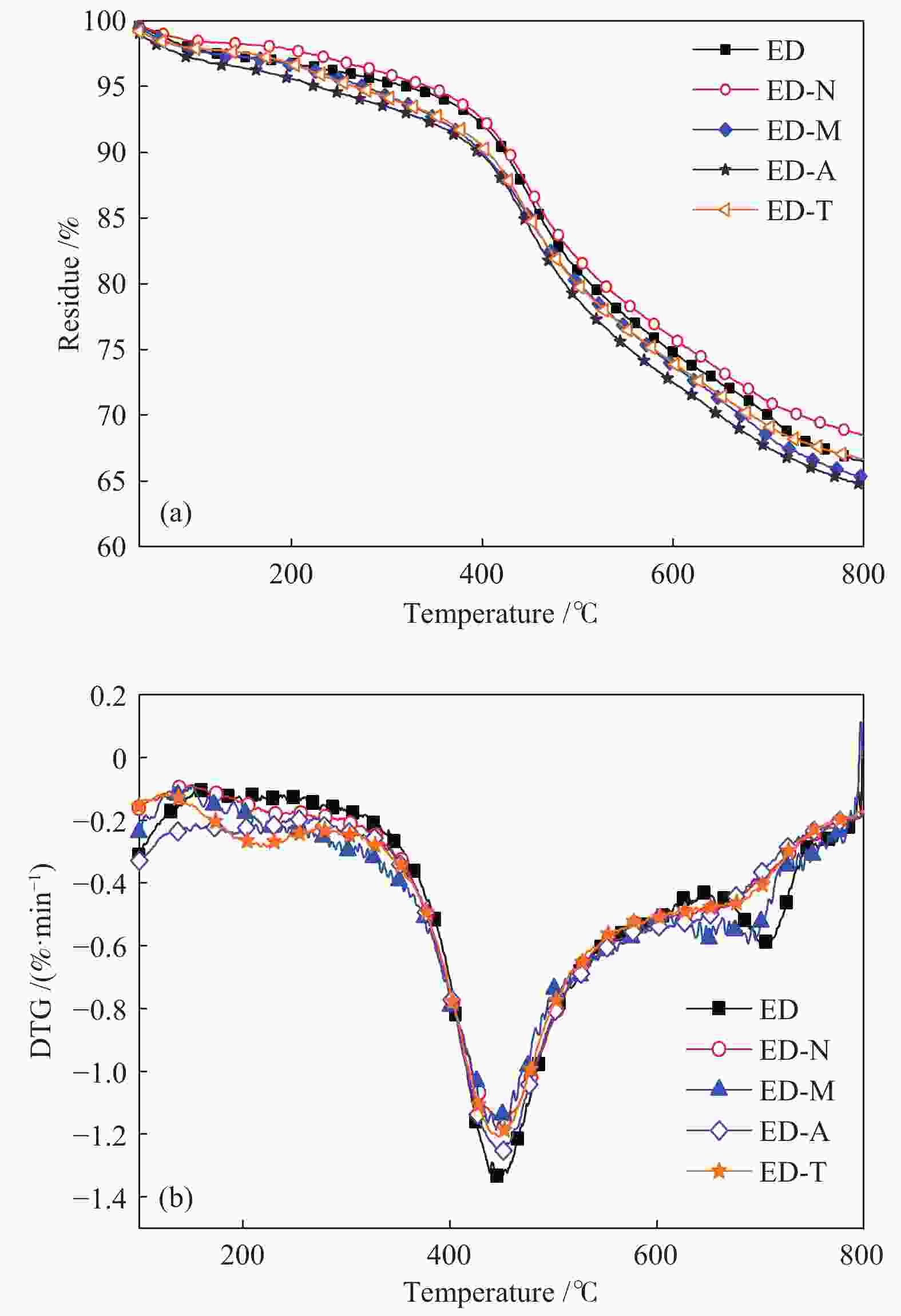
Table 4 TG parameters
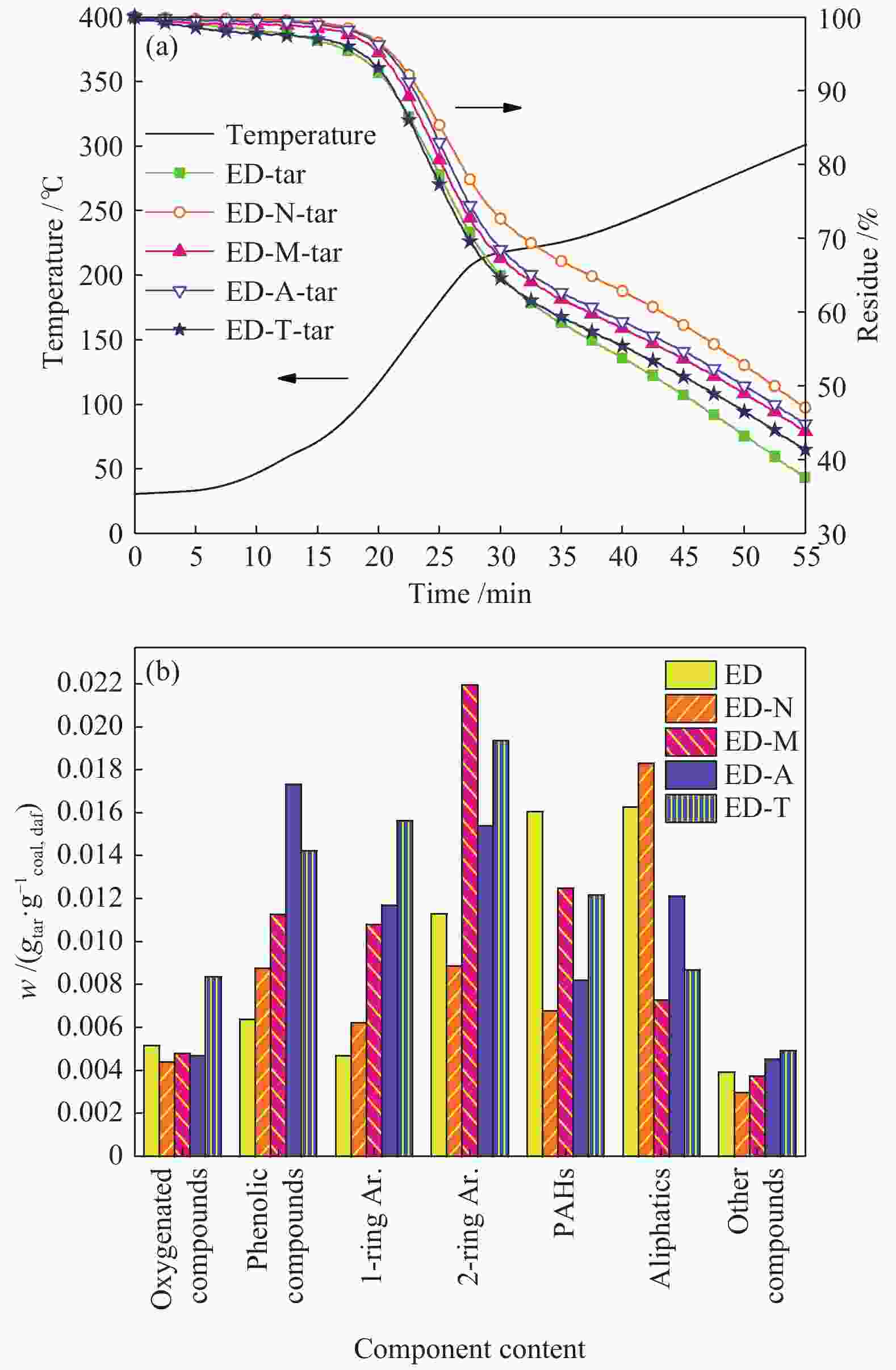
Sample w/% ti/℃ tmax/℃ tf/℃ tp/℃ Rmax/(%·min−1) ED 33.48 355 452 549 700 1.34 ED-N 31.51 344 450 560 − 1.16 ED-M 34.41 322 445 568 676 1.18 ED-A 35.28 344 447 550 − 1.26 ED-T 33.37 339 449 559 − 1.21 note: − means that there is no obvious peak 表 5 XRD分峰拟合参数
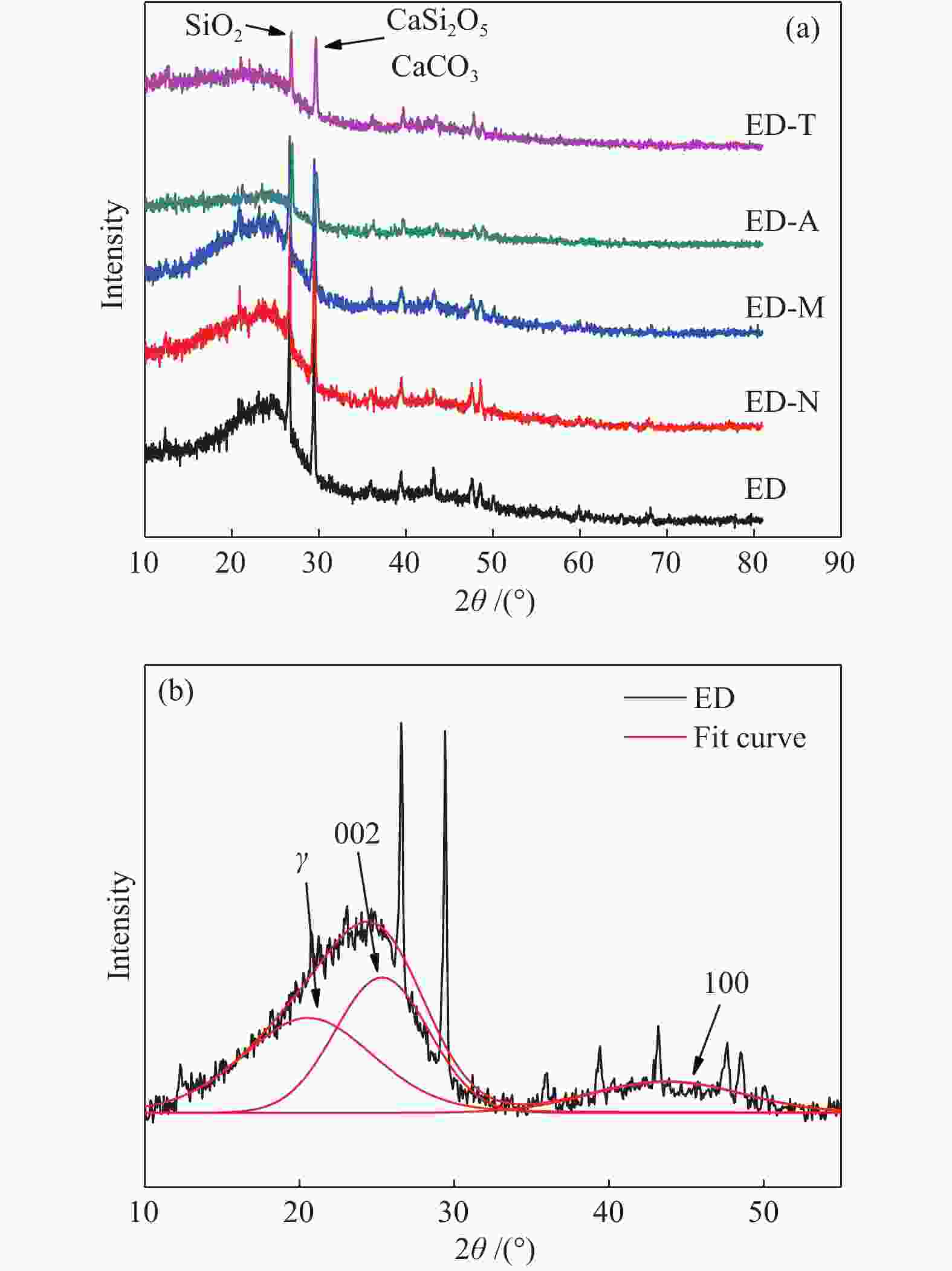
Table 5 Fitting parameters of XRD peaks
Sample 2θ002/(°) d002/nm Lc/nm n 2θ100/(°) La/nm ED 25.32 0.1802 1.2157 6.75 43.74 1.9479 ED-N 25.28 0.1804 1.1982 6.64 43.77 1.7195 ED-M 25.21 0.1809 1.1206 6.20 43.25 1.8019 ED-A 25.01 0.1822 1.0594 5.81 44.11 2.0501 ED-T 24.92 0.1829 0.9445 5.16 43.12 1.9966 表 6 煤样的核磁分峰数据
Table 6 NMR peak data of coal samples
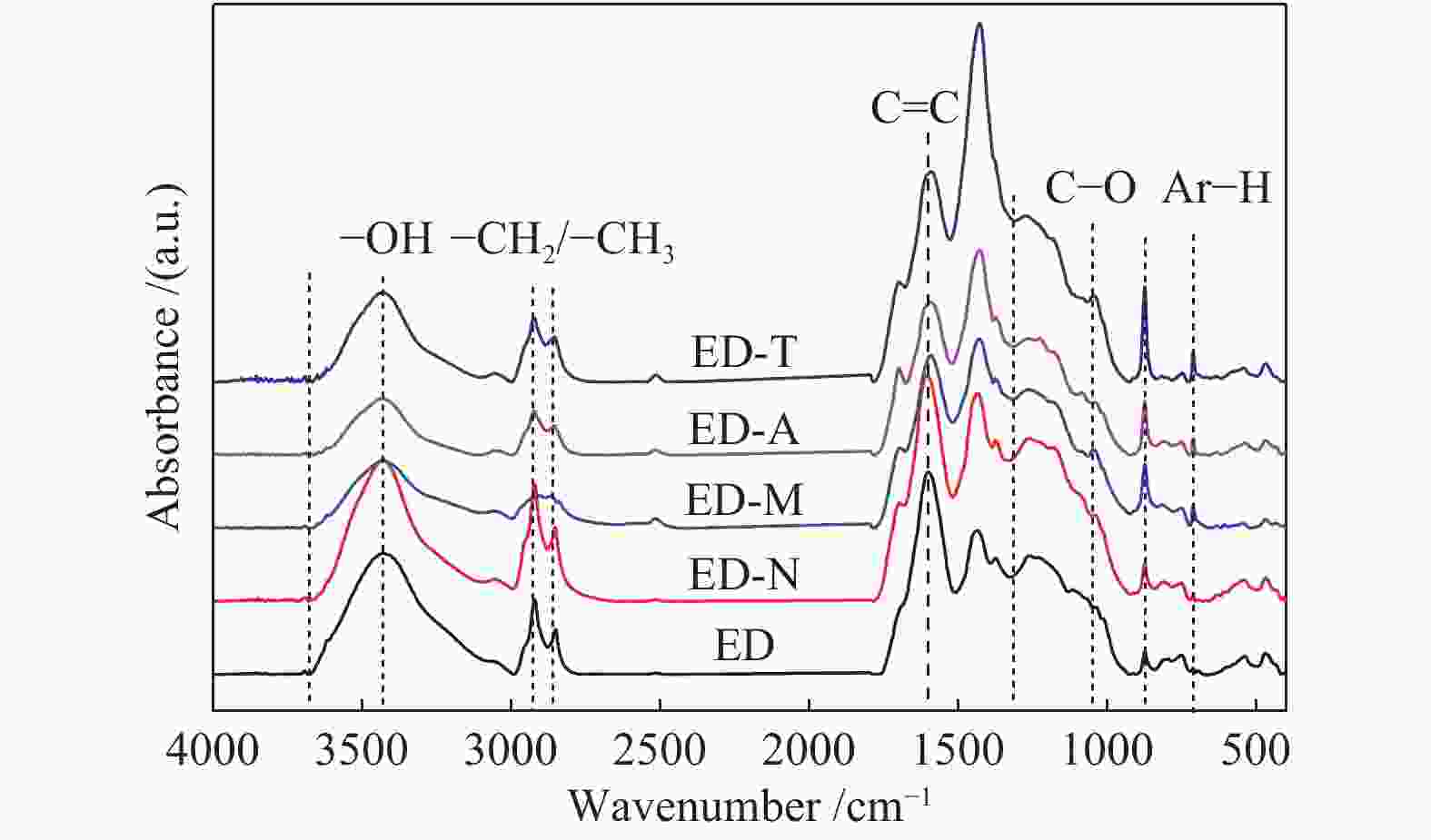
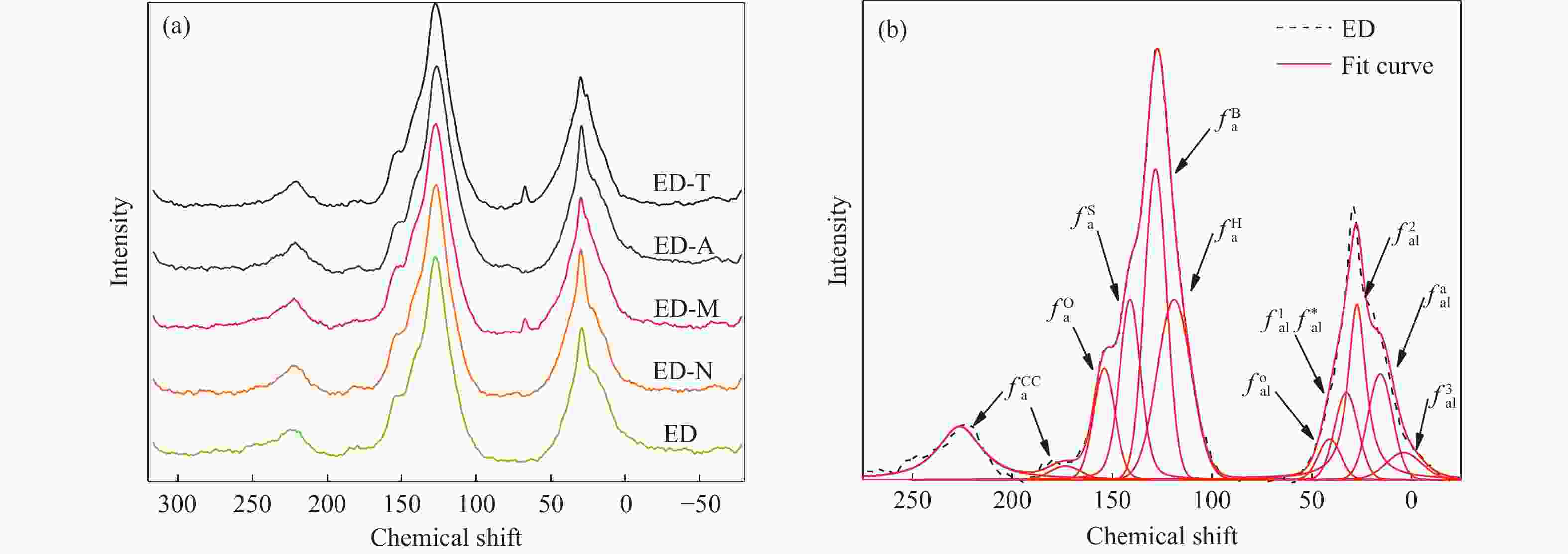
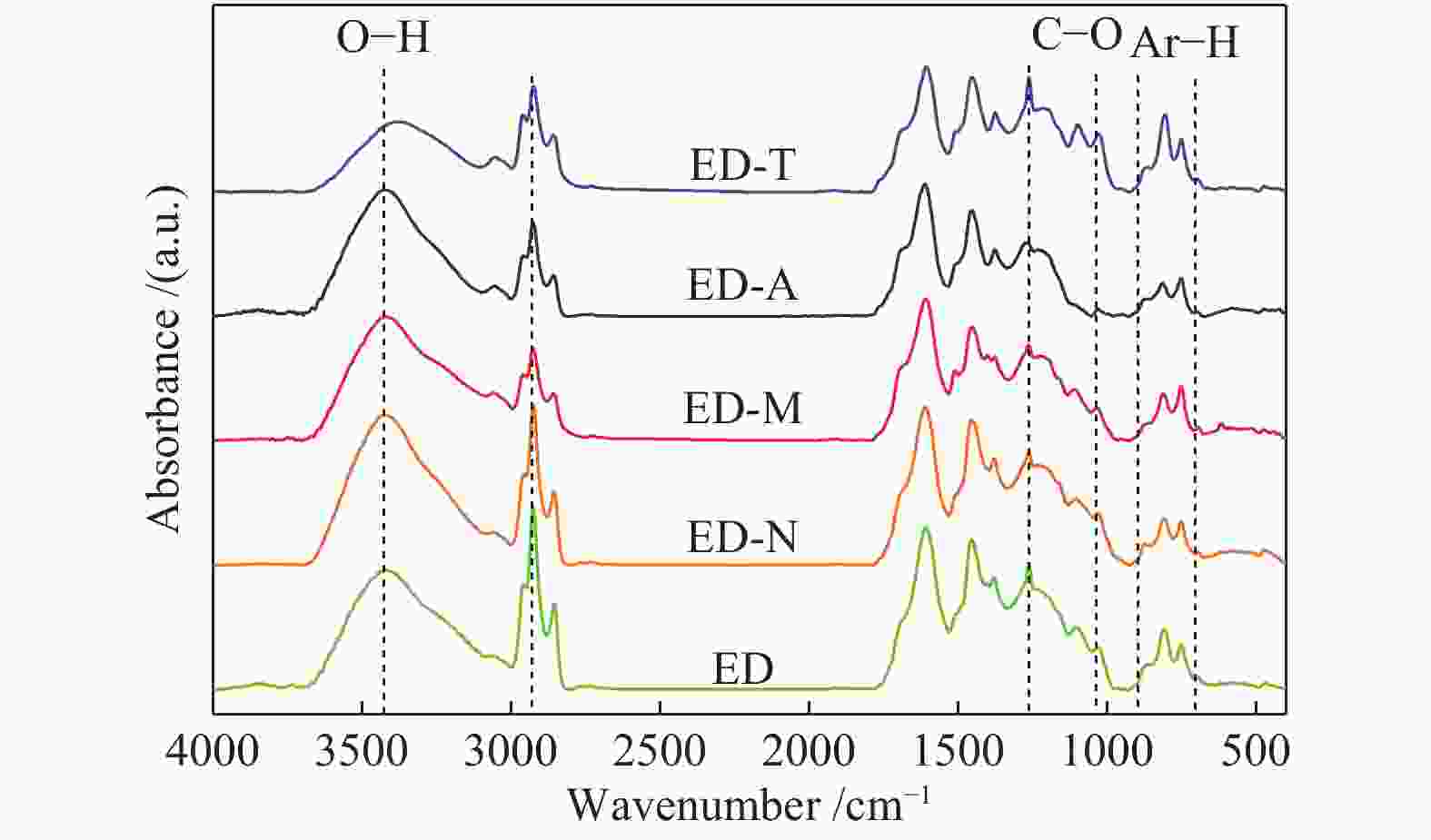
Sample Al-C Ar-C Carboxy-C CH2/CH3 ED 31.99 57.66 10.35 1.23 ED-N 32.03 57.58 10.39 1.37 ED-M 32.30 57.37 10.33 1.20 ED-A 32.76 57.34 9.90 1.19 ED-T 32.99 57.35 9.66 1.17 note: aliphatic carbon content Al-C=$ f_{\mathrm{a}\mathrm{l}}^{3} $+$ f_{\mathrm{a}\mathrm{l}}^{\mathrm{a}} $+$ f_{\mathrm{a}\mathrm{l}}^{2} $+$ f_{\mathrm{a}\mathrm{l}}^{1} $+$ f_{\mathrm{a}\mathrm{l}}^{\mathrm{*}} $+$ f_{\mathrm{a}\mathrm{l}}^{\mathrm{O}} $;aromatic carbon content Ar-C=$ f_{\mathrm{a}}^{\mathrm{H}} $+$ f_{\mathrm{a}}^{\mathrm{B}} $+$ f_{\mathrm{a}}^{\mathrm{S}} $+$ f_{\mathrm{a}}^{\mathrm{O}} $;carbonyl carbon content Carboxy-C=$ f_{\mathrm{a}}^{\mathrm{C}\mathrm{C}} $;CH2/CH3=($ f_{\mathrm{a}\mathrm{l}}^{3} $+$ f_{\mathrm{a}\mathrm{l}}^{\mathrm{a}} $)/$ f_{\mathrm{a}\mathrm{l}}^{2} $ 表 7 煤样热解产物的收率
Table 7 Yield of pyrolysis products of coal samples
Yield w/% ED ED-N ED-M ED-A ED-T Tar 9.32 9.56 11.08 11.81 12.45 Char 71.01 70.82 69.39 68.72 68.14 Gas 16.44 16.47 17.03 17.12 17.25 Water 3.23 3.15 2.50 2.35 2.16 note: the data above were obtained after eliminating the residual solvent in tar, and the gas yield was obtained by subtraction method 表 8 热解气相产物分布
Table 8 Distribution of pyrolysis gas products
Sample Component content/($ {\rm{mmol}} \cdot {\rm{g}}_{{\rm{coal{\text{,}}daf}}}^{-1} $) CO CH4 CO2 C2 C3 H2 ED 1.05 1.04 0.48 0.15 0.07 0.21 ED-N 0.97 0.75 0.49 0.13 0.08 0.12 ED-M 1.12 0.88 0.62 0.17 0.09 0.21 ED-A 1.17 0.68 0.66 0.15 0.07 0.14 ED-T 1.23 0.94 0.65 0.17 0.09 0.16 -
[1] WANG J, LU X, YAO J, LIN W, CUI L. Experimental study of coal topping process in a downer reactor[J]. Ind Eng Chem Res, 2005, 44(3): 463−470. [2] LIU L, KUMAR S, WANG Z, HE Y, LIU J, CEN K. Catalytic effect of metal chlorides on coal pyrolysis and gasification part I. Combined TG-FTIR study for coal pyrolysis[J]. Thermochim Acta,2017,655:331−336. doi: 10.1016/j.tca.2017.07.007 [3] LIU L, YUAN Y, KUMAR S, WANG Z, HE Y, LV Y, LIU J, GUL-E-RANA J, CEN K. Catalytic effect of metal chlorides on coal pyrolysis and gasification part Ⅱ. Effects of acid washing on coal characteristics[J]. Thermochim Acta,2018,666:41−50. doi: 10.1016/j.tca.2018.06.001 [4] 王勇. 溶胀预处理对煤结构及煤液化性能的影响[D]. 大连: 大连理工大学, 2000.WANG Yong. Effect of Solvent Swelling on the Coal Structure and Liquefaction[D]. Dalian: Dalian University of Technology, 2000. [5] ZHU P, LUO A, ZHANG F, LEI Z, ZHANG J, ZHANG J. Effects of extractable compounds on the structure and pyrolysis behaviours of two Xinjiang coal[J]. J Anal Appl Pyrolysis,2018,133:128−135. doi: 10.1016/j.jaap.2018.04.012 [6] LI Y, HUANG S, WU Y, WU S, GAO J. Effects of thermal dissolution in different solvents on structural characteristics and pyrolysis behaviors of lignite[J]. Fuel,2019,241:550−557. doi: 10.1016/j.fuel.2018.12.042 [7] YAN J, BAI Z, BAI J, LI W. Chemical structure and reactivity alterations of brown coals during thermal treatment with aromatic solvents[J]. Fuel Process Technol,2015,137:117−123. doi: 10.1016/j.fuproc.2015.04.009 [8] LI Z, WEI X, YAN H, YU X, ZONG Z. Characterization of soluble portions from thermal dissolution of Zhaotong lignite in cyclohexane and methanol[J]. Fuel Process Technol,2016,151:131−138. doi: 10.1016/j.fuproc.2016.05.029 [9] LIU M, LI J, DUAN Y. Effects of solvent thermal treatment on the functional groups transformation and pyrolysis kinetics of Indonesian lignite[J]. Energy Convers Manage,2015,103:66−72. doi: 10.1016/j.enconman.2015.06.047 [10] TIAN B, QIAO Y Y, TIAN Y Y, XIE K C, LIU Q, ZHOU H F. FTIR study on structural changes of different–rank coals caused by single/multiple extraction with cyclohexanone and NMP/CS2 mixed solvent[J]. Fuel Process Technol,2016,154:210−218. doi: 10.1016/j.fuproc.2016.08.035 [11] XIE K, LI F, FENG J, LIU J. Study on the structure and reactivity of swollen coal[J]. Fuel Process Technol,2000,64(1):241−251. [12] 王娜. 预处理对胜利褐煤微结构及其热化学性能影响的研究[D]. 呼和浩特: 内蒙古工业大学, 2015.WANG Na. Study on the Effect of pretreatment on micro-structure and thermal-chemical performance of Shengli lignite[D]. Huhehot: Inner Mongolia University of Technology, 2015. [13] YAN J, BAI Z, BAI J, GUO Z, LI W. Effects of organic solvent treatment on the chemical structure and pyrolysis reactivity of brown coal[J]. Fuel,2014,128:39−45. doi: 10.1016/j.fuel.2014.03.001 [14] 黄澎. 芳香族溶剂对低阶煤轻度加氢改质的影响[J]. 洁净煤技术,2016,22(3):49−54.HUANG Peng. Effects of aromatic solvents on mild hydro - upgrading reaction of low rank coal[J]. Clean Coal Technol,2016,22(3):49−54. [15] HOU R, BAI Z, HAO P, DAI X, XU J, ZHENG H, GUO Z, KONG L, BAI J, LI W. Effects of temperature and solvents on structure variation of Yunnan lignite in preheating stage of direct liquefaction[J]. Fuel,2019,239:917−925. doi: 10.1016/j.fuel.2018.11.075 [16] 杨振建. 预处理对褐煤结构及低温热解特性的影响[D]. 太原: 太原理工大学, 2016.YANG Zhen-jian. Effect of pretreatment on the structure properties and pyrolysis behaviours of lignite[D]. Taiyuan: Taiyuan University of Technology, 2016. [17] 何超. 低变质煤/溶胀煤原位担载金属离子的热解特性研究[D]. 西安: 西北大学, 2018.HE Chao. Study on the pyrolysis characteristics of the low rank coal/swelling coal in-situ impregnated with metal ions[D]. Xi'an: Northwestern University, 2018. [18] 李沙沙, 盛清涛, 范济民, 申峻. 鑫源煤在有机溶剂中的溶胀特性研究[J]. 煤炭转化,2014,37(1):11−14. doi: 10.3969/j.issn.1004-4248.2014.01.003LI Sha-sha, SHENG Qing-tao, FAN Ji-min, SHEN Jun. Swelling characteristics of Xinyuan coal in organic solvents[J]. Coal Convers,2014,37(1):11−14. doi: 10.3969/j.issn.1004-4248.2014.01.003 [19] 贺新福, 张小琴, 吴红菊, 周安宁. 甲醇蒸气预处理对煤热解产物分布的影响[J]. 煤炭技术,2017,36(8):277−279.HE Xin-fu, ZHANG Xiao-qin, WU Hong-ju, ZHOU An-ning. Effect of methanol vapor pretreatment on yields of coal pyrolysis products[J]. Coal Technol,2017,36(8):277−279. [20] HE C, MIN X, ZHENG H, FAN Y, YAO Q, ZHANG D, TANG X, WAN C, SUN M, MA X, JIA C Q. Study on the volatiles and kinetic of in-situ catalytic pyrolysis of swelling low-rank coal[J]. Energy Fuels,2017,31(12):13558−13571. doi: 10.1021/acs.energyfuels.7b02952 [21] VAN NIEKERK D, MATHEWS J P. Molecular dynamic simulation of coal–solvent interactions in Permian-aged South African coals[J]. Fuel Process Technol, 2011, 92(4):729−734. [22] 陈茺, 许学敏, 高晋生, 颜涌捷, 郭新闻. 氢键在煤大分子溶胀行为中的作用[J]. 燃料化学学报,1997,25(6):45−48.CHEN Chong, XU Xue-min, GAO Jin-sheng, YAN Yong-jie, GUO Xin-wen. Role of hydrogen bond in swelling behavior of coal macromolecules[J]. J Fuel Chem Technol,1997,25(6):45−48. [23] 王晓华, 魏贤勇. 煤的溶剂萃取研究进展[J]. 现代化工,2003,23(7):19. doi: 10.3321/j.issn:0253-4320.2003.07.005WANG Xiao-hua, WEI Xian-yong. Advances in coal solvent extraction[J]. Mod Chem Ind,2003,23(7):19. doi: 10.3321/j.issn:0253-4320.2003.07.005 [24] 吉庆军, 黄克权. 甲醇处理煤的微孔性质及反应性研究[J]. 燃料化学学报,1991,19(1):49−56.JI Qing-jun, HUANG Ke-quan. Study on microporous properties and reactivity of coal treated with methanol[J]. J Fuel Chem Technol,1991,19(1):49−56. [25] SHI L, LIU Q, GUO X, WU W, LIU Z. Pyrolysis behavior and bonding information of coal — A TGA study[J]. Fuel Process Technol,2013,108:125−132. doi: 10.1016/j.fuproc.2012.06.023 [26] MENG, HU H, ZHANG Q, LI X, WU B. Pyrolysis behaviors of tumuji oil sand by thermogravimetry (TG) and in a fixed bed reactor[J]. Energy Fuels,2007,21(4):2245−2249. doi: 10.1021/ef070048z [27] 刘劲松, 冯杰, 李凡, 谢克昌. 溶胀作用在煤结构与热解研究中的应用[J]. 煤炭转化,1998,21(2):3−5.LIU Jin-song, FENG jie, LI fan, XIE Ke-chang. Application of swelling in the study of coal structure and pyrolysis[J]. Coal Convers,1998,21(2):3−5. [28] HE Q, HUANG Y, DING L, GUO Q, GONG Y, YU G. Effect of partial rapid pyrolysis on bituminous properties: from structure to reactivity[J]. Energy Fuels,2020,34(5):5476−5484. doi: 10.1021/acs.energyfuels.9b04439 [29] ZHAO Y, QIU P, CHEN G, PEI J, SUN S, LIU L, LIU H. Selective enrichment of chemical structure during first grinding of Zhundong coal and its effect on pyrolysis reactivity[J]. Fuel (Guildford),2017,189:46−56. doi: 10.1016/j.fuel.2016.10.083 [30] ZOU L, JIN L, LI Y, ZHU S, HU H. Effect of tetrahydrofuran extraction on lignite pyrolysis under nitrogen[J]. J Anal Appl Pyrolysis, 2015, 112:113−120. [31] ZHANG X P, ZHANG C, PENG T, LI X, FANG Q Y, GANG C. Effects of hydrothermal upgrading on the physicochemical structure and gasification characteristics of Zhundong coal[J]. Fuel Process Technol, 2018, 172: 200−208. [32] 常慧. 预处理对低变质煤热解特性影响的研究[D]. 西安: 西北大学, 2019.CHANG Hui. Study on the pyrolysis characteristic of low rank coals with pretreatments[D]. Xi'an: Northwestern University, 2019. [33] IBARRA J, MUÑOZ E, MOLINER R. FTIR study of the evolution of coal structure during the coalification process[J]. Org Geochem,1996,24(6):725−735. [34] 刘鹏. 水热处理对低阶煤碳结构及热解行为的作用机理研究[D]. 上海: 华东理工大学, 2016.LIU Peng. Research on mechanism of hydro thermal on carbon structure and pyrolysis behavior of low rank coal[D]. Shanghai: East China University of Science and Technology, 2016. [35] LIU J, JIANG X, SHEN J, ZHANG H. Pyrolysis of superfine pulverized coal. Part 2. Mechanisms of carbon monoxide formation[J]. Energy Convers Manage,2014,87:1039−1049. doi: 10.1016/j.enconman.2014.07.055 [36] HAN L, WANG Q, MA Q, YU C, LUO Z, CEN K. Influence of CaO additives on wheat-straw pyrolysis as determined by TG-FTIR analysis[J]. J Anal Appl Pyrolsis,2010,88(2):199−206. doi: 10.1016/j.jaap.2010.04.007 [37] SUN M, ZHANG D, YAO Q, LIU Y, SU X, JIA C Q, HAO Q, MA X. Separation and composition analysis of GC/MS analyzable and unanalyzable parts from coal tar[J]. Energy Fuels,2018,32(7):7404−7411. doi: 10.1021/acs.energyfuels.8b01054 -




 下载:
下载: