Synthesis of small crystal NiY zeolites and their catalytic performance in hydrocracking
-
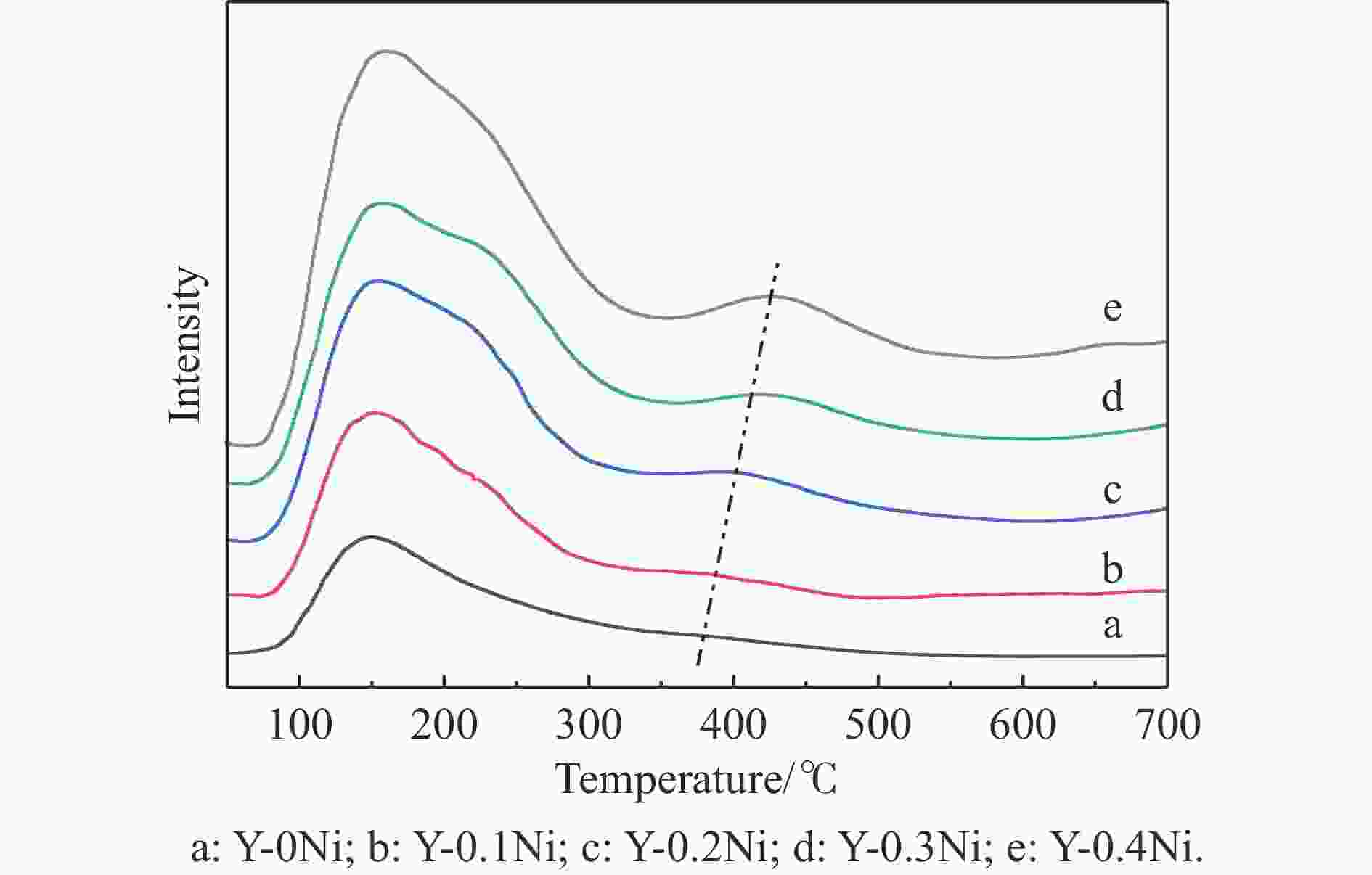
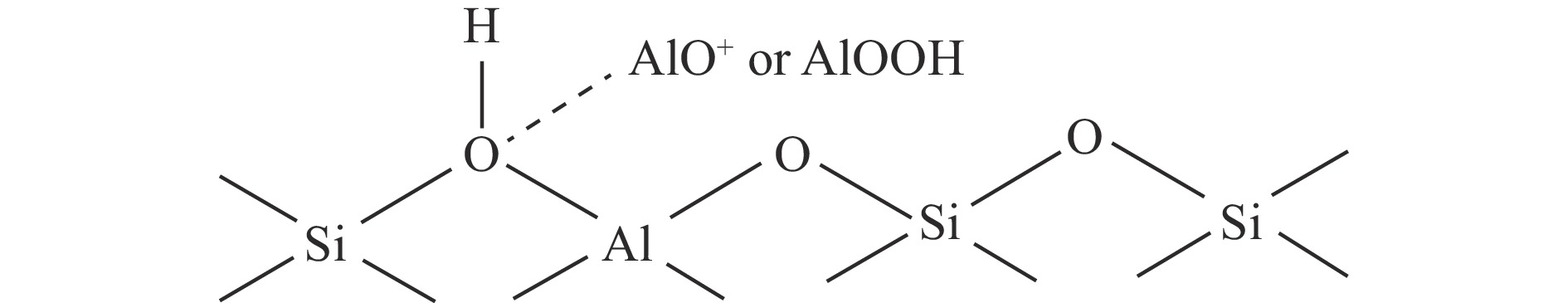
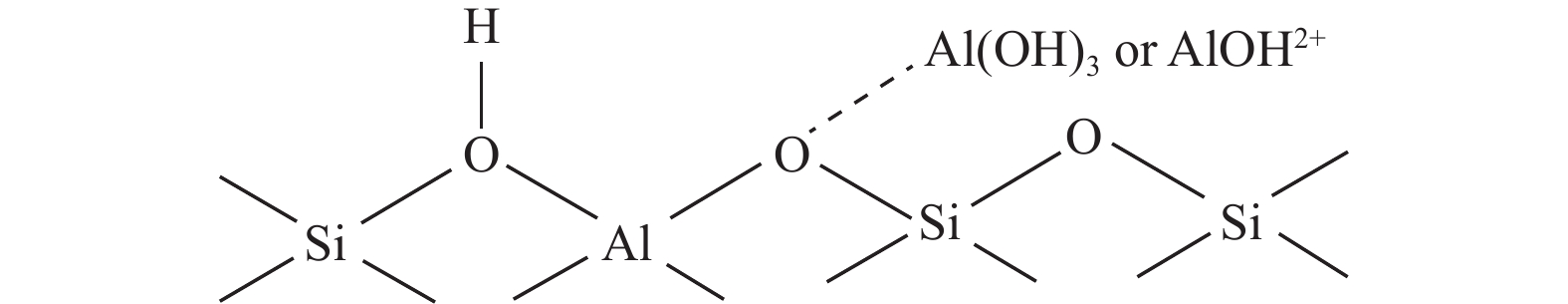
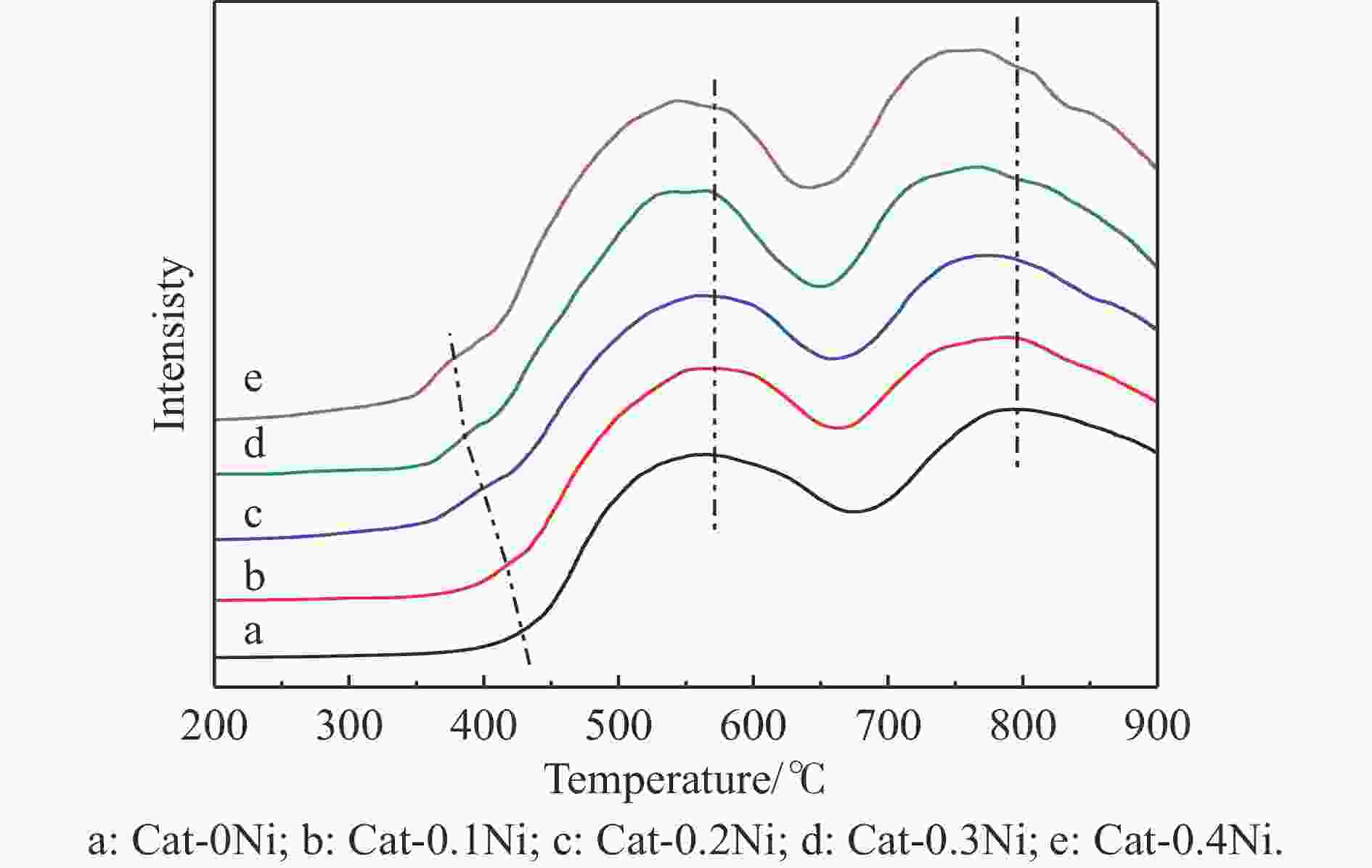
摘要: 通过在小晶粒Y分子筛合成的过程中原位引入Ni源合成了一系列不同Ni掺入量的小晶粒Y-xNi分子筛,将活性金属Ni预浸渍到Y分子筛的骨架中。将Y-xNi分子筛和ASA混合物作为载体并采用等体积浸渍法负载Ni和W,制备了系列Cat-xNi加氢裂化催化剂,以正十六烷为反应物,探究其加氢裂化催化反应性能。采用扫描电子显微镜(SEM)、X射线衍射(XRD)、N2吸附-脱附、氨气程序升温脱附(NH3-TPD)、氢气程序升温还原(H2-TPR)、透射电子显微镜(TEM)和X射线光电子能谱(XPS)等表征手段分析了Ni的掺入对Y分子筛及Cat-xNi催化剂理化性质的影响。结果表明,Ni主要取代Al引入Y分子筛骨架;在Y分子筛中适量掺入Ni会提高Y分子筛的相对结晶度以及Brønsted酸和Lewis酸位点的数量,但过量的Ni掺入不利于Y分子筛的结晶。Ni的掺入削弱了金属与载体间的相互作用,提高了活性金属的硫化度及NiWS活性相的堆积数及分散度,调节了催化剂上金属中心与酸中心的匹配。催化性能评价结果显示,由于Ni改性能同时增加Brønsted酸中心与NiWS活性中心数量,增强金属中心与酸中心之间的协同作用,因而在提高正十六烷加氢裂化活性的同时可避免其过度裂化,获得较高的中间馏分产物(C8–C12)选择性及收率。在360 °C反应条件下,与Cat-0Ni催化剂相比,Cat-0.2Ni催化剂具有较高的n-C16转化率和C8–C12产物收率(达65.4%)。综上可知,采用原位合成法将活性金属Ni预浸渍在Y分子筛上可以有效调节裂化活性中心与加氢活性中心之间的平衡,从而提高其催化活性和中间馏分产物的收率。Abstract: A series of small crystal Y-xNi zeolites with different amounts of Ni doping were synthesized by in situ introducing the Ni precursors during the synthesis, through which the active Ni metal was incorporated into the framework of the Y zeolites. With the mechanical mixture of Y-xNi zeolites and amorphous silica-alumina (ASA) as the support, a series of Cat-xNi catalysts were prepared through loading the Ni and W components by incipient wet impregnation and the catalytic performance of Cat-xNi in the hydrocracking of n-hexadecane was then investigated. In addition, the effect of Ni doping on the physicochemical properties of Y zeolite and Cat-xNi catalysts was elucidated with the help of scanning electron microscopy (SEM), X-ray diffraction (XRD), N2-adsorption desorption, NH3 temperature programmed desorption (NH3-TPD), H2 temperature programmed reduction (H2-TPR), transmission electron microscopy (TEM), X-ray photoelectron spectroscopy (XPS), and so on. The results indicate that Ni can replace Al to enter the framework of the Y zeolites. The incorporation of appropriate amount of Ni into the Y zeolites can increase their relative crystallinity and the number of Brønsted and Lewis acid sites; however, excessive Ni incorporation is detrimental to the crystallization of Y zeolite and excessive non-framework Ni species will cover the surface Brønsted acid sites. Moreover, Ni doping can weaken the metal-support interaction, increase the sulfation extent of the active metal and the stacking number and dispersion of the active NiWS phase, and then improve the matching between the metal and acid sites on the Cat-xNi catalysts. As a results, in comparison with the counterpart Cat-0Ni catalyst, the Cat-xNi catalysts display more Brønsted acid sites and active NiWS sites as well as improved the synergy between the metal sites and acid sites, which can enhance the conversion of n-hexadecane whereas inhibit the over-cracking, and then booster the yield of the middle distillate products (C8–C12). In particular, for the n-hexadecane hydrocracking at 360 °C, the Cat-0.2Ni catalyst exhibits a C8–C12 product yield of 65.4%, with a much higher n-C16 conversion than the Cat-0Ni counterpart. All these suggest that the pre-impregnation of active metal Ni on the Y zeolites can effectively regulate the balance between the hydrogenation and cracking performance and improve the catalytic activity and the yield of middle distillate products in the hydrocracking of paraffins.
-
Key words:
- Y zeolite /
- in-situ Ni modification /
- catalyst /
- hydrocracking /
- middle distillate
-
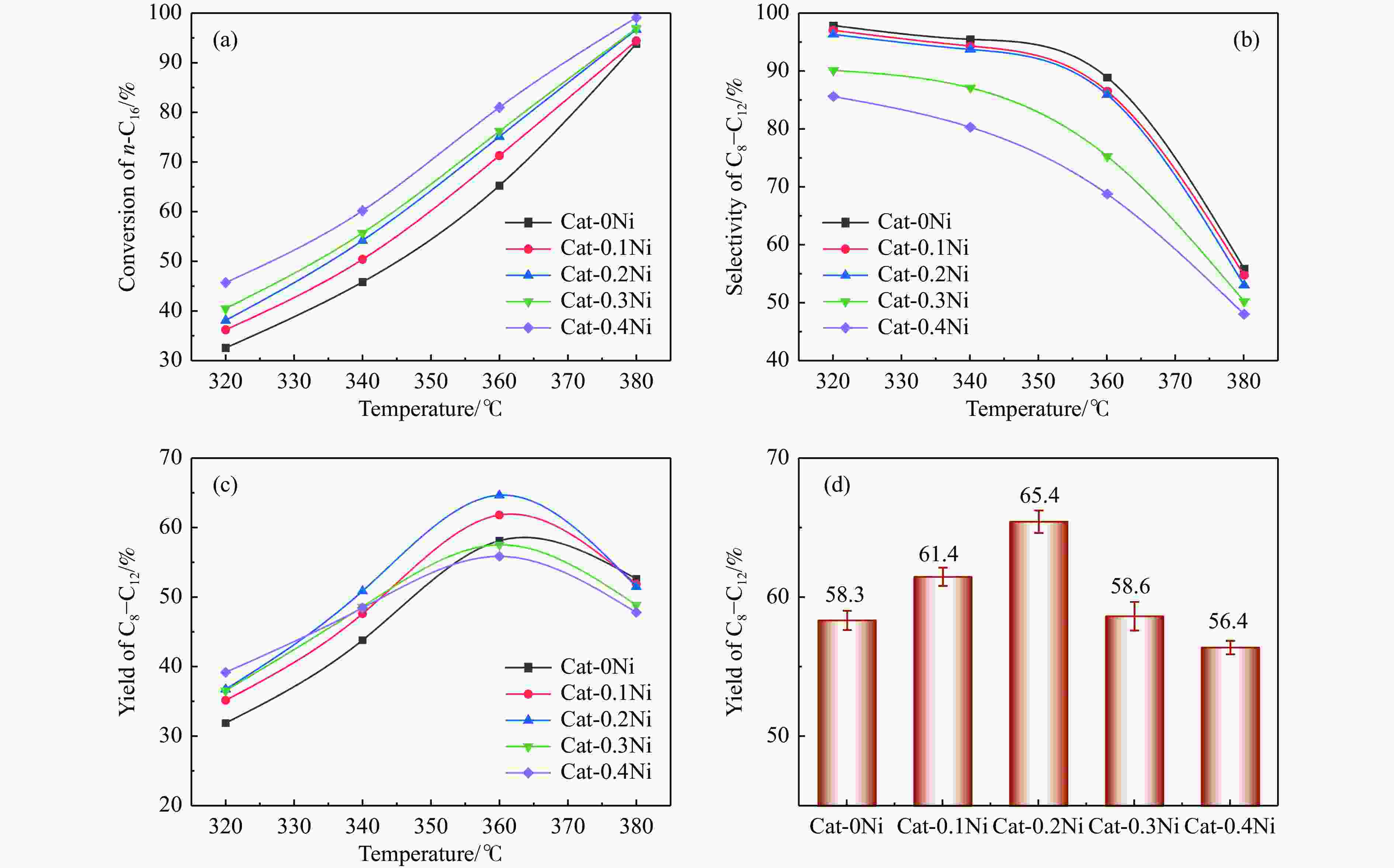
图 11 Cat-xNi系列硫化催化剂在不同反应温度下n-C16的转化率(a)、C8−C12的选择性(b)和收率(c)以及在360 ℃条件下重复三次实验后不同催化剂的C8−C12的收率(d)
Figure 11 Conversion of n-C16 (a), selectivity of C8−C12 (b) and yield of C8−C12 (c) over the sulfided NiW catalysts at different reaction temperature; the average yield of C8–C12 (with the error bar) derived from three repetitive tests at 360 °C for different sulfided Cat-xNi catalysts (d)
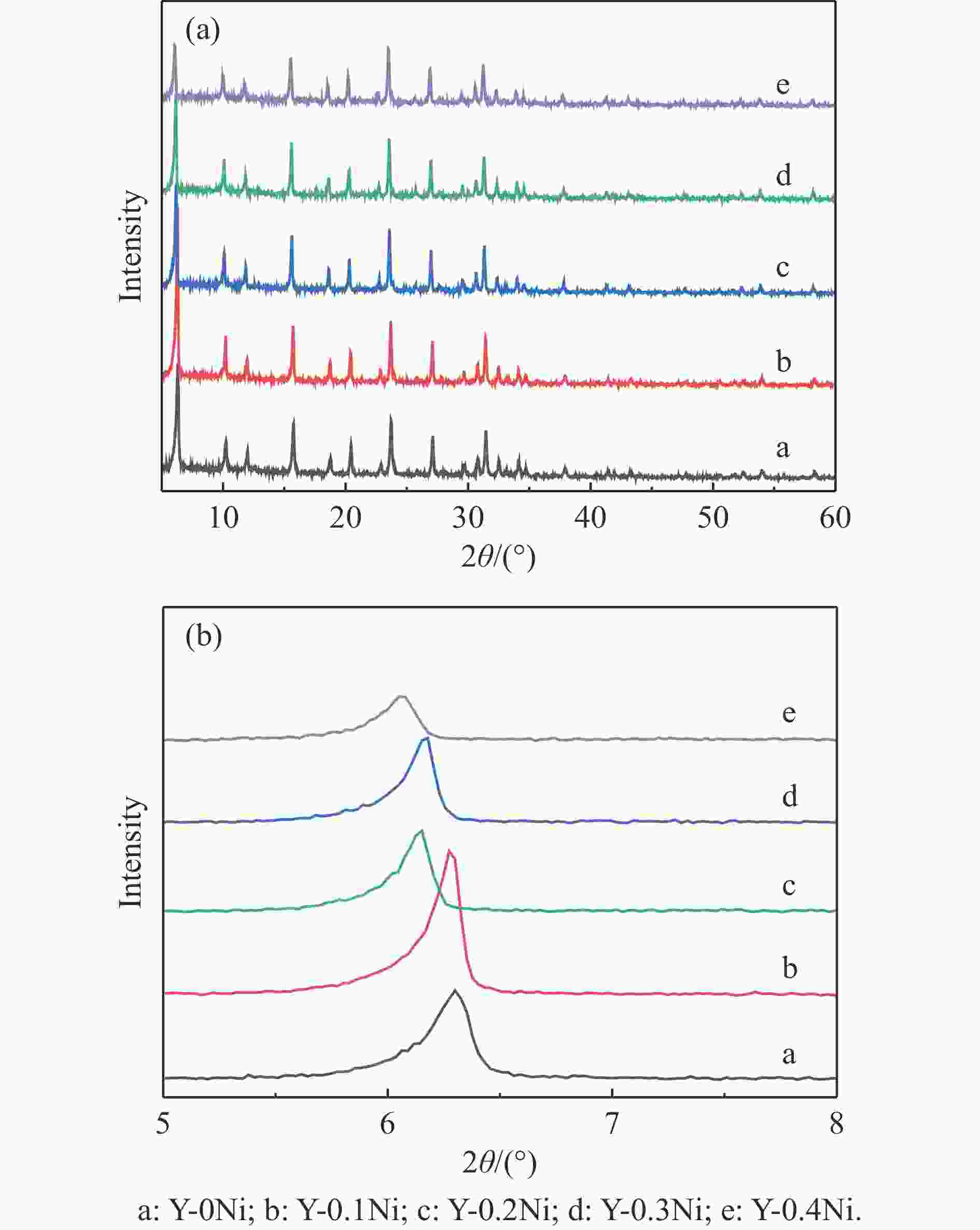
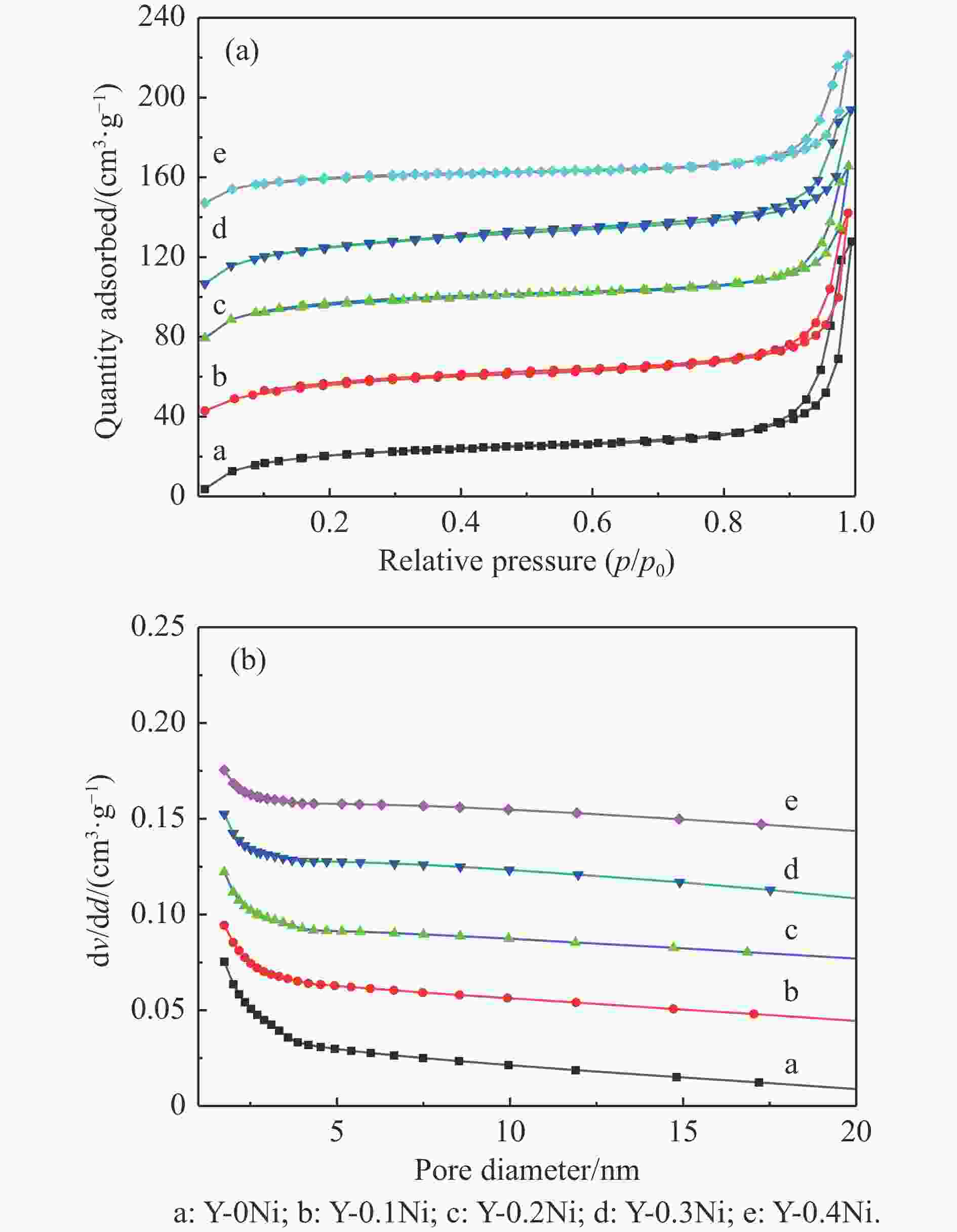
表 1 不同Ni含量杂原子Y-xNi分子筛的理化性质
Table 1 Physicochemical properties of the synthesized Y-xNi zeolites with different Ni contents
Sample n(NiO/Al2O3) Relative crystallinityb/
%Crystal sizec/
nmSBET/
(m2·g−1)vtotal/
(cm3·g−1)Average pore diameter/
nmtheoretical actuala Y-0Ni 0 0 100 125 634.4 0.64 6.7 Y-0.1Ni 0.1 0.09 102 136 612.1 0.62 6.4 Y-0.2Ni 0.2 0.17 97 141 600.6 0.61 6.1 Y-0.3Ni 0.3 0.26 92 146 588.4 0.59 5.9 Y-0.4Ni 0.4 0.37 88 152 560.2 0.55 5.1 a: Calculated from XRF results (test sample was HY-xNi zeolites after ion-exchange); b: Calculated from XRD results and Eq. (1); c: Statistically calculated from SEM image results. 表 2 Y-xNi系列分子筛上各物种分峰拟合
Table 2 XPS deconvolution results of the synthesized Y-xNi zeolites
Catalyst Si−O−Si/% Si−O−H/% Si−O−Al/% Ni−O/% Ni2+/% Ni3+/% Y-0Ni 39.6 28.1 32.3 − − − Y-0.1Ni 37.3 25.8 23.5 13.4 43.0 57.0 Y-0.2Ni 38.1 29.1 17.6 15.2 46.7 53.3 Y-0.3Ni 38.8 30.0 13.4 17.8 50.1 49.9 Y-0.4Ni 39.1 30.8 10.9 19.2 55.4 44.6 表 3 Y-xNi系列分子筛的酸类型及酸量
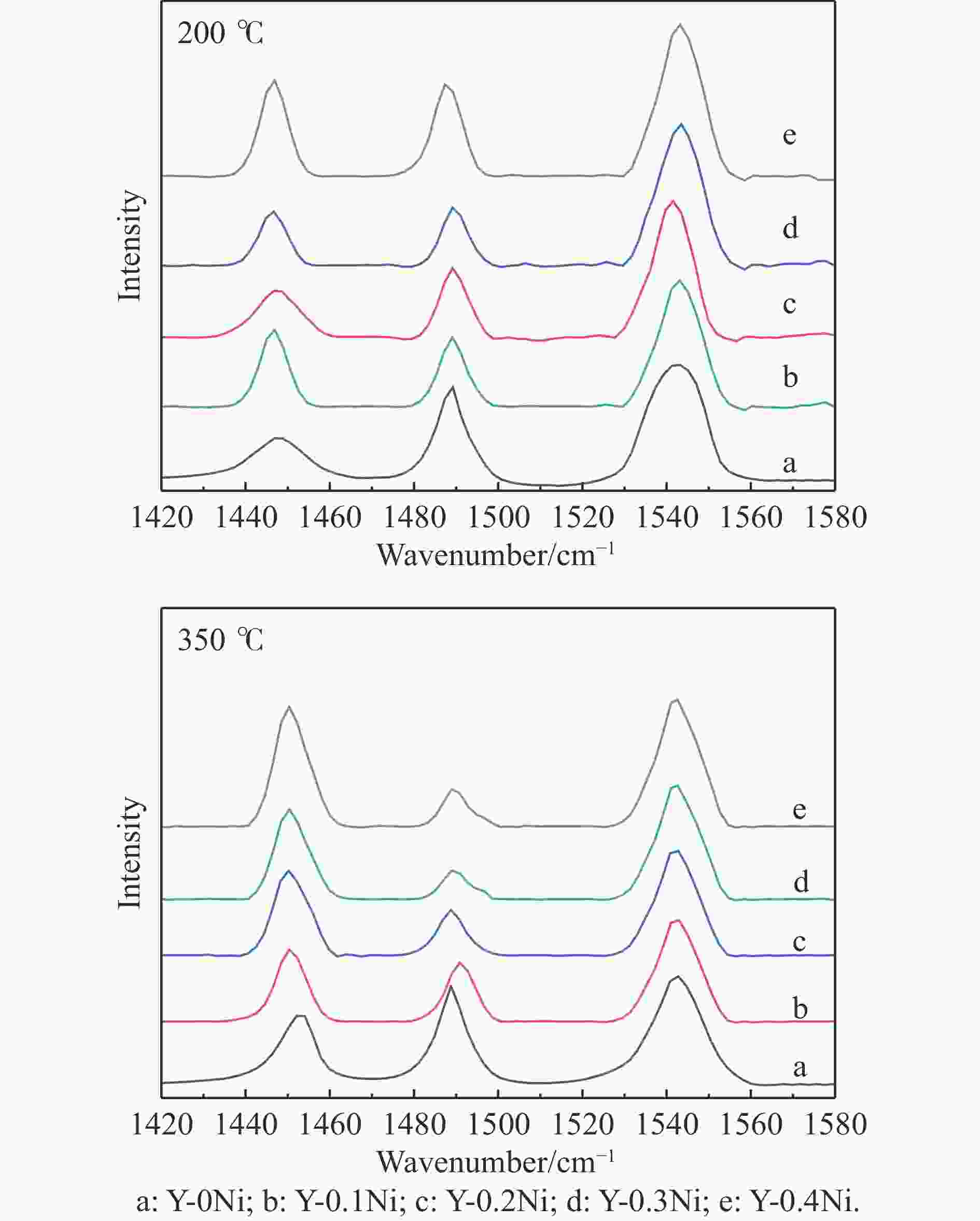
Table 3 Acidity of the synthesized Y-xNi zeolites
Sample Acidity/(μmol·g−1) total acid sites (200 ℃) medium and strong acid sites (350 ℃) totald L B L+B B/L L B L+B B/L Y-0Ni 96 220 316 2.3 45 158 206 3.5 414 Y-0.1Ni 125 233 358 1.9 63 164 227 2.6 452 Y-0.2Ni 162 242 404 1.5 88 171 259 1.9 486 Y-0.3Ni 193 254 447 1.3 107 182 289 1.7 522 Y-0.4Ni 228 257 485 1.1 124 189 313 1.5 591 d: Calculated from NH3-TPD results. 表 4 硫化催化剂上WS2的平均长度、堆垛层数、fw值
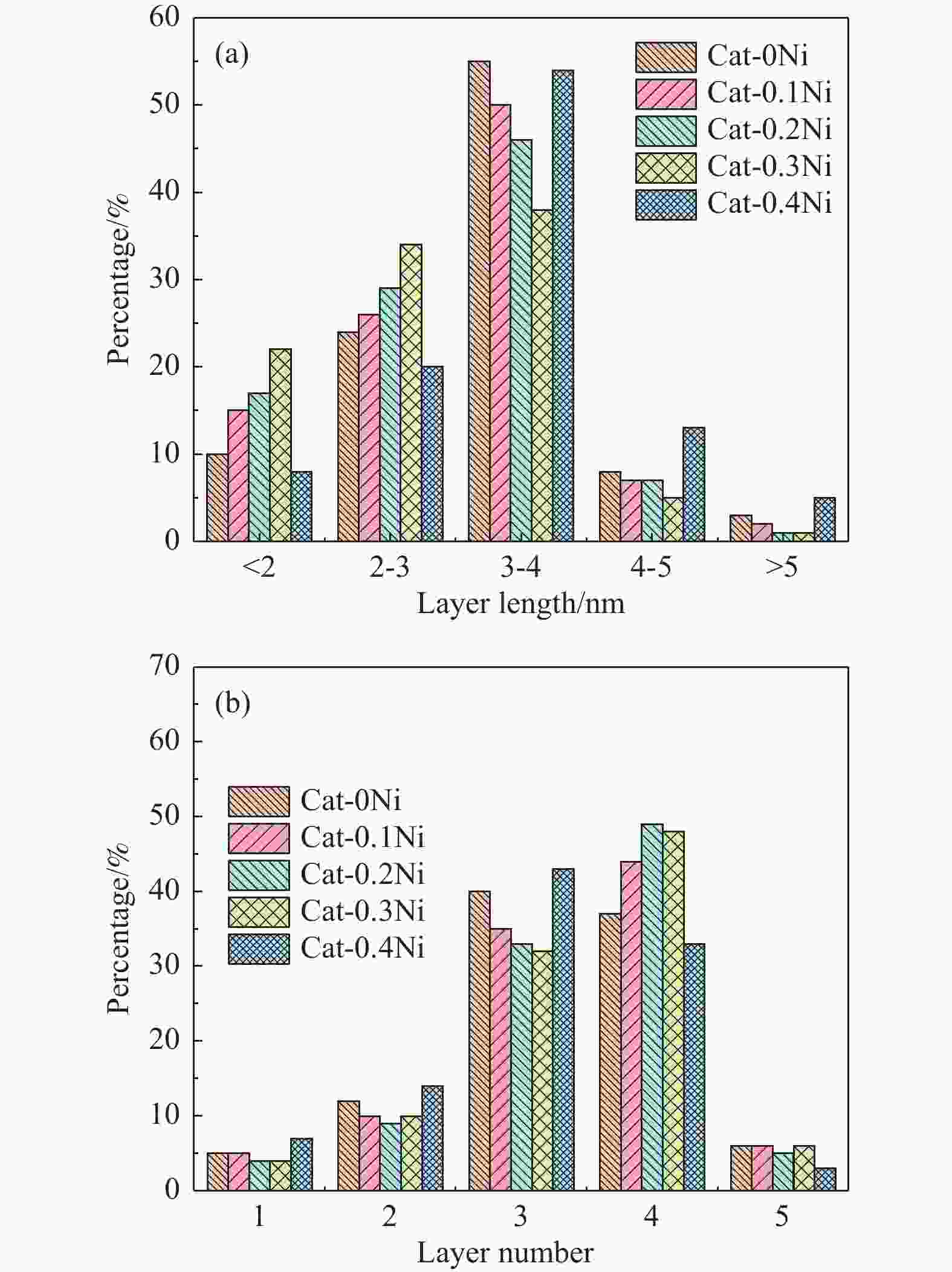
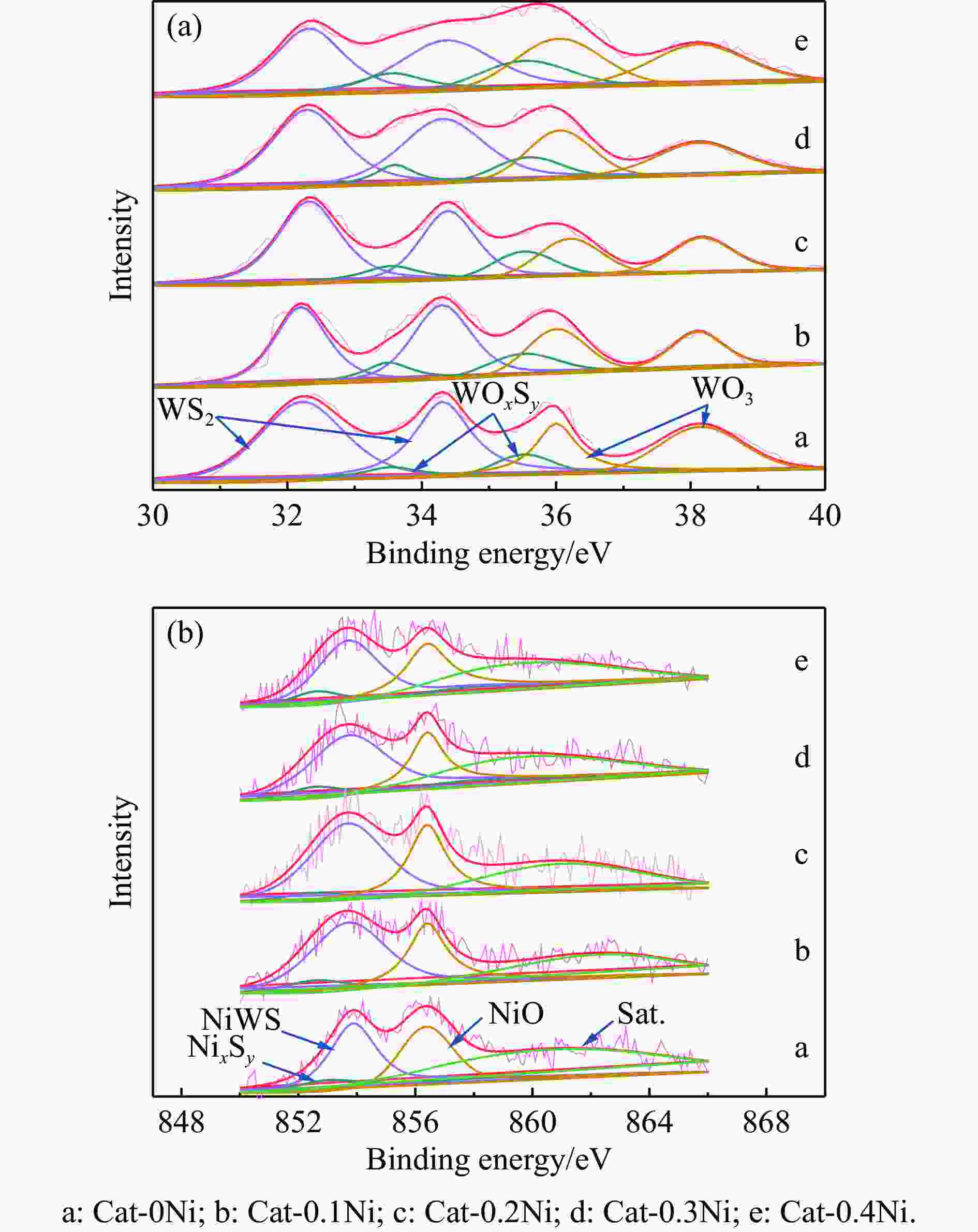
Table 4 Average length, stacking number, fw values of WS2 slabs for the sulfided Cat-xNi catalysts
Catalyst Length/nm Stacking number fw Cat-0Ni 3.65 3.54 0.34 Cat-0.1Ni 3.61 3.55 0.35 Cat-0.2Ni 3.56 3.58 0.36 Cat-0.3Ni 3.48 3.58 0.37 Cat-0.4Ni 3.65 3.47 0.32 表 5 硫化NiW系列催化剂上各物种分峰拟合结果
Table 5 XPS deconvolution results of the sulfide NiW series catalysts
Catalyst WS2/% WOxSy/% WO3/% Nisulfidation/% NiWS/% NixSy/% NiO/% Cat-0Ni 68.1 10.2 21.7 70.1 58.6 11.5 32.9 Cat-0.1Ni 68.7 11.4 19.9 68.3 59.1 9.2 31.7 Cat-0.2Ni 69.0 11.8 19.2 69.5 59.7 9.8 30.5 Cat-0.3Ni 69.2 12.1 18.7 70.2 60.8 9.4 29.8 Cat-0.4Ni 51.8 9.8 38.4 63.6 52.9 10.7 36.4 表 6 360 ℃下五种催化剂的n-C16加氢裂化活性数据
Table 6 Results of the n-C16 hydrocracking reaction over five catalysts at 360 ℃
Catalyst ka/(mol·g–1·h−1) TOFa,b/h−1 n-C16 conversion/% C8−C12 selectivity/% C8−C12 yield/% Cat-0Ni 1.09×10−2 25.79 65.2 88.9 58.1 Cat-0.1Ni 1.22×10−2 27.32 71.3 86.6 61.7 Cat-0.2Ni 1.31×10−2 28.05 75.1 86.0 64.6 Cat-0.3Ni 1.44×10−2 29.44 76.2 75.4 57.5 Cat-0.4Ni 1.68×10−2 38.19 81.0 68.9 55.8 a: The rate constant k and TOF values were determined at an n-C16 conversion of about 30% by changing the LHSV for reaction; b: The TOF value is number of n-C16 molecules converted per hour per mole of W atoms. -
[1] BROWNING B E, PITAULT I, COUENNE F, et al. Effects of bifunctional catalyst geometry on vacuum gas oil hydrocracking conversion and selectivity for middle distillate[J]. Ind Eng Chem Res,2018,57(49):16579−16592. doi: 10.1021/acs.iecr.8b03003 [2] LABABIDI H M S, CHEDADEH D, RIAZI M R, et al. Prediction of product quality for catalytic hydrocracking of vacuum gas oil[J]. Fuel,2011,90(2):719−727. doi: 10.1016/j.fuel.2010.09.046 [3] DING L, SITEPU H, AL-BOGAMI S A, et al. Effect of zeolite-Y modification on crude-oil direct hydrocracking[J]. ACS Omega,2021,6(43):28654−28662. doi: 10.1021/acsomega.1c03029 [4] SAHU R, SONG B J, IM J S, et al. A review of recent advances in catalytic hydrocracking of heavy residues[J]. J Ind Eng Chem,2015,27:12−24. doi: 10.1016/j.jiec.2015.01.011 [5] FRANCIS J, GUILLON E, BATS N, et al. Design of improved hydrocracking catalysts by increasing the proximity between acid and metallic sites[J]. Appl Catal A: Gen,2011,409−410:140−147. doi: 10.1016/j.apcata.2011.09.040 [6] WANG W, LIU C J, WU W. Bifunctional catalysts for the hydroisomerization of n-alkanes: The effects of metal-acid balance and textural structure[J]. Catal Sci Technol,2019,9(16):4162−4187. doi: 10.1039/C9CY00499H [7] LI D, NISHIJIMA A, MORRIS D E, et al. Activity and structure of hydrotreating Ni, Mo, and Ni-Mo sulfide catalysts supported on γ-Al2O3-USY zeolite[J]. J Catal,1999,188(1):111−124. doi: 10.1006/jcat.1999.2663 [8] RANA M S, ANCHEYTA J, MAITY S K, et al. Heavy crude oil hydroprocessing: A zeolite-based CoMo catalyst and its spent catalyst characterization[J]. Catal Today,2008,130(2):411−420. [9] CUI Q, WANG S, WEI Q, et al. Synthesis and characterization of Zr incorporated small crystal size Y zeolite supported NiW catalysts for hydrocracking of vacuum gas oil[J]. Fuel,2019,237:597−605. doi: 10.1016/j.fuel.2018.10.040 [10] BATALHA N, PINARD L, BOUCHY C, et al. n-Hexadecane hydroisomerization over Pt-HBEA catalysts. Quantification and effect of the intimacy between metal and protonic sites[J]. J Catal,2013,307:122−131. doi: 10.1016/j.jcat.2013.07.014 [11] DING L, ZHENG Y, YANG H, et al. LCO hydrotreating with Mo-Ni and W-Ni supported on nano- and micro-sized zeolite beta[J]. Appl Catal A: Gen,2009,353(1):17−23. doi: 10.1016/j.apcata.2008.10.023 [12] 黄坚, 李先锋, 谢军, 等. 离子交换法改性的NaY分子筛对吸附含硫VOCs的性能提升[J]. 环境工程学报,2022,16(10):3335−3345. doi: 10.12030/j.cjee.202204053HUANG Jian, LI Xianfeng, XIE Jun, et al. Improvement of the adsorption performance of sulfur-containing VOCs by NaY zeolite modified by ion exchange method[J]. Chin J Environ Eng,2022,16(10):3335−3345. doi: 10.12030/j.cjee.202204053 [13] GU D, WANG H, LI Z, et al. Green synthesis of anthraquinone by one-pot method with Ni-modified Hβ zeolite[J]. Mol Catal,2023,538:112969. doi: 10.1016/j.mcat.2023.112969 [14] JABŁOŃSKA M, KRÓL A, KUKULSKA-ZAJAC E, et al. Zeolite Y modified with palladium as effective catalyst for selective catalytic oxidation of ammonia to nitrogen[J]. J Catal,2014,316:36−46. doi: 10.1016/j.jcat.2014.04.022 [15] VIEIRA L H, CARVALHO K T, URQUIETA-GONZÁLEZ E A, et al. Effects of crystal size, acidity, and synthesis procedure on the catalytic performance of gallium and aluminum MFI zeolites in glycerol dehydration[J]. J Mol Catal A: Chem , 2016, 422 : 148−157. [16] ZHOU W, LIU M, ZHOU Y, et al. 4, 6-Dimethyldibenzothiophene hydrodesulfurization on nickel-modified USY-supported NiMoS catalysts: effects of modification method[J]. Energy Fuels,2017,31(7):7445−7455. doi: 10.1021/acs.energyfuels.7b01113 [17] LIN T J, MENG X, SHI L. Ni-exchanged Y-zeolite: An efficient heterogeneous catalyst for acetylene hydrocarboxylation[J]. Appl Catal A: Gen,2014,485:163−171. doi: 10.1016/j.apcata.2014.07.036 [18] 于悦, 张佳, 吕忠武, 等. Ni改性HZSM-5催化剂对混合C4中异丁烯齐聚的影响[J]. 工业催化,2023,31(10):63−68. doi: 10.3969/j.issn.1008-1143.2023.10.009YU Yue, ZHANG Jia, LÜ Zhongwu, et al. Effect of Ni - modified HZSM-5 catalysts on oligomerization of isobutylene in mixed C4[J]. Ind Catal,2023,31(10):63−68. doi: 10.3969/j.issn.1008-1143.2023.10.009 [19] CUI Q, ZHOU Y, WEI Q, et al. Performance of Zr-and P-modified USY-based catalyst in hydrocracking of vacuum gas oil[J]. Fuel Process Technol,2013,106:439−446. doi: 10.1016/j.fuproc.2012.09.010 [20] WANG J, LIU P, BORONAT M, et al. Organic-Free synthesis of zeolite Y with high Si/Al ratios: Combined strategy of in situ hydroxyl radical assistance and post‐synthesis treatment[J]. Angew Chem,2020,132(39):17378−17381. doi: 10.1002/ange.202005715 [21] TAYEB K B, LAMONIER C, LANCELOT C, et al. Study of the active phase of NiW hydrocracking sulfided catalysts obtained from an innovative heteropolyanion based preparation[J]. Catal Today,2010,150(3/4):207−212. doi: 10.1016/j.cattod.2009.07.094 [22] ZHOU W, WEI Q, ZHOU Y, et al. Hydrodesulfurization of 4, 6-dimethyldibenzothiophene over NiMo sulfide catalysts supported on meso-microporous Y zeolite with different mesopore sizes[J]. Appl Catal B: Environ,2018,238:212−224. doi: 10.1016/j.apcatb.2018.07.042 [23] LI X, WANG A, EGOROVA M, et al. Kinetics of the HDS of 4, 6-dimethyldibenzothiophene and its hydrogenated intermediates over sulfided Mo and NiMo on γ-Al2O3[J]. J Catal,2007,250(2):283−293. doi: 10.1016/j.jcat.2007.06.005 [24] CALEMMA V, PERATELLO S, PEREGO C. Hydroisomerization and hydrocracking of long chain n-alkanes on Pt/amorphous SiO2-Al2O3 catalyst[J]. Appl Catal A: Gen,2000,190(1/2):207−218. doi: 10.1016/S0926-860X(99)00292-6 [25] MURAOKA K, SADA Y, SHIMOJIMA A, et al. Tracking the rearrangement of atomic configurations during the conversion of FAU zeolite to CHA zeolite[J]. Chem Sci,2019,10(37):8533−8540. doi: 10.1039/C9SC02773D [26] MENG B, REN S, LI Z, et al. A facile organic-free synthesis of high silica zeolite Y with small crystal in the presence of Co2+[J]. Microporous Mesoporous Mater,2021,323:111248. doi: 10.1016/j.micromeso.2021.111248 [27] LIU S, REN J, ZHU S, et al. Synthesis and characterization of the Fe-substituted ZSM-22 zeolite catalyst with high n-dodecane isomerization performance[J]. J Catal,2015,330:485−496. doi: 10.1016/j.jcat.2015.07.027 [28] HUANG W, WEI Q, ZHOU Y, et al. Hydrotreating of diesel fuel over in-situ nickel modified Y zeolite supported Ni-Mo-S catalyst[J]. Catal Today,2023,407:135−145. doi: 10.1016/j.cattod.2022.02.002 [29] WEI B, JIN L, WANG D, et al. Effect of different acid-leached USY zeolites on in-situ catalytic upgrading of lignite tar[J]. Fuel,2020,266:117089. doi: 10.1016/j.fuel.2020.117089 [30] CHOO M-Y, JUAN J C, OI L E, et al. The role of nanosized zeolite Y in the H2-free catalytic deoxygenation of triolein[J]. Catal Sci Technol,2019,9(3):772−782. doi: 10.1039/C8CY01877D [31] LV P, YAN L, LIU Y, et al. Catalytic conversion of coal pyrolysis vapors to light aromatics over hierarchical Y-type zeolites[J]. J Energy Inst,2020,93(4):1354−1363. doi: 10.1016/j.joei.2019.12.005 [32] NG E-P, NG D T-L, AWALA H, et al. Microwave synthesis of colloidal stable AlPO-5 nanocrystals with high water adsorption capacity and unique morphology[J]. Mater Lett,2014,132:126−129. doi: 10.1016/j.matlet.2014.06.022 [33] SING K S W. Reporting physisorption data for gas/solid systems with special reference to the determination of surface area and porosity (Recommendations 1984)[J]. Pure Appl Chem,1985,57(4):603−619. doi: 10.1351/pac198557040603 [34] VALLE B, PALOS R, BILBAO J, et al. Role of zeolite properties in bio-oil deoxygenation and hydrocarbons production by catalytic cracking[J]. Fuel Process Technol,2022,227:107130. [35] PARLITZ B, SCHREIER E, ZUBOWA H, et al. Isomerization of n-heptane over Pd-loaded silico-alumino-phosphate molecular sieves[J]. J Catal,1995,155(1):1−11. doi: 10.1006/jcat.1995.1182 [36] ALSOBAAI A, ZAKARIA R, HAMEED B. Gas oil hydrocracking on NiW/USY catalyst: Effect of tungsten and nickel loading[J]. Chem Eng J,2007,132(1/3):77−83. doi: 10.1016/j.cej.2007.01.021 [37] 祖运, 秦玉才, 高雄厚, 等. 催化裂化条件下噻吩与改性Y分子筛的作用机制[J]. 燃料化学学报,2015,43(7):862−869.ZU Yun, QIN Yucai, GAO Xionghou, et al. Mechanisms of thiophene conversion over the modified Y zeolites under catalytic cracking conditions[J]. J Fuel Chem Technol,2015,43(7):862−869. [38] MOSQUEDA-JIMÉNEZ B, JENTYS A, SESHAN K, et al. Reduction of nitric oxide by propene and propane on Ni-exchanged mordenite[J]. Appl Catal B: Environ,2003,43(2):105−115. doi: 10.1016/S0926-3373(02)00280-1 [39] SAAB R, POLYCHRONOPOULOU K, ANJUM D H, et al. Carbon nanostructure/zeolite Y composites as supports for monometallic and bimetallic hydrocracking catalysts[J]. Nanomaterials,2022,12(18):3246. doi: 10.3390/nano12183246 [40] LU K, JIN F, WU G, et al. The synergetic effect of acid and nickel sites on bifunctional MWW zeolite catalysts for ethylene oligomerization and aromatization[J]. Sustainable Energy Fuels,2019,3(12):3569−3581. doi: 10.1039/C9SE00771G [41] PIETERSE J A, SESHAN K, DOMOKOS L, et al. On the accessibility of acid sites in ferrierite for pyridine[J]. J Catal,1999,187(2):518−520. doi: 10.1006/jcat.1999.2629 [42] EMEIS C. Determination of integrated molar extinction coefficients for infrared absorption bands of pyridine adsorbed on solid acid catalysts[J]. J Catal,1993,141(2):347−354. doi: 10.1006/jcat.1993.1145 [43] MIRODATOS C, BARTHOMEUF D. Superacid sites in zeolites[J]. J Chem Soc, Chem Commun,1981,(2):39−40. doi: 10.1039/c39810000039 [44] LI S, ZHENG A, SU Y, et al. Brønsted/Lewis acid synergy in dealuminated HY zeolite: A combined solid-state NMR and theoretical calculation study[J]. J Am Chem Soc,2007,129(36):11161−11171. doi: 10.1021/ja072767y [45] WANG Q, GIANNETTO G, GUISNET M. Dealumination of zeolites III. Effect of extra-framework aluminum species on the activity, selectivity, and stability of Y zeolites in n-heptane cracking[J]. J Catal,1991,130(2):471−482. doi: 10.1016/0021-9517(91)90129-R [46] GUO F, LI J, LI W, et al. Quinoline hydrodenitrogenation over NiW/Al-MCM-41 catalysts with different al contents[J]. Russ J Appl Chem,2017,90(12):2055−2063. doi: 10.1134/S1070427217120242 [47] KOSTYNIUK A, KEY D, MDLELENI M. 1-hexene isomerization over bimetallic M-Mo-ZSM-5 (M: Fe, Co, Ni) zeolite catalysts: Effects of transition metals addition on the catalytic performance[J]. J Energy Inst,2020,93(2):552−564. doi: 10.1016/j.joei.2019.06.009 [48] DE LEÓN J D, PICQUART M, VILLARROEL M, et al. Effect of gallium as an additive in hydrodesulfurization WS2/γ-Al2O3 catalysts[J]. J Mol. Catal A: Chem , 2010, 323(1-2): 1-6. [49] ZHOU W, ZHOU Y, WEI Q, et al. Continuous synthesis of mesoporous Y zeolites from normal inorganic aluminosilicates and their high adsorption capacity for dibenzothiophene (DBT) and 4, 6-dimethyldibenzothiophene (4, 6-DMDBT)[J]. Chem Eng J,2017,330:605−615. doi: 10.1016/j.cej.2017.08.005 [50] TAYEB K B, LAMONIER C, LANCELOT C, et al. Active phase genesis of NiW hydrocracking catalysts based on nickel salt heteropolytungstate: Comparison with reference catalyst[J]. Appl Catal B: Environ,2012,126:55−63. doi: 10.1016/j.apcatb.2012.06.025 [51] ALSALME A, ALZAQRI N, ALSALEH A, et al. Efficient Ni-Mo hydrodesulfurization catalyst prepared through Keggin polyoxometalate[J]. Appl Catal B: Environ,2016,182:102−108. doi: 10.1016/j.apcatb.2015.09.018 [52] HUR Y G, LEE D-W, LEE K-Y J F. Hydrocracking of vacuum residue using NiWS (x) dispersed catalysts[J]. Fuel,2016,185:794−803. doi: 10.1016/j.fuel.2016.08.027 [53] CORTES J C, RODRIGUEZ C, MOLINA R, et al. Hydrocracking of 1-methylnaphtalene (1MN) over modified clays-supported NiMoS and NiWS catalyst[J]. Fuel,2021,295:120612. doi: 10.1016/j.fuel.2021.120612 -




 下载:
下载: