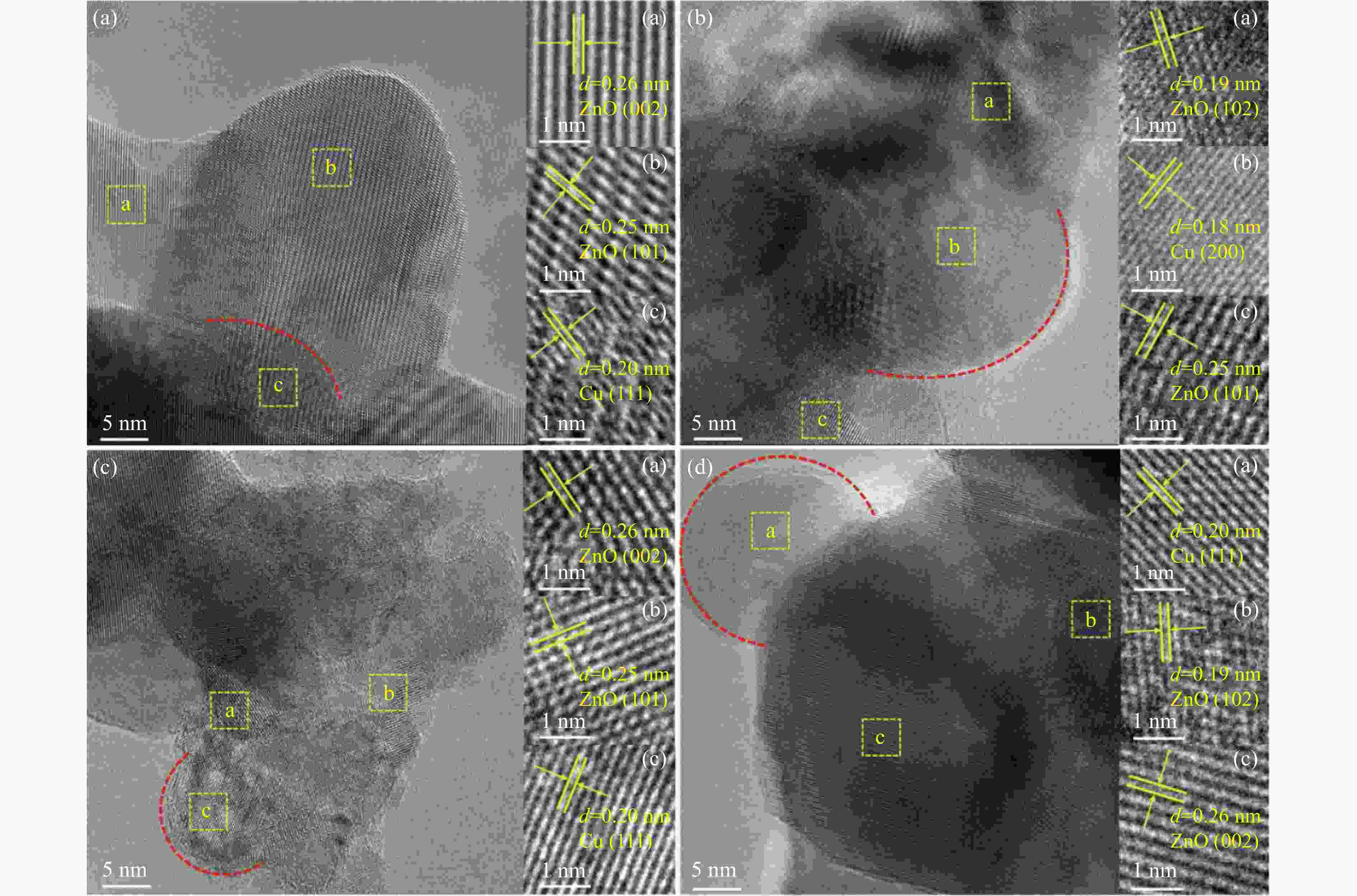
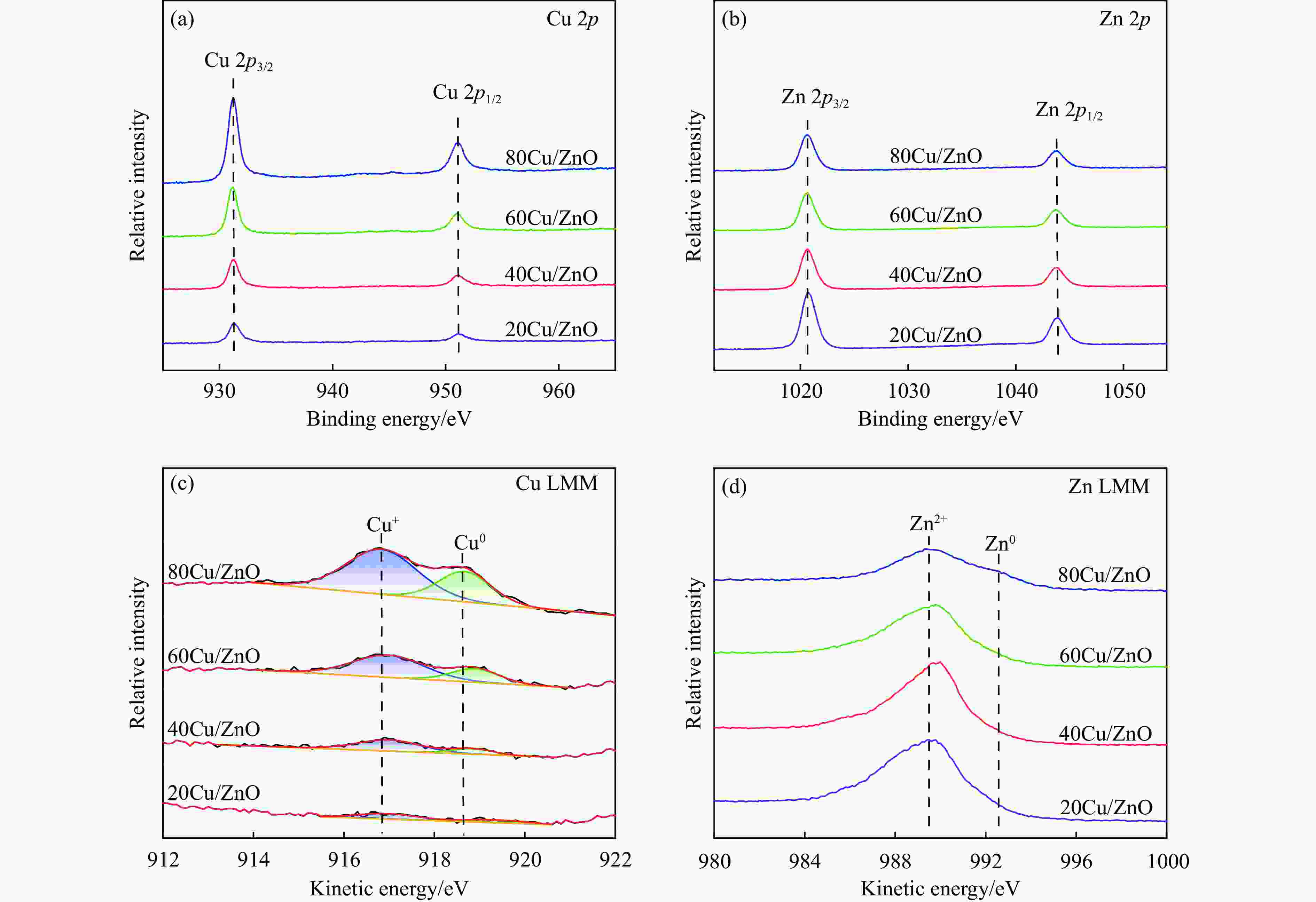
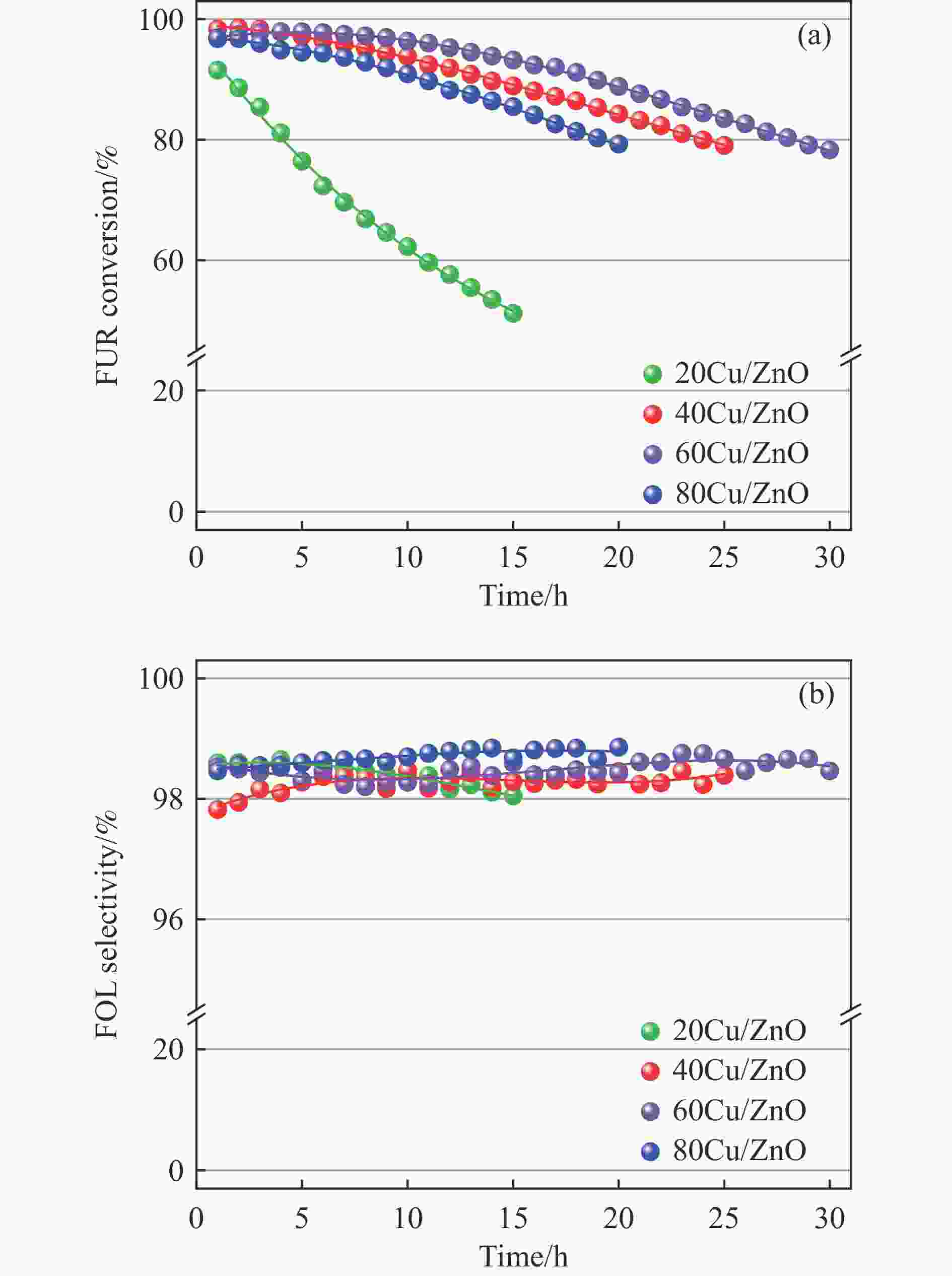
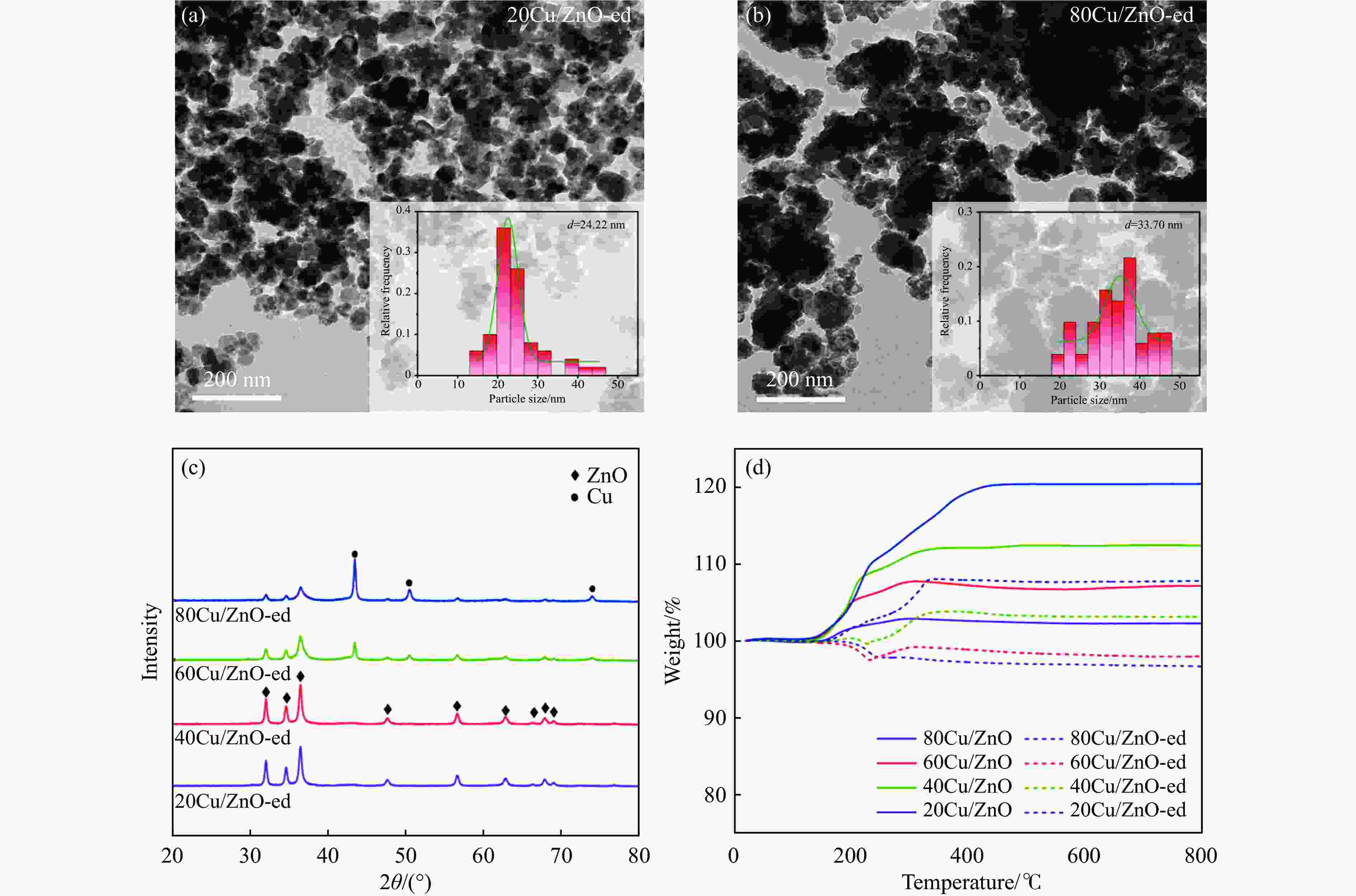
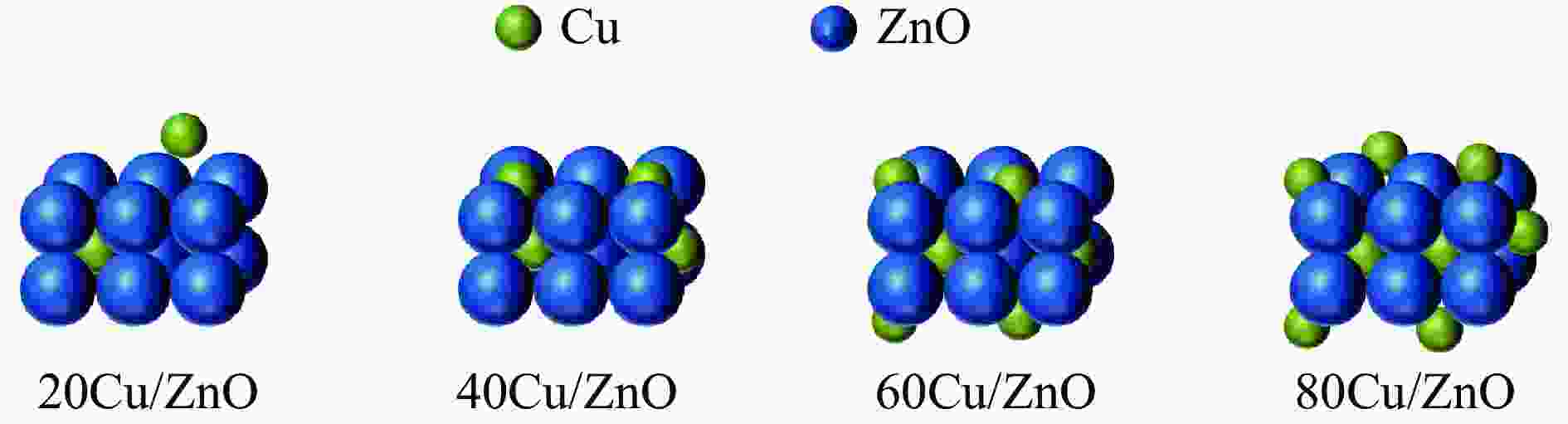
Effect of the metal-support interaction in the Cu/ZnO catalyst on its performance in the hydrogenation of furfural to furfuryl alcohol
-
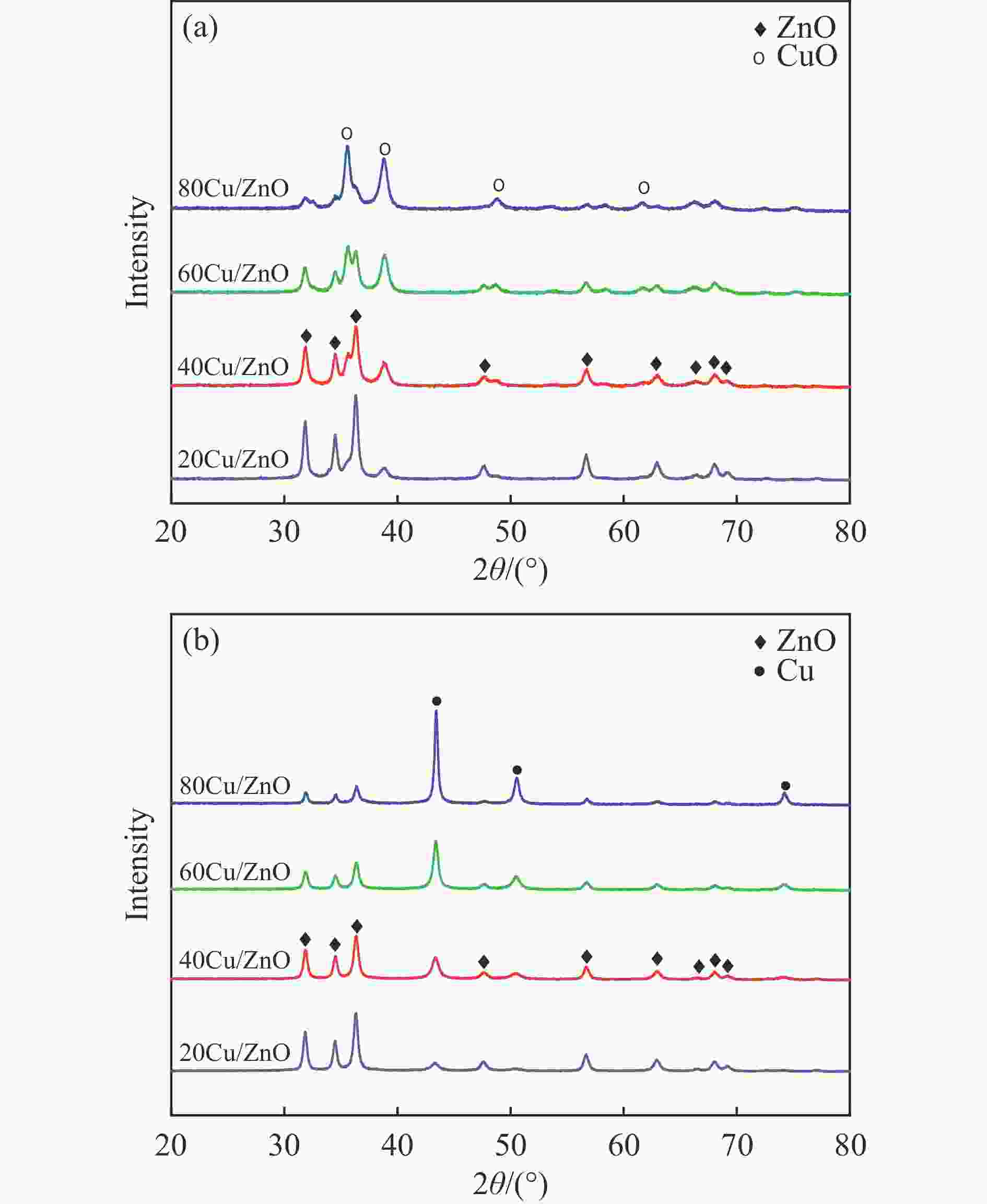
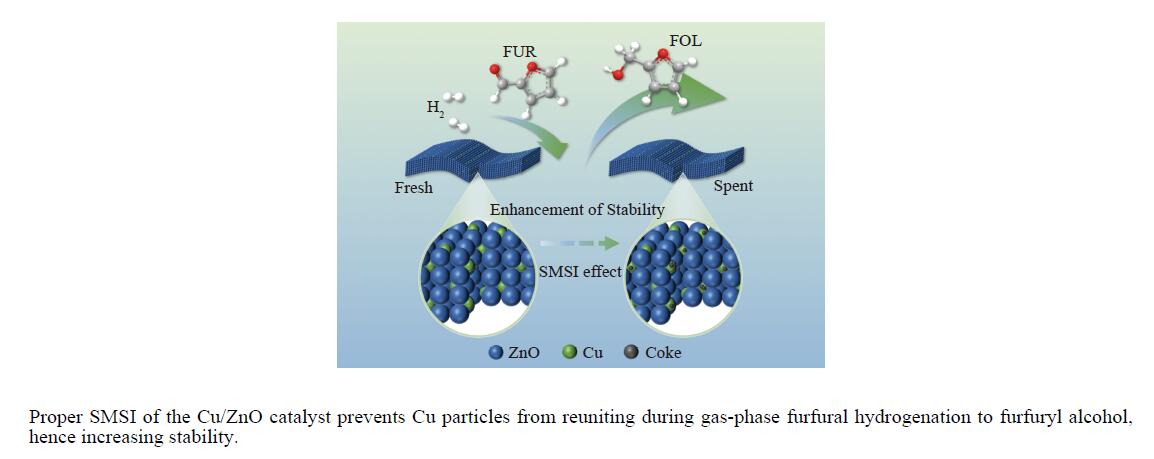
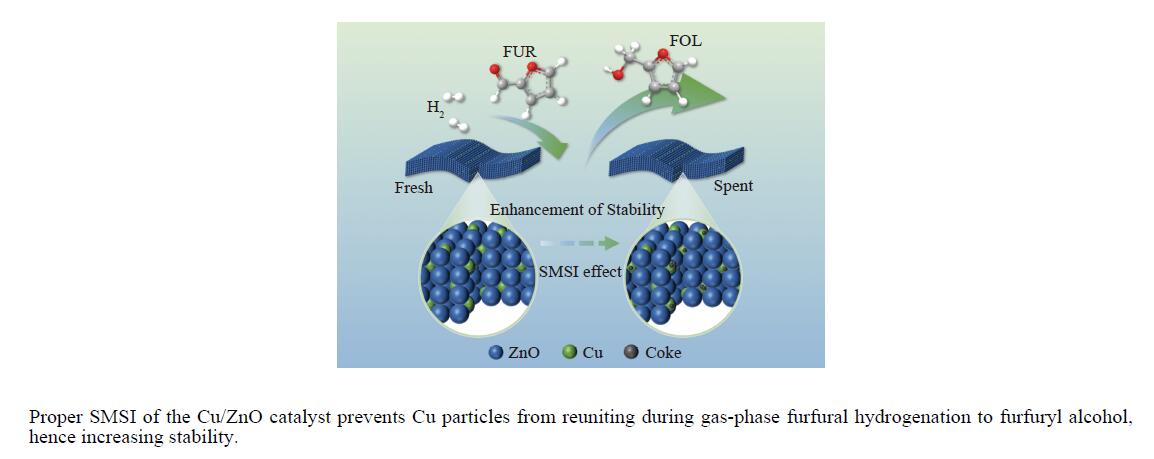
摘要: 采用共沉淀法制备了系列Cu/Zn比不同的Cu/ZnO催化剂,研究了Cu/Zn比与金属载体强相互作用(SMSI)的关系及其对糠醛气相加氢制糠醇催化反应性能的影响。XRD、H2-TPR、SEM、HRTEM和XPS等表征结果显示,Cu/ZnO催化剂中的金属载体强相互作用改变了催化剂的微观结构。ZnO载体对活性金属Cu颗粒具有不同程度的几何修饰,影响了Cu表面的电子状态。不同Cu/Zn比的Cu/ZnO催化剂的SMSI作用顺序为:20Cu/ZnO > 40Cu/ZnO > 60Cu/ZnO > 80Cu/ZnO。在同一反应条件下,20Cu/ZnO催化剂的糠醛转化率高于80%的时间仅为5 h,而60Cu/ZnO催化剂的糠醛转化率高于80%的时间可以达到28 h,表明适当的SMSI作用有利于提升Cu/ZnO催化剂在糠醛加氢反应中的稳定性,而过强的SMSI作用会抑制其催化活性。Abstract: A series of Cu/ZnO catalysts were prepared by the coprecipitation method and the effect of Cu/Zn ratio on the strong metal support interaction (SMSI) as well as its relation to the catalytic performance of Cu/ZnO in the gaseous hydrogenation of furfural to furfuryl alcohol was investigated. The H2-TPR, XRD, SEM, TEM and XPS characterization results reveal that there exists the SMSI effect in the Cu/ZnO catalyst that influences the catalyst microstructure. ZnO support, acting as a geometric modifier on the active metal Cu particles, has a significant influence on the electronic state of the surface Cu species. The strength of SMSI is related to the Cu/Zn ratio and the SMSI strength of various Cu/ZnO catalysts follows the order of 20Cu/ZnO > 40Cu/ZnO > 60Cu/ZnO > 80Cu/ZnO. Under the same reaction conditions, the lifetime of the 20Cu/ZnO catalyst with a furfural conversion of above 80% is only 5 h, in comparison with the lifetime of 28 h for the 60Cu/ZnO catalyst. That is, appropriate SMSI can enhance the stability of the Cu/ZnO catalyst in the hydrogenation of furfural to furfuryl alcohol, whereas excessive SMSI is detrimental to the catalyst activity.
-
表 1 新鲜Cu/ZnO催化剂的物理化学性质
Table 1 Physicochemical properties of fresh Cu/ZnO catalysts
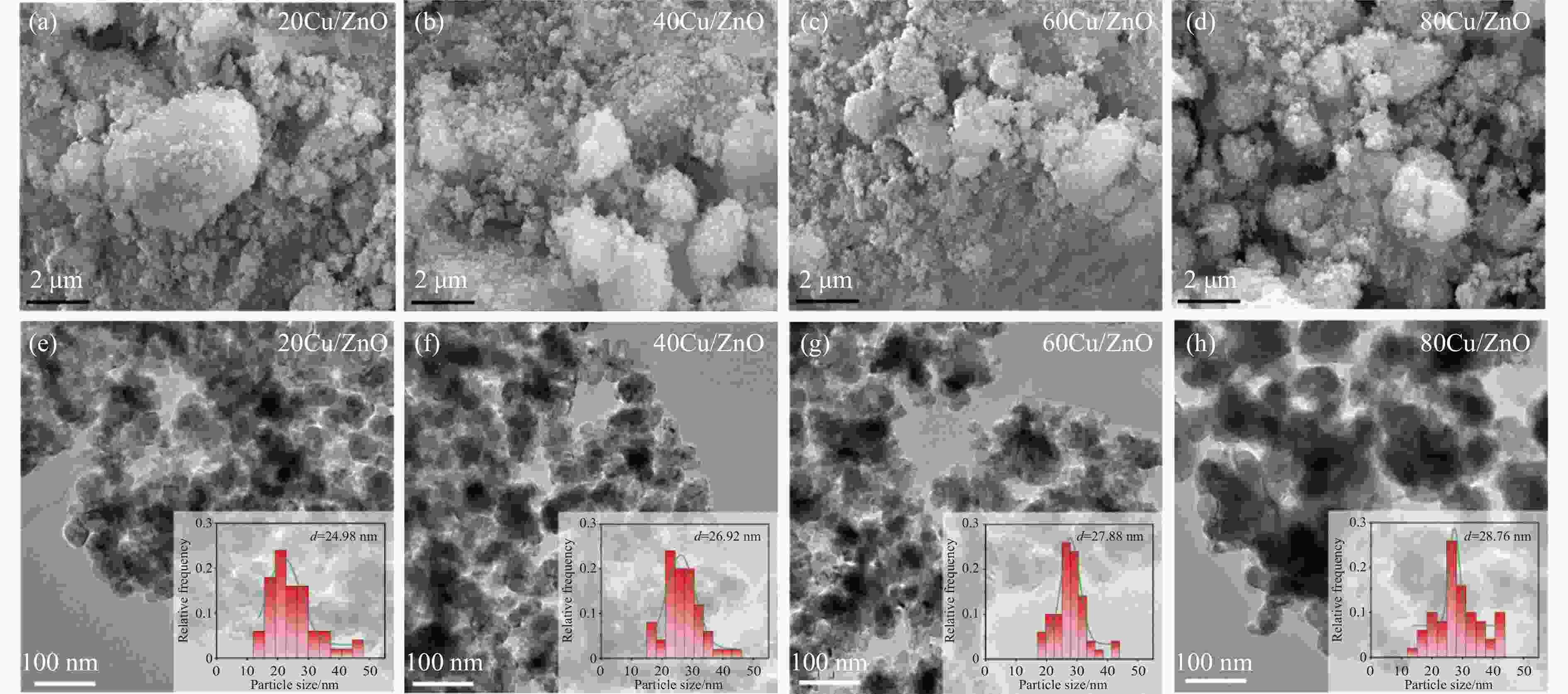
Catalyst SBETa/(m2·g−1) vtotala/(cm3·g−1) dporea/nm dCub/nm dparticlesb/nm dCud/nm DCud/% SCud/(m2·g−1) 20Cu/ZnO 22.9 0.14 24.1 9.3 25.0 8.3 12.1 13.1 40Cu/ZnO 42.5 0.17 18.8 10.6 26.0 13.6 7.4 15.9 60Cu/ZnO 28.5 0.17 24.2 14.4 28.0 21.0 4.8 15.4 80Cu/ZnO 24.2 0.18 24.3 20.2 29.0 16.3 6.1 26.5 a: Determined by nitrogen adsorption; b: Average Cu particle size was calculated using the Scherrer equation; c: Determined by TEM; d: Copper particle size, copper dispersion and exposed metallic copper surface area were determined using N2O-titration. 表 2 催化剂还原峰及酸量
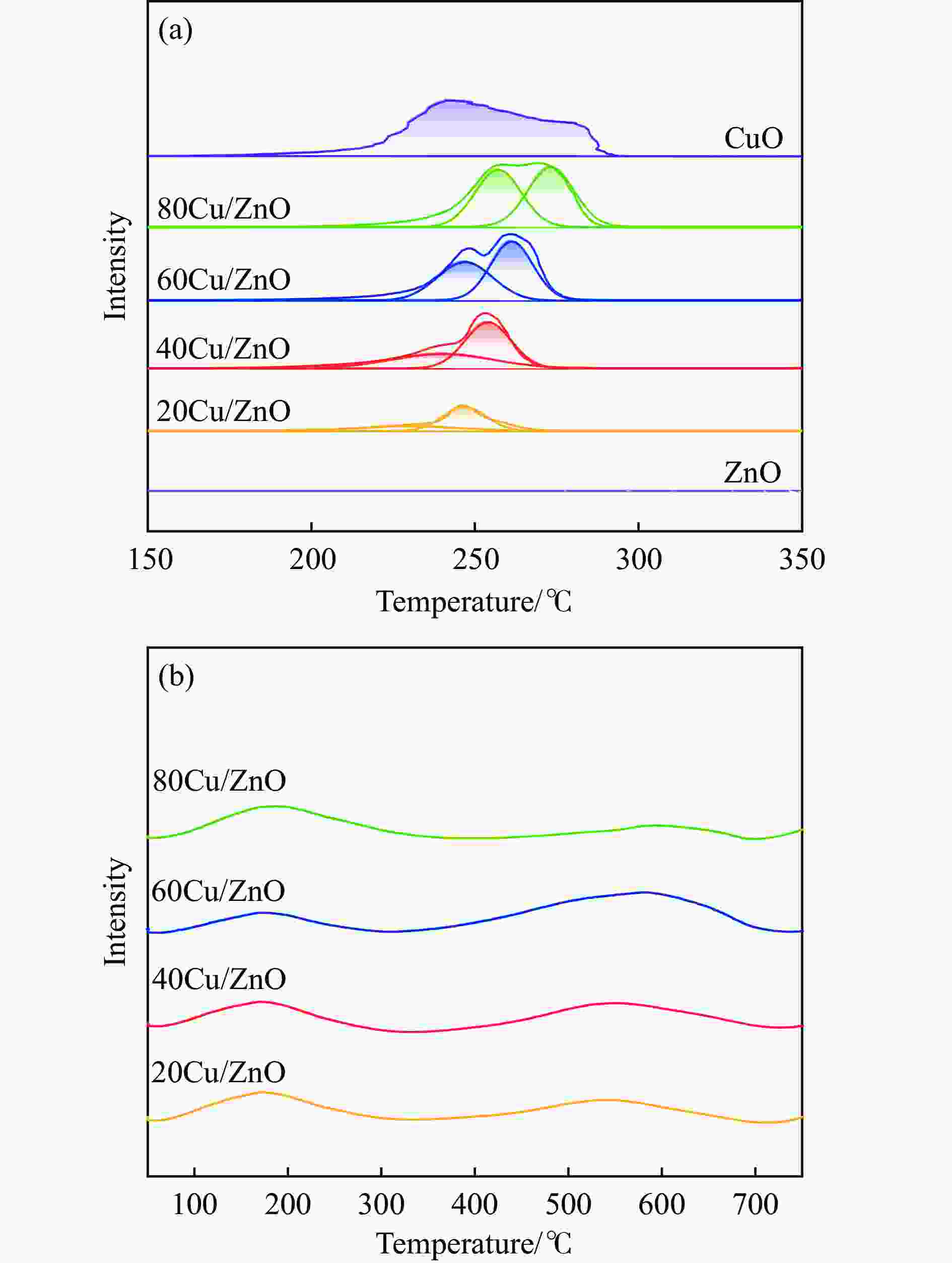
Table 2 Reduction temperature and acidity distribution of different catalysts
Catalyst α β Relative area Acidity/
(mmol·g−1)Acid site density/
(mmol·m−2)t/℃ fraction/% t/℃ fraction/% 20Cu/ZnO 229.7 28.0 246.9 72.0 2765.8 0.059 0.0025 40Cu/ZnO 240.1 40.6 254.1 59.4 6349.5 0.197 0.0046 60Cu/ZnO 247.0 46.4 261.3 53.6 9424.3 0.113 0.0039 80Cu/ZnO 257.1 49.0 273.2 51.0 10944.3 0.072 0.0029 表 3 还原态Cu/ZnO催化剂表面金属物种在XPS中的分布
Table 3 Distribution of various metal species on the surface of reduced Cu/ZnO catalysts determined by XPS
Catalyst BE/eV KE/eV Cu0/ (Cu++ Cu0)a Cu 2p3/2 Zn 2p3/2 Cu+ Cu0 Zn2+ Zn0 20Cu/ZnO 932.4 1020.8 916.96 919.40 989.7 − 33.58% 40Cu/ZnO 932.3 1020.6 917.00 918.85 990.1 − 23.55% 60Cu/ZnO 932.4 1020.5 916.98 918.90 990.3 − 29.16% 80Cu/ZnO 932.5 1020.6 916.83 918.67 988.9 992.4 32.83% a: Ratio of Cu0 to (Cu++Cu0) was obtained by deconvolution of the Cu LMM spectra. 表 4 使用后Cu/ZnO催化剂的物理化学性质
Table 4 Physicochemical properties of the spent Cu/ZnO catalysts after the reaction test
Catalyst dCua/nm dparticles b/nm △wc/% 20Cu/ZnO-spent 5.2 24.22 5.6 40Cu/ZnO-spent 6.0 − 9.2 60Cu/ZnO-spent 14.4 − 9.2 80Cu/ZnO-spent 20.5 33.70 12.66 a: Average Cu particle size was calculated from the XRD patterns by using the Scherrer equation; b: Mean particle size was determined by TEM; c: The coke deposition was determined by TGA. -
[1] LIU H W, HU Q, FAN G L, et al. Surface synergistic effect in well-dispersed Cu/MgO catalysts for highly efficient vapor-phase hydrogenation of carbonyl compounds[J]. Catal Sci Technol,2015,5(8):3960−3969. doi: 10.1039/C5CY00437C [2] DONG F, ZHU Y L, ZHENG H Y, et al. Cr-free Cu-catalysts for the selective hydrogenation of biomass-derived furfural to 2-methylfuran: The synergistic effect of metal and acid sites[J]. J Mol Catal A: Chem,2015,398:140−148. doi: 10.1016/j.molcata.2014.12.001 [3] AN J W, WANG X H, ZHAO J X, et al. Density-functional theory study on hydrogenation of dimethyl oxalate to methyl glycolate over copper catalyst: Effect of copper valence state[J]. Mol Catal,2020,482:110667. doi: 10.1016/j.mcat.2019.110667 [4] KURNIAWAN E, HAYASHI T, HOSAKA S, et al. Selective vapor-phase hydrogenation of furfural to furfuryl alcohol over Cu/silica catalysts[J]. Bull Chem Soc Jpn,2023,96(1):8−15. doi: 10.1246/bcsj.20220285 [5] GHASHGHAEE M, SHIRVANI S, GHAMBARIAN M. Kinetic models for hydroconversion of furfural over the ecofriendly Cu-MgO catalyst: An experimental and theoretical study[J]. App Catal A: Gen,2017,545:134−147. doi: 10.1016/j.apcata.2017.07.040 [6] SADJADI S, FARZANEH V, SHIRVANI S, et al. Preparation of Cu-MgO catalysts with different copper precursors and precipitating agents for the vapor-phase hydrogenation of furfural[J]. Korean J Chem Eng,2017,34(3):692−700. doi: 10.1007/s11814-016-0344-7 [7] R. BERTOLINI G, JIMÉNEZ-GÓMEZ C P, CECILIA J A, et al. Gas-phase hydrogenation of furfural to furfuryl alcohol over Cu-ZnO-Al2O3 catalysts prepared from layered double hydroxides[J]. Catalysts,2020,10(5):486. [8] ALGORABI S, AKMAZ S, KOÇ S N. The investigation of hydrogenation behavior of furfural over sol-gel prepared Cu/ZrO2 catalysts[J]. J Sol-Gel Sci Technol,2020,96(1):47−55. doi: 10.1007/s10971-020-05352-6 [9] JIMÉNEZ-GÓMEZ C P, CECILIA J A, FRANCO-DURO F I, et al. Promotion effect of Ce or Zn oxides for improving furfuryl alcohol yield in the furfural hydrogenation using inexpensive Cu-based catalysts[J]. Mol Catal,2018,455:121−131. doi: 10.1016/j.mcat.2018.06.001 [10] GARCÍA-SANCHO C, MÉRIDA-ROBLES J M, CECILIA-BUENESTADO J A, et al. The role of Copper in the hydrogenation of furfural and levulinic acid[J]. Int J Mol Sci,2023,24(3):2443. doi: 10.3390/ijms24032443 [11] ZHENG J W, ZHOU J F, LIN H Q, et al. CO-mediated deactivation mechanism of SiO2-supported copper catalysts during dimethyl oxalate hydrogenation to ethylene glycol[J]. J Phys Chem C,2015,119(24):13758−13766. doi: 10.1021/acs.jpcc.5b03569 [12] LI Y W, ZHANG R M, DU L K, et al. Catalytic mechanism of C–F bond cleavage: Insights from QM/MM analysis of fluoroacetate dehalogenase[J]. Catal Sci Technol,2016,6(1):73−80. doi: 10.1039/C5CY00777A [13] CERÓN M R, IZQUIERDO M, ALEGRET N, et al. Reactivity differences of Sc3N@C2n(2n = 68 and 80). Synthesis of the first methanofullerene derivatives of Sc3N@D5h -C80[J]. Chem Commun,2016,52(1):64−67. doi: 10.1039/C5CC07416A [14] TAUSTER S J, FUNG S C, BAKER R T K, et al. Strong interactions in supported-metal catalysts[J]. Science,1981,211(4487):1121−1125. doi: 10.1126/science.211.4487.1121 [15] TAUSTER J S, FUNG C S, GARTEN L R. Strong metal-support interactions. Group 8 noble metals supported on titanium dioxide[J]. J Am Chem Soc,2002,100(1):170−175. [16] TAUSTER J. Strong metal-support interactions[J]. Accounts Chem Res,1987,20(11):121−140. [17] WANG W X, LI X K, ZHANG Y, et al. Strong metal-support interactions between Ni and ZnO particles and their effect on the methanation performance of Ni/ZnO[J]. Catal Sci Technol,2017,7(19):4413−4421. doi: 10.1039/C7CY01119A [18] LIU X Y, LIU M H, LUO Y C, et al. Strong metal-support interactions between gold nanoparticles and ZnO nanorods in CO oxidation[J]. J Am Chem Soc,2012,134(24):10251−10258. doi: 10.1021/ja3033235 [19] HANSEN P L, WAGNER J B, HELVEG S, et al. Atom-resolved imaging of dynamic shape changes in supported copper nanocrystals[J]. Science,2002,295(5562):2053−2055. doi: 10.1126/science.1069325 [20] JIMÉNEZ-GÓMEZ C P, CECILIA J A, DURÁN-MARTÍN D, et al. Gas-phase hydrogenation of furfural to furfuryl alcohol over Cu/ZnO catalysts[J]. J Catal,2016,336:107−115. doi: 10.1016/j.jcat.2016.01.012 [21] 萧垚鑫, 张军, 胡升, 等. 甲醇供氢体系铜锌双金属催化糠醛加氢转化[J]. 化工进展,2023,42(3):1341−1352.XIAO Yaoxin, ZHANG Jun, HU Sheng, et al. Catalytic hydrogenation of furfural by copper and zinc bimetal in methanol hydrogen supply system[J]. Chem Ind Eng Prog,2023,42(3):1341−1352. [22] LI K, CHEN J G. CO2 hydrogenation to methanol over ZrO2-containing catalysts: Insights into ZrO2 induced synergy[J]. ACS Catal,2019,9(9):7840−7861. doi: 10.1021/acscatal.9b01943 [23] SCHUMANN J, EICHELBAUM M, LUNKENBEIN T, et al. Promoting strong metal support interaction: doping ZnO for enhanced activity of Cu/ZnO: M (M = Al, Ga, Mg) catalysts[J]. ACS Catal,2015,5(6):3260−3270. doi: 10.1021/acscatal.5b00188 [24] ZHANG S Y, HU Q, FAN G L, et al. The relationship between the structure and catalytic performance Cu/ZnO/ZrO2 catalysts for hydrogenation of dimethyl 1, 4-cyclohexane dicarboxylate[J]. Catal Commun,2013,39:96−101. doi: 10.1016/j.catcom.2013.05.011 [25] ZHANG S Y, LIU Q Y, FAN G L, et al. Highly-dispersed copper-based catalysts from Cu-Zn-Al layered double hydroxide precursor for gas-phase hydrogenation of dimethyl oxalate to ethylene glycol[J]. Catal Lett,2012,142(9):1121−1127. doi: 10.1007/s10562-012-0871-8 [26] 孔祥鹏, 游新明, 元培红, 等. 助剂对于Cu/ZnO催化剂结构特征及催化草酸二甲酯加氢合成乙二醇反应性能的影响[J]. 燃料化学学报(中英文),2023,51(6):794−803. doi: 10.1016/S1872-5813(22)60073-2KONG Xiangpeng, YOU Xinming, YUAN Peihong, et al. Effects of Additives on structural characteristics and catalytic performance of Cu/ZnO catalyst for hydrogenation of Dimethyl oxalate to ethylene glycol[J]. J Fuel Chem Technol,2023,51(6):794−803. doi: 10.1016/S1872-5813(22)60073-2 [27] 黄玉辉, 任国卿, 孙蛟, 等. Cu/ZnO催化糠醛气相加氢制2-甲基呋喃的研究[J]. 燃料化学学报,2016,44(11):1349−1355.HUANG Yuhui, REN Guoqing, SUN Jiao, et al. Study on gas phase hydrogenation of furfural catalyzed by Cu/ZnO to 2-methylfuran[J]. J Fuel Chem Technol,2016,44(11):1349−1355. [28] 黄玉辉, 任国卿, 孙蛟, 等. 沉淀剂对CuZnAl催化剂糠醛气相加氢制糠醇选择性的影响[J]. 燃料化学学报,2016,44(6):726−731.HUANG Yuhui, REN Guoqing, SUN Jiao, et al. Effect of precipitator on selectivity of furfural gas phase hydrogenation to furfuryl alcohol by CuZnAl catalyst[J]. J Fuel Chem Technol,2016,44(6):726−731. [29] YUAN Z L, WANG L N, WANG J H, et al. Hydrogenolysis of glycerol over homogenously dispersed copper on solid base catalysts[J]. Appl Catal B: Environ,2011,101(3/4):431−440. doi: 10.1016/j.apcatb.2010.10.013 [30] LI H B, CUI Y Y, LIU Y X, et al. Highly efficient Ag-modified copper phyllosilicate nanotube: Preparation by co-ammonia evaporation hydrothermal method and application in the selective hydrogenation of carbonate[J]. J Mater Sci Technol,2020,47(12):29−37. [31] 于冬冬, 于欣瑞, 张雅静, 等. 糠醛气相加氢制备糠醇Cu/SiO2催化剂的失活机理研究[J]. 燃料化学学报(中英文),2023,51(12):1751−1760. doi: 10.1016/S1872-5813(23)60362-7YU Dongdong, YU Xinrui, ZHANG Yajing, et al. Study on deactivation mechanism of Furfuryl alcohol Cu/SiO2 catalyst prepared by furfural Gasification[J]. J Fuel Chem Technol,2023,51(12):1751−1760. doi: 10.1016/S1872-5813(23)60362-7 [32] PARK S W, JOO O S, JUNG K D, et al. Development of ZnO/Al2O3 catalyst for reverse-water-gas-shift reaction of CAMERE (carbon dioxide hydrogenation to form methanol via a reverse-water-gas-shift reaction) process[J]. Appl Catal A: Gen,2001,211(1):81−90. doi: 10.1016/S0926-860X(00)00840-1 [33] MENG H, LIU J G, DU Y L, et al. Novel Cu-based oxides catalyst from one-step carbothermal reduction decomposition method for selective catalytic reduction of NO with NH3[J]. Catal Commun,2019,119:101−105. doi: 10.1016/j.catcom.2018.10.023 [34] 丛昱, 包信和, 张涛, 等. CO2加氢合成甲醇的超细Cu-ZnO-ZrO2催化剂的表征[J]. 催化学报,2000,(4):314−318.CONG Yu, BAO Xinhe, ZHANG Tao, et al. Characterization of ultrafine Cu-ZnO-ZrO2 catalyst for hydrogenation of CO2 to methanol[J]. Chin J Catal,2000,(4):314−318. [35] JIMÉNEZ-GÓMEZ C P, CECILIA J A, ALBA-RUBIO A C, et al. Tailoring the selectivity of Cu-based catalysts in the furfural hydrogenation reaction: Influence of the morphology of the silica support[J]. Fuel,2022,319:123827. doi: 10.1016/j.fuel.2022.123827 [36] TU Y J, CHEN Y W. Effects of alkaline-earth oxide additives on silica-supported copper catalysts in ethanol dehydrogenation[J]. Ind Eng Chem Res,1998,37(7):2618−2622. doi: 10.1021/ie9708135 [37] BEHRENS M, STUDT F, KASATKIN I, et al. The active site of methanol synthesis over Cu/ZnO/Al2O3 industrial catalysts[J]. Science,2012,336(6083):893−897. doi: 10.1126/science.1219831 [38] NIE R F, LEI H, PAN S Y, et al. Core-shell structured CuO-ZnO@H-ZSM-5 catalysts for CO hydrogenation to dimethyl ether[J]. Fuel,2012,96:419−425. [39] TURCO M, BAGNASCO G, CAMMARANO C, et al. Cu/ZnO/Al2O3 catalysts for oxidative steam reforming of methanol: The role of Cu and the dispersing oxide matrix[J]. Appl Catal B: Environ,2007,77(1/2):46−57. doi: 10.1016/j.apcatb.2007.07.006 [40] CHEN F, LIANG J M, WANG F, et al. Improved catalytic activity and stability of Cu/ZnO catalyst by boron oxide modification for low-temperature methanol synthesis[J]. Chem Eng J,2023,458:141401. [41] KARIM W, SPREAFICO C, KLEIBERT A, et al. Catalyst support effects on hydrogen spillover[J]. Nature,2017,541(7635):68−71. doi: 10.1038/nature20782 [42] ZHANG S, PLESSOW P N, WILLIS J J, et al. Dynamical observation and detailed description of catalysts under strong metal-support interaction[J]. Nano Lett,2016,16(7):4528−4534. doi: 10.1021/acs.nanolett.6b01769 [43] YANG X, CHEN H, MENG Q, et al. Insights into influence of nanoparticle size and metal–support interactions of Cu/ZnO catalysts on activity for furfural hydrogenation[J]. Catal Sci Technol,2017,7(23):5625−5634. doi: 10.1039/C7CY01284E [44] 陈列. 晶化的介孔金属氧化物的合成与电化学储能性能研究[D]. 南京: 南京大学, 2015.CHEN Lie. Synthesis and electrochemical energy storage Performance of crystallized mesoporous metal oxides[D]. Nanjing: Nanjing University, 2015.) [45] VAN DEELEN T W, HERNÁNDEZ MEJÍA C, DE JONG K P. Control of metal-support interactions in heterogeneous catalysts to enhance activity and selectivity[J]. Nat Catal,2019,2(11):955−970. doi: 10.1038/s41929-019-0364-x [46] CHEN H, CUI H S, LV Y, et al. CO2 hydrogenation to methanol over Cu/ZnO/ZrO2 catalysts: Effects of ZnO morphology and oxygen vacancy[J]. Fuel,2022,314:123035. doi: 10.1016/j.fuel.2021.123035 [47] 海雪清, 谭静静, 何静, 等. CuCo双金属催化剂催化糠醛加氢制备1, 5-戊二醇的研究[J]. 燃料化学学报(中英文),2023,51(7):959−969. doi: 10.1016/S1872-5813(23)60334-2HAI Xueqing, TAN Jingjing, HE Jing, et al. Preparation of 1, 5-pentanediol by hydrogenation of furfural with CuCo bimetallic catalyst[J]. J Fuel Chem Technol,2023,51(7):959−969. doi: 10.1016/S1872-5813(23)60334-2 [48] GUO T, GUO Q, LI S Z, et al. Effect of surface basicity over the supported Cu-ZnO catalysts on hydrogenation of CO2 to methanol[J]. J Catal,2022,407:312−321. doi: 10.1016/j.jcat.2022.01.035 [49] ZHANG J Y, JIA Z, YU S T, et al. Regulating the Cu0-Cu+ ratio to enhance metal-support interaction for selective hydrogenation of furfural under mild conditions[J]. Chem Eng J, 2023, 468. [50] JIMÉNEZ-GÓMEZ C P, CECILIA J A, MORENO-TOST R, et al. Selective production of 2-methylfuran by gas-phase hydrogenation of furfural on copper incorporated by complexation in mesoporous silica catalysts[J]. ChemSusChem,2017,10(7):1448−1459. doi: 10.1002/cssc.201700086 [51] LI F, CAO B, MA R, et al. Performance of Cu/TiO2 -SiO2 catalysts in hydrogenation of furfural to furfuryl alcohol[J]. Can J Chem Eng,2016,94(7):1368−1374. doi: 10.1002/cjce.22503 [52] VASILIADOU E, EGGENHUISEN T, MUNNIK P, et al. Synthesis and performance of highly dispersed Cu/SiO2 catalysts for the hydrogenolysis of glycerol[J]. Appl Catal B: Environ,2014,145:108−119. -




 下载:
下载: