Acidity regulation of Fe-based catalysts and its effect on the selectivity of HDS reaction pathways
-
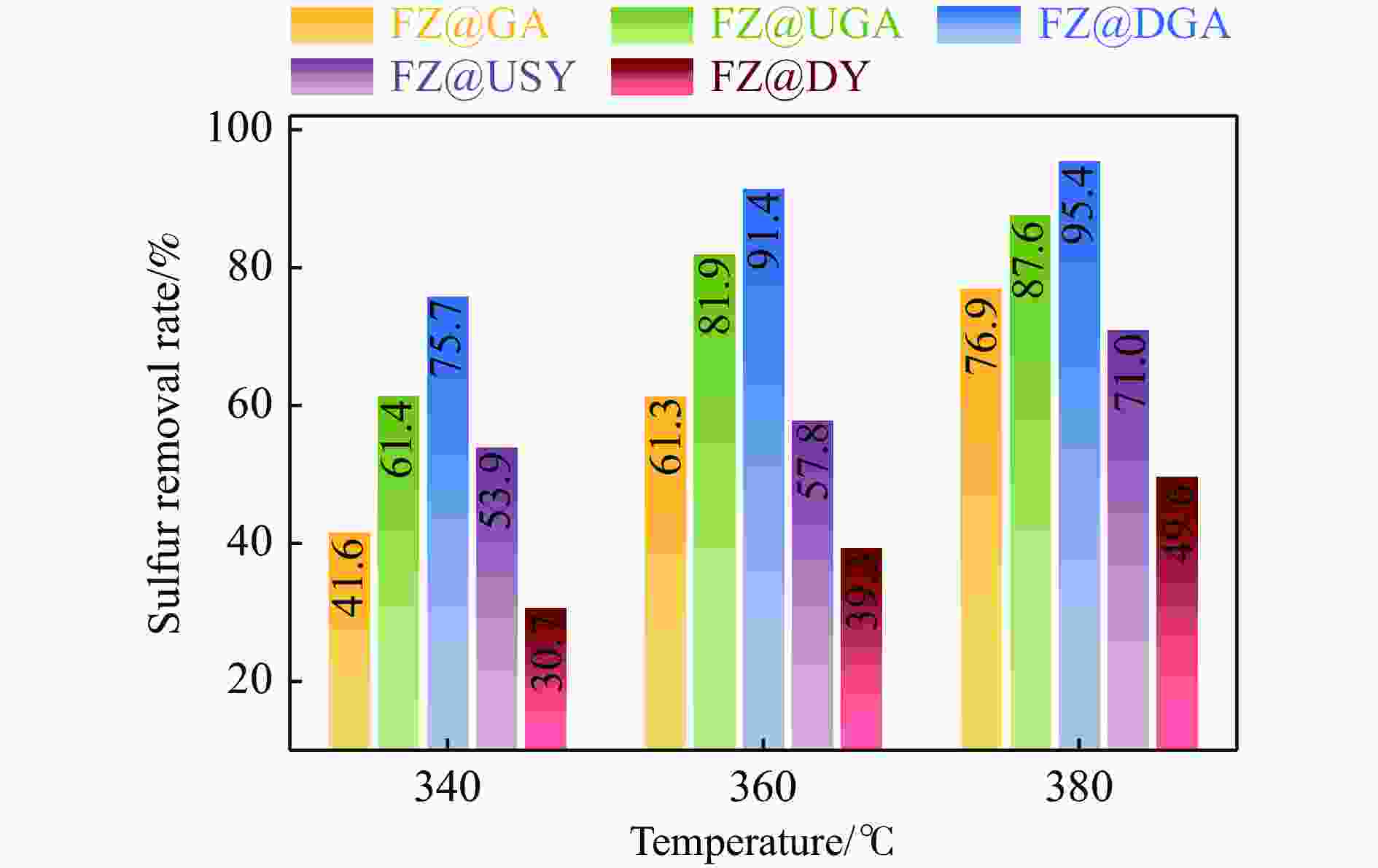
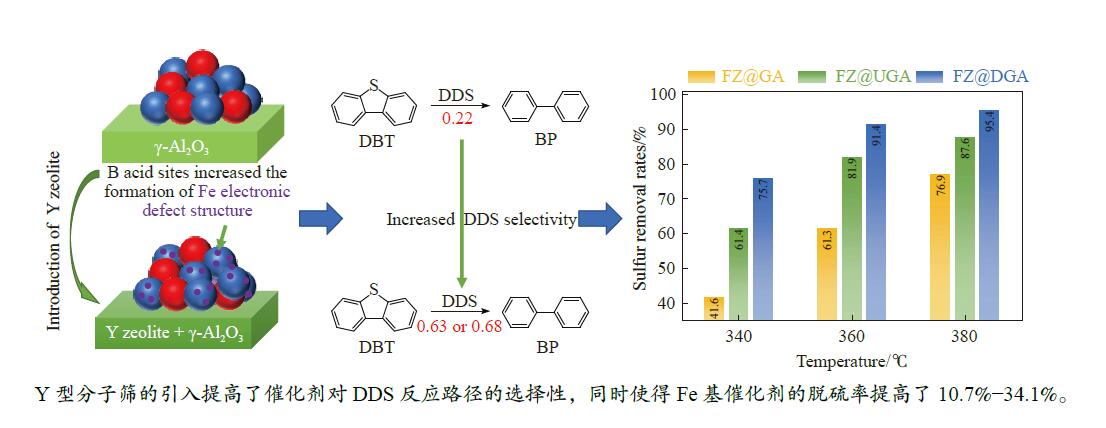
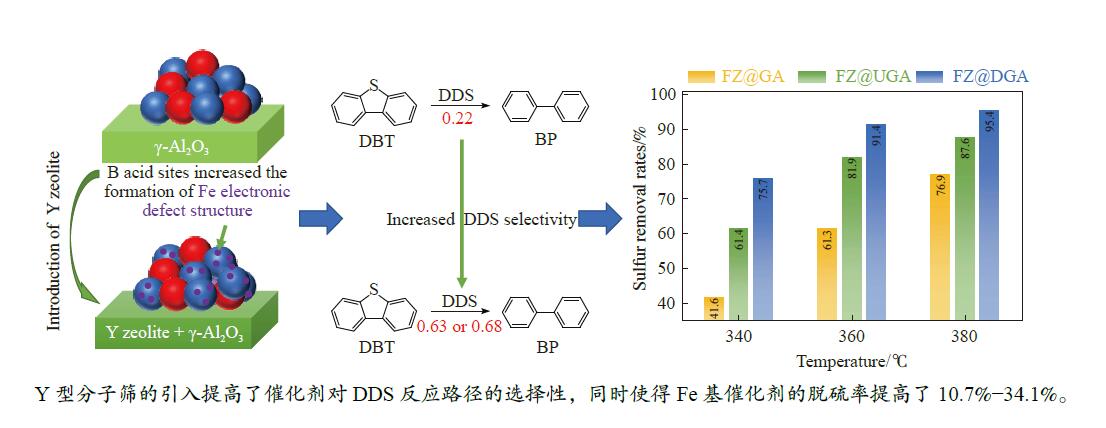
摘要: 以Fe作为主活性金属、Zn作为助活性金属,制备了Y型分子筛改性的Fe基加氢脱硫(HDS)催化剂。采用低温氮气物理吸附、X射线衍射(XRD)、氢气程序升温还原(H2-TPR)、氨气程序升温脱附(NH3-TPD)、扫描电子显微镜(SEM)、X射线光电子能谱(XPS)和吡啶红外(Py-IR)等表征方法对改性前后Fe基催化剂的形貌、孔结构、分散性、还原性、电子缺陷结构以及酸性等变化进行了研究,并使用固定床反应器对Fe基催化剂的HDS性能进行了评价。结果表明,Y型分子筛的引入提供了Brønsted(B)酸中心,使得Fe基催化剂的脱硫率提高了10.7%−34.1%。同时,B酸中心提高了催化剂的直接脱硫(DDS)反应路径的选择性。此外,B酸中心在促进DDS反应路径选择性增加的同时,抑制了预加氢脱硫(HYD)反应路径中四氢二苯并噻吩(THDBT)和六氢二苯并噻吩(HHDBT)更进一步的深度加氢,从而在保证脱硫率提升的同时又降低了氢耗。其根本原因可能是Y型分子筛的引入增强了催化剂的酸性,特别是B酸中心和活性金属之间的相互作用促进了电子转移,从而调节了Fe物种的电子缺陷结构,进而提升了催化剂的HDS性能。Abstract: An Fe-based hydrodesulfurization (HDS) catalyst modified by Y zeolite was developed using Fe as the main active metal and Zn as a promoter. The change of morphology, pore structure, dispersity, reducibility, electronic defect structure and acidity of the Fe-based catalysts before and after modification were investigated using low-temperature nitrogen physical adsorption, X-ray diffraction (XRD), H2-temperature programmed reduction (H2-TPR), NH3-temperature programmed desorption (NH3-TPD), scanning electron microscopy (SEM), X-ray photoelectron spectroscopy (XPS) and pyridine infrared spectroscopy (Py-IR). Meanwhile, the HDS performance of the Fe-based catalyst was evaluated using a fixed-bed reactor. The results showed that the introduction of Y zeolite provided the Brønsted (B) acid sites, which increased the sulfur removal rates of Fe based catalysts by 10.7% −34.1%. Meanwhile, the B acid sites improved the selectivity of the direct desulfurization (DDS) reaction pathway. In addition, the B acid sites not only promoted the increase of DDS selectivity but also inhibited further deep hydrogenation of tetrahydrodibenzothiophene (THDBT) and hexahydrodibenzothiophene (HHDBT) in the hydrogenation (HYD) reaction pathway, thereby ensuring an increase in desulfurization efficiency while reducing hydrogen consumption. The fundamental reason was that the introduction of Y zeolite enhanced the acidity of the modified catalyst, especially the interaction between B acid sites and active metal promoted electron transfer, which adjusted the Fe species electronic defect structure, resulting in the improvement of HDS performance.
-
Key words:
- Fe-based catalyst /
- Y zeolite /
- acidity /
- electronic defect structure /
- HDS /
- dibenzothiophene
-
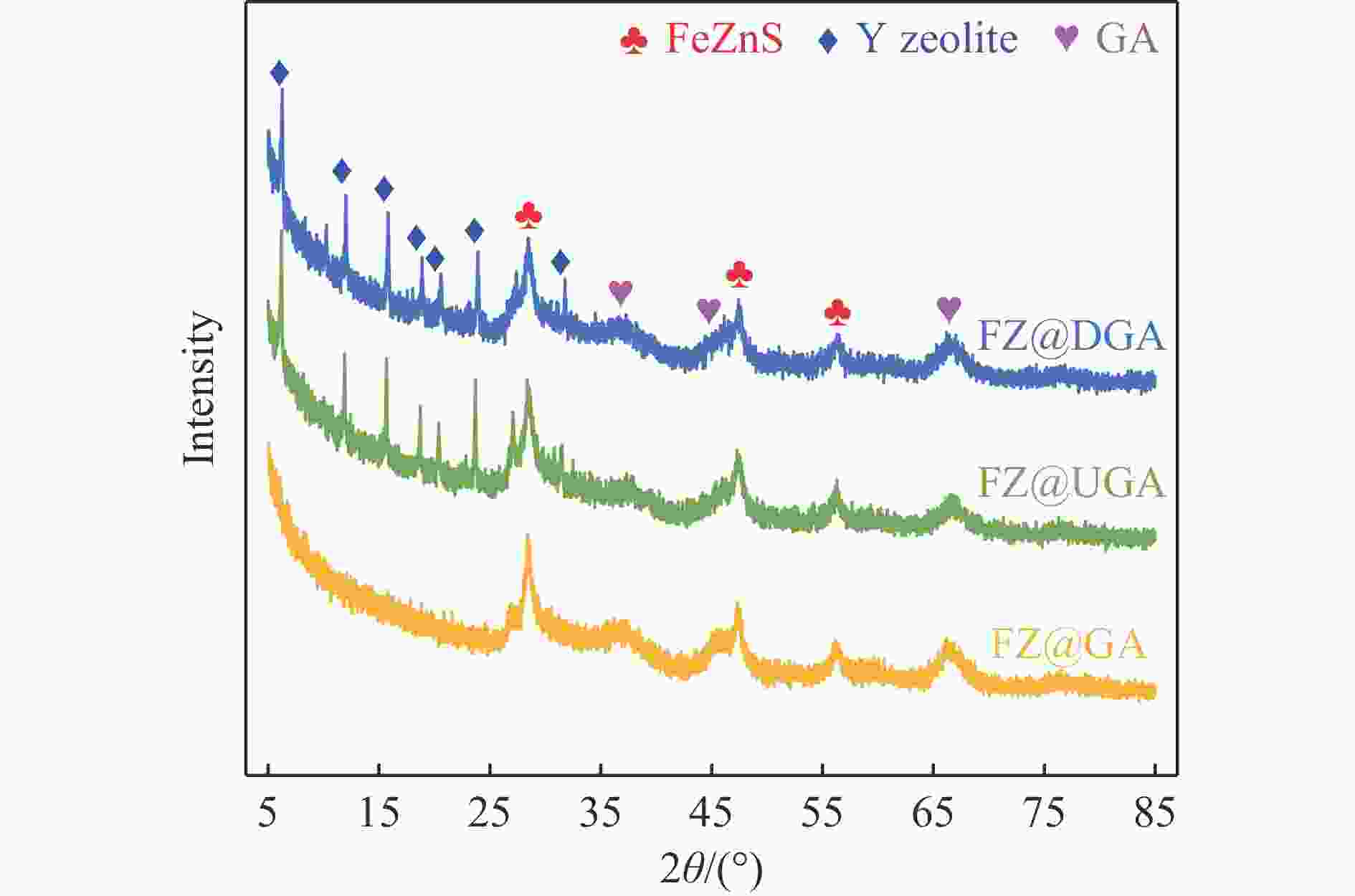
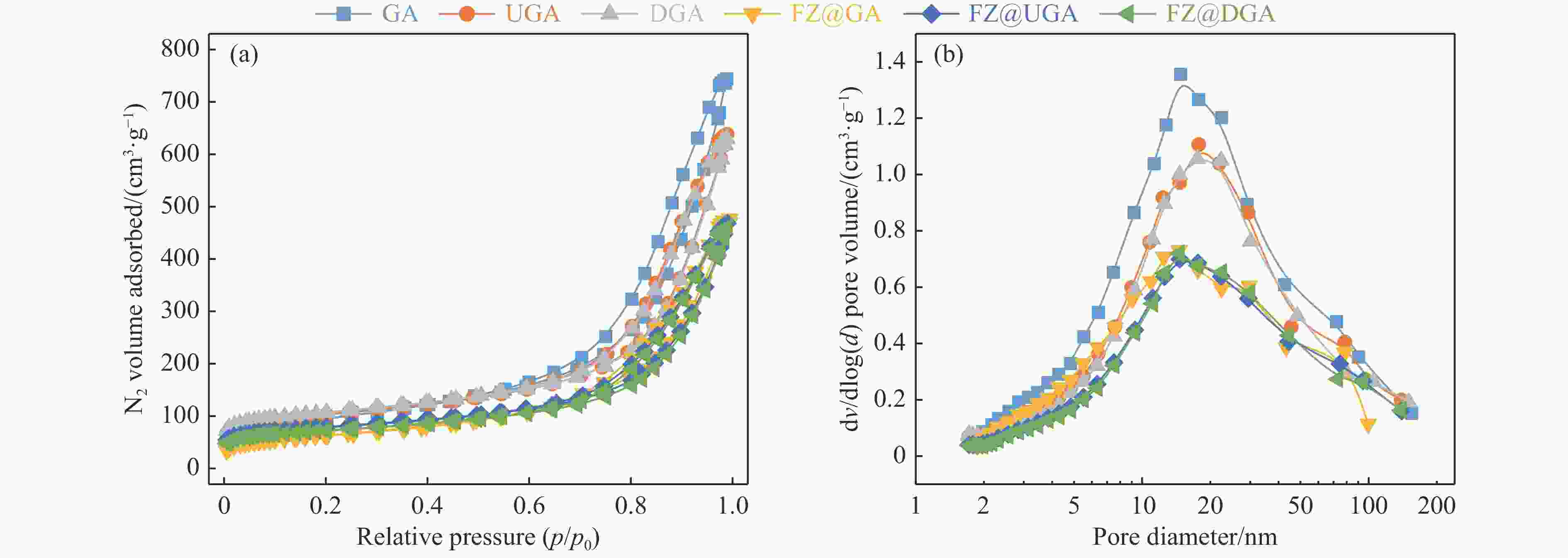
表 1 载体以及Fe基催化剂的织构性质
Table 1 Textural properties of supports and Fe-based catalysts
Sample Surface area/(m2·g−1) Pore volume/(cm3·g−1) BETa external microporeb totalc mesopore microporeb USY 586 509 77 0.389 0.140 0.249 DY 578 533 45 0.364 0.103 0.261 GA 336 296 40 1.150 1.133 0.017 UGA 353 232 121 0.987 0.929 0.058 DGA 366 220 146 0.975 0.905 0.070 FZ@GA 225 200 25 0.737 0.727 0.010 FZ@UGA 270 159 111 0.724 0.671 0.053 FZ@DGA 248 154 94 0.714 0.669 0.045 a: BET method; b: t-plot method; c: Volume adsorbed at p/p0 =0.99. 表 2 Fe基催化剂的H2-TPR谱图对应的还原温度
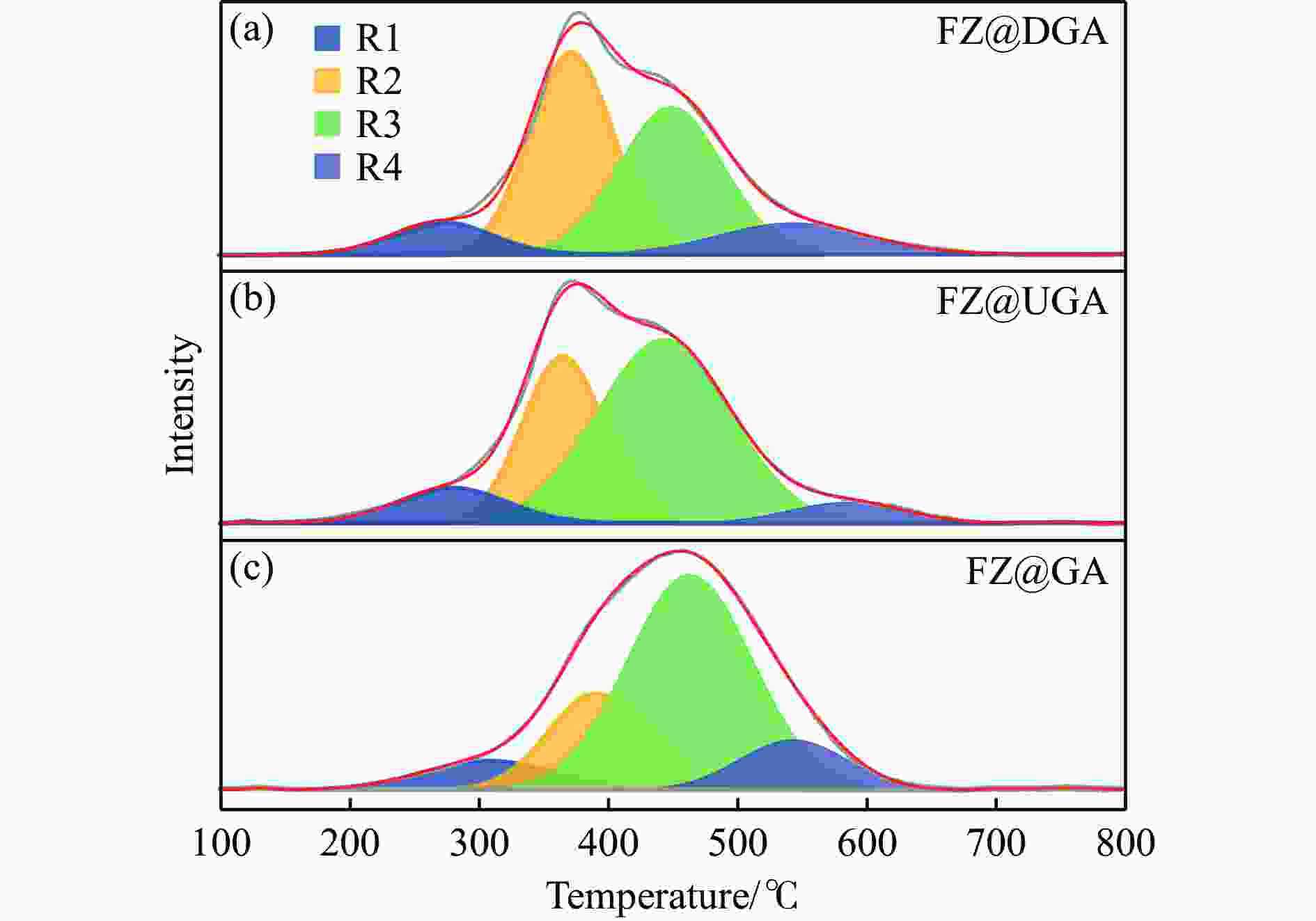
Table 2 Reduction temperature corresponding to peak of oxide Fe-based catalysts from H2-TPR
Sample Temperature corresponding to reduction peak/℃ R1 R2 R3 R4 FZ@GA 307 390 463 540 FZ@UGA 279 365 443 585 FZ@DGA 271 371 448 542 表 3 由Py-IR测得的改性前后载体和Fe基催化剂表面酸中心的类型和含量
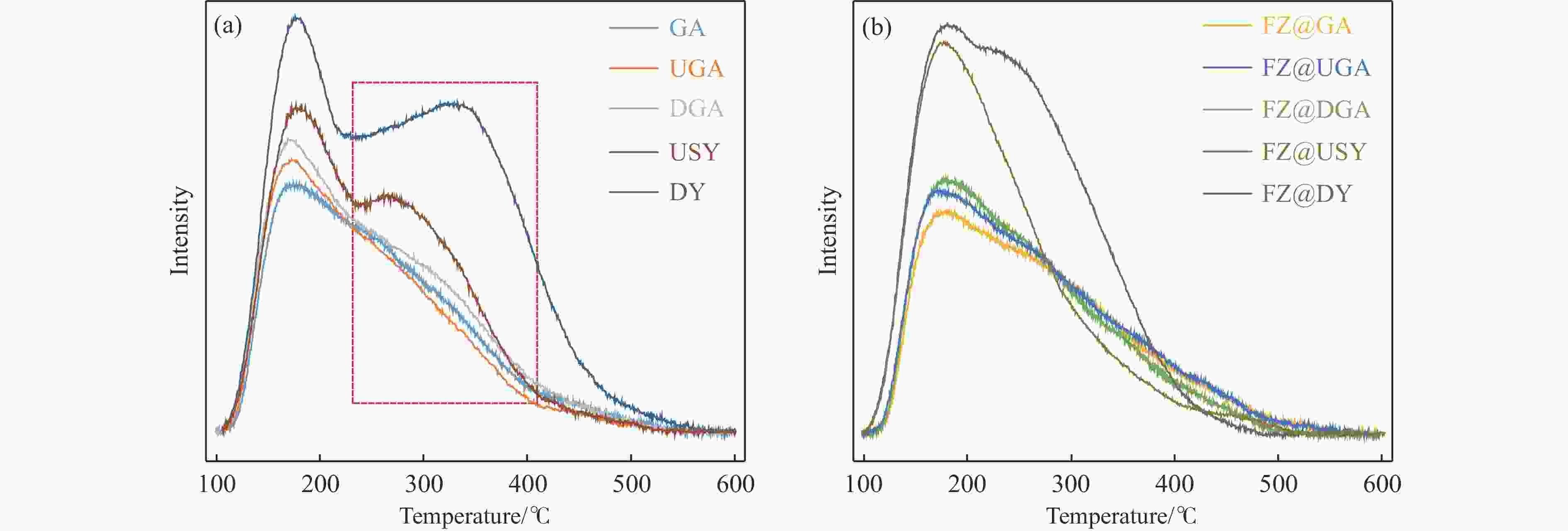
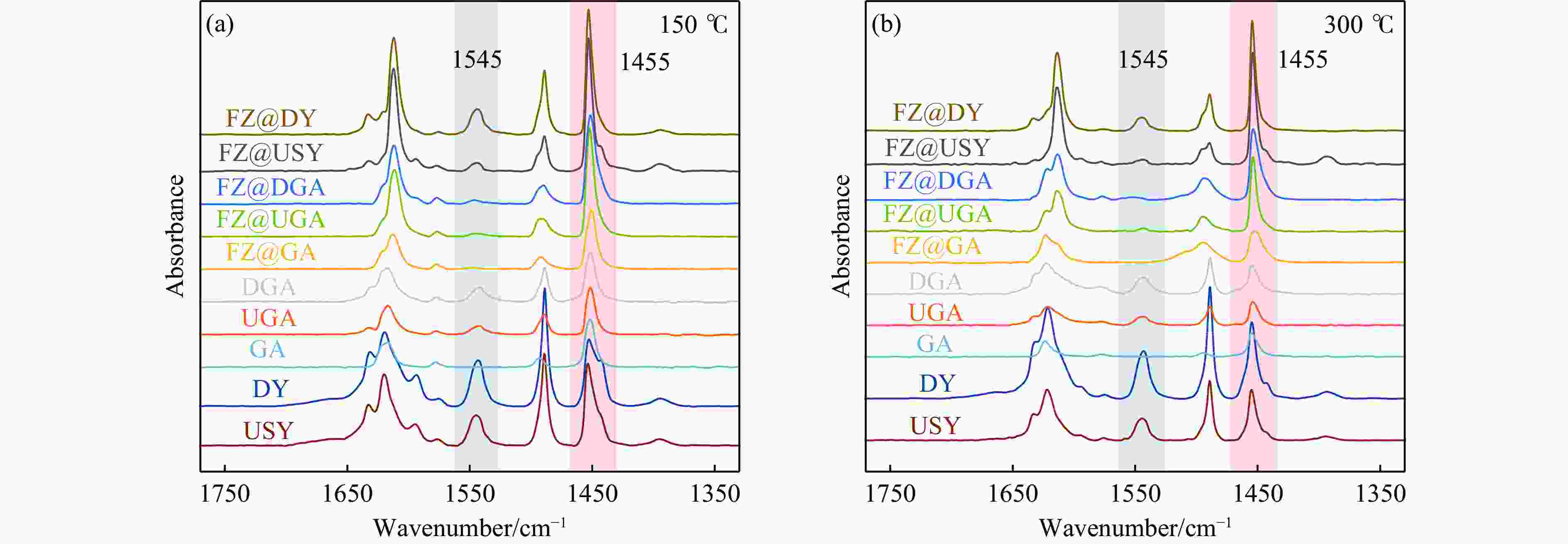
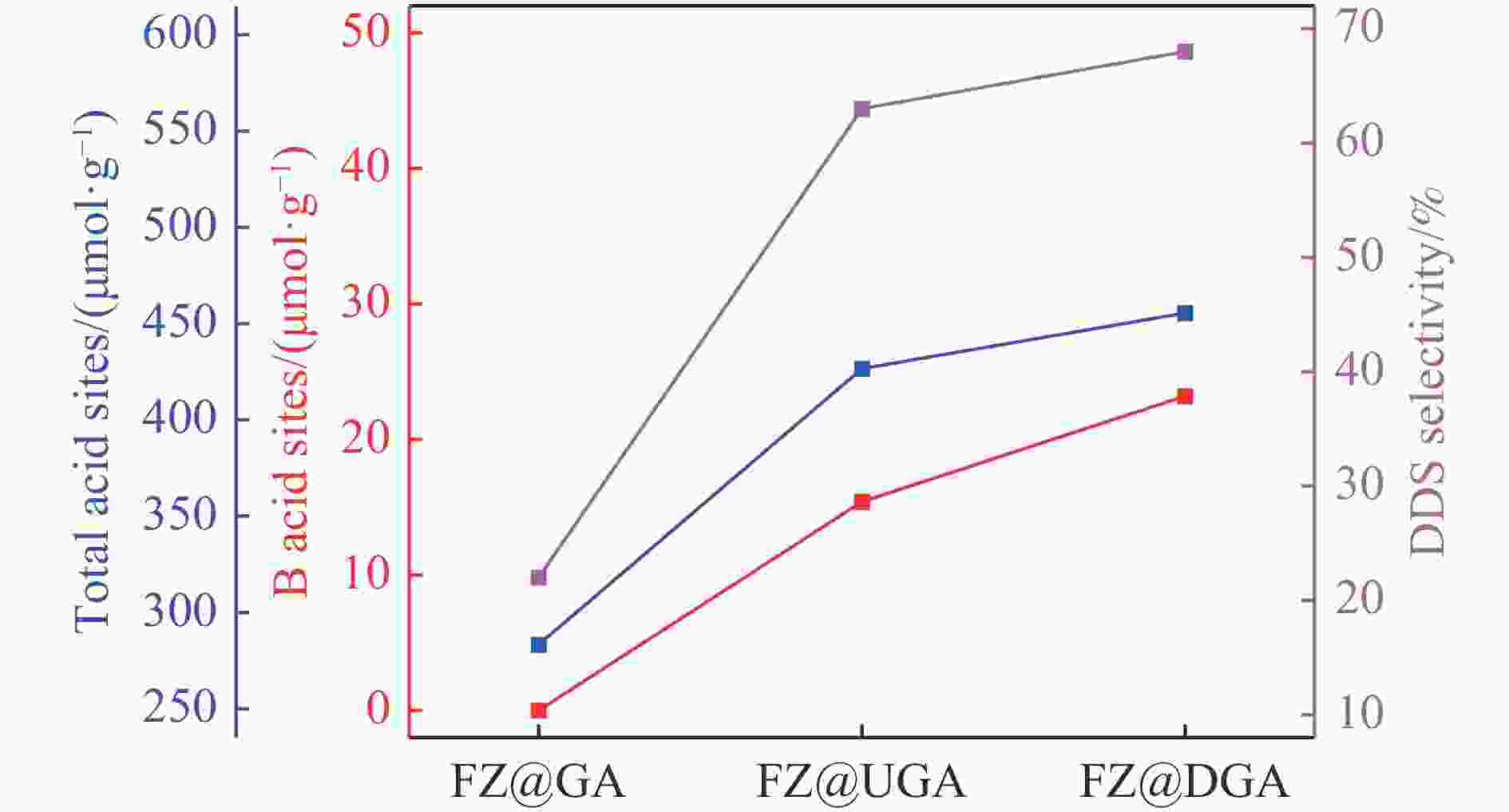
Table 3 Concentrations of B acid sites (1545 cm−1) and L acid sites (1455 cm−1) acquired by Py-IR spectra
Sample Weak acid sites/(μmol·g−1) Strong acid sites/(μmol·g−1) Total/(μmol·g−1) L B L B USY 189.9 105.3 133.0 101.0 529.1 DY 171.8 108.7 258.2 118.3 657.0 GA 198.0 0.0 77.6 0.0 275.7 UGA 150.7 24.9 67.2 46.4 289.2 DGA 148.7 33.9 115.1 98.0 395.7 FZ@GA 159.5 0.0 123.9 0.0 283.3 FZ@UGA 269.5 13.3 141.6 2.1 426.6 FZ@DGA 224.1 17.0 208.2 6.2 455.4 FZ@USY 203.9 90.4 164.0 45.2 503.5 FZ@DY 226.2 136.9 191.7 63.3 618.1 表 4 硫化态Fe基催化剂中Fe物种的组成
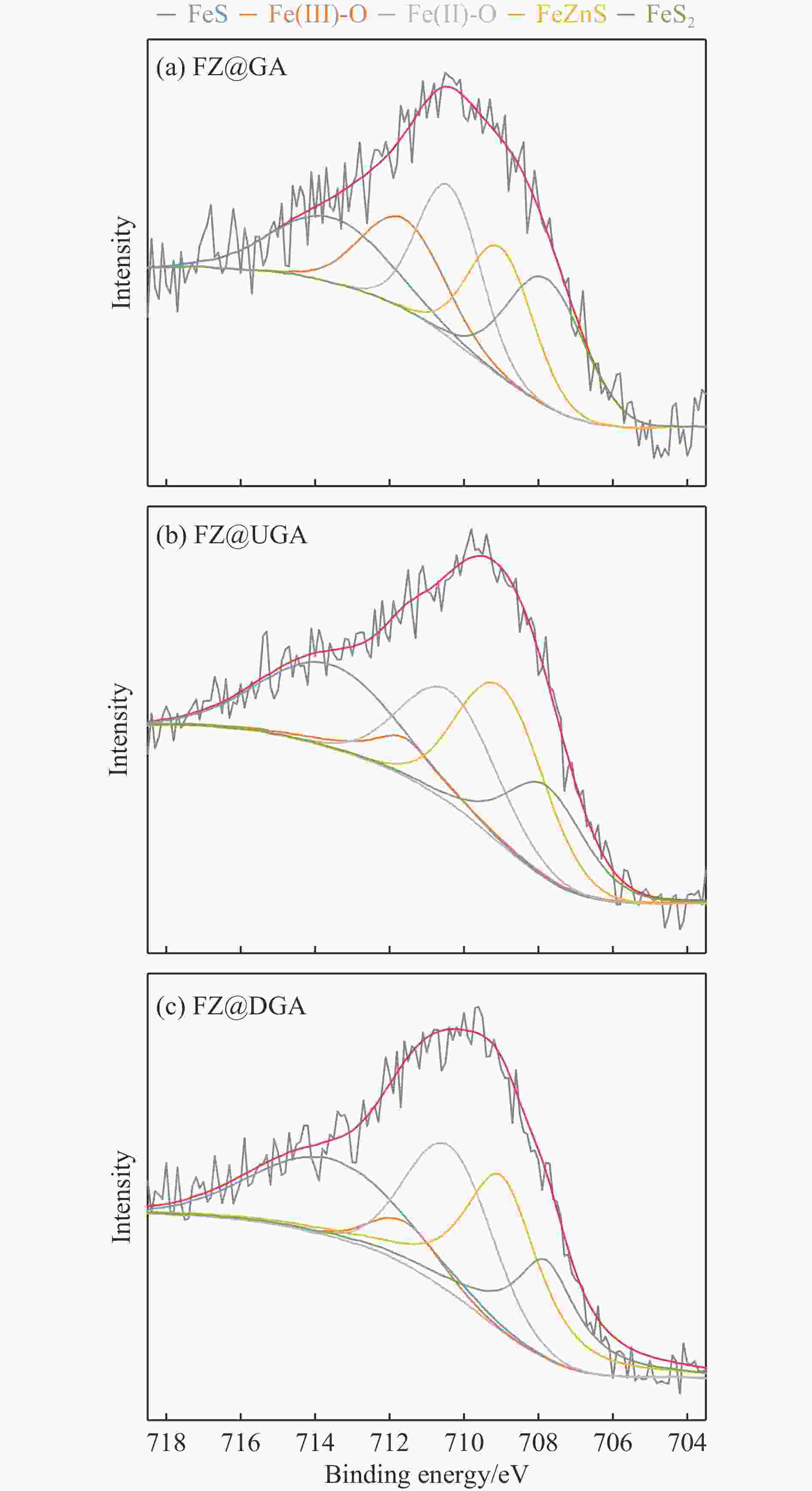
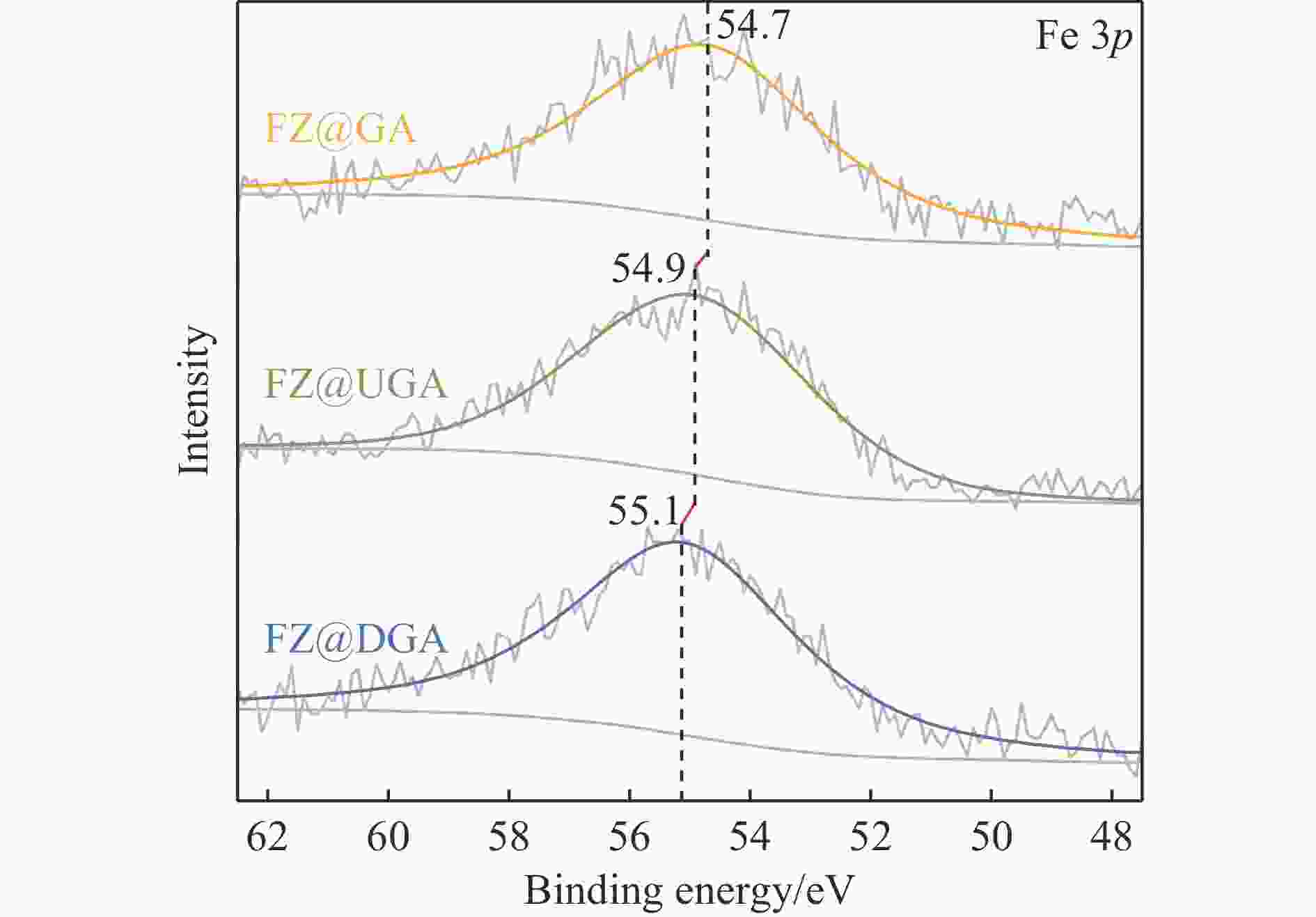
Table 4 Composition of Fe species of sulfide Fe-based catalysts from XPS data
Sample Concentration of Fe species/% SD/% FeS Fe(III)-O Fe(II)-O FeZnS FeS2 FZ@GA 17.7 17.3 22.3 19.6 23.0 60.4 FZ@UGA 23.7 7.8 22.4 28.7 17.4 69.8 FZ@DGA 22.4 6.0 24.0 29.6 18.0 70.0 表 5 Fe基催化剂在催化DBT的HDS过程中的产物分布、反应选择性和反应速率常数
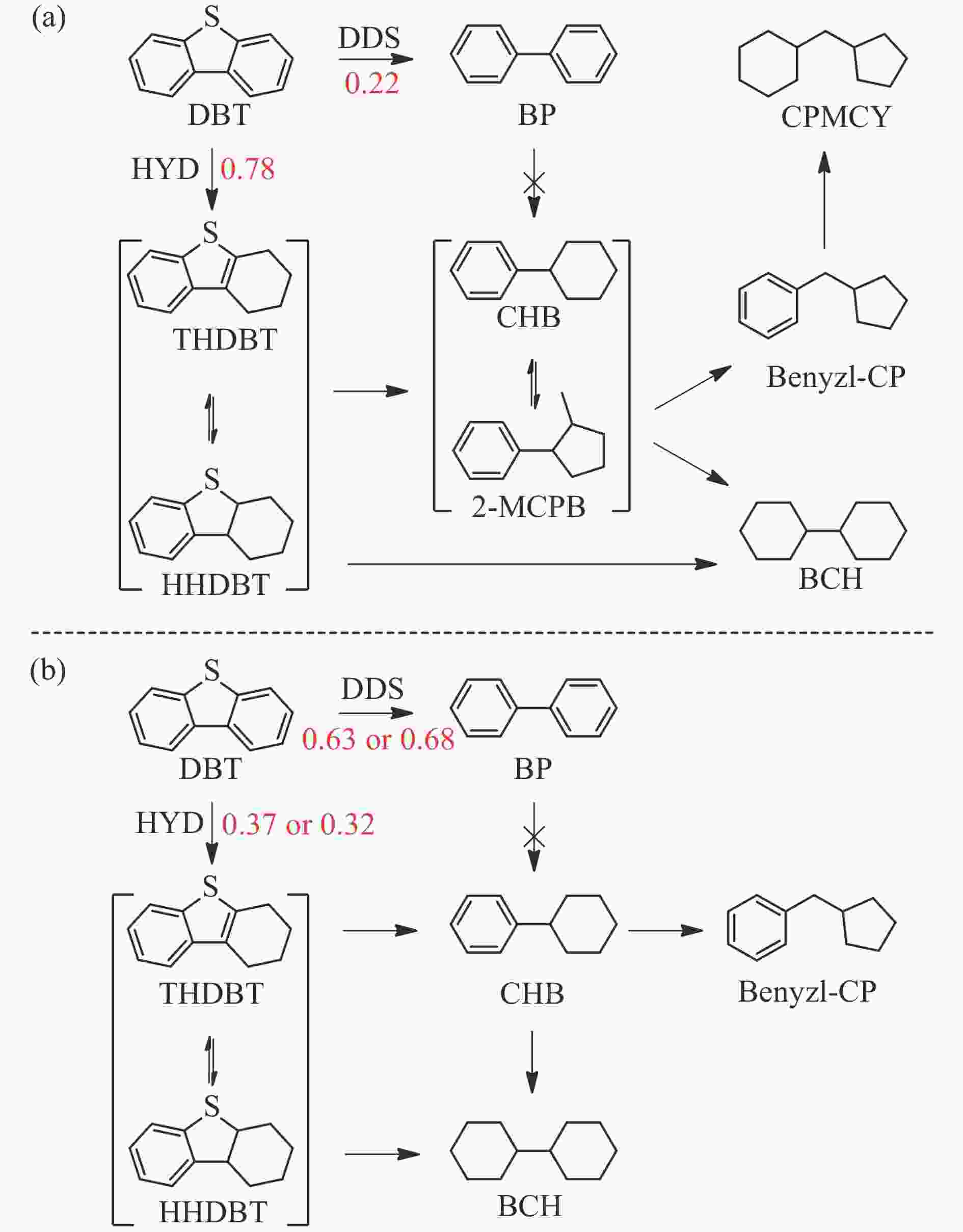
Table 5 Product distributions, pathway selectivity and kHDS of HDS reaction for DBT over Fe-based catalysts
Sample FeZn@GA FeZn@UGA FeZn@DGA CPMCY 9.01 0 0 2-MCPB 3.19 0 0 Benzyl-CP 16.74 4.42 5.32 BCH 6.58 0.97 0.76 CHB 27.63 3.12 2.37 BP 22.20 62.96 68.21 THDBT+HHDBT 14.65 26.75 23.33 kHDS ( × 10−4 mol/(g·h)) 1.05 2.02 2.67 SDDS 22.20 62.96 68.21 SHYD 77.8 37.04 31.79 DDS/HYD 0.29 1.70 2.15 *: The data was determined with approximately 30% of the HDS rate via changing the WHSV at 360 ℃. -
[1] SRIVASTAVA V C. An evaluation of desulfurization technologies for sulfur removal from liquid fuels[J]. RSC Adv,2012,2(3):759−783. doi: 10.1039/C1RA00309G [2] 尹海亮, 刘新亮, 周同娜, 等. NiMo催化剂载体中纳米HY分子筛和氧化铝混合方式对柴油加氢脱硫性能的影响[J]. 燃料化学学报,2018,46(8):950−956. doi: 10.1016/S1872-5813(18)30038-0YIN Hailiang, LIU Xinliang, ZHOU Tongna, et al. Effect of preparation method of nanosized zeolite HY-Al2O3 composite as NiMo catalyst support on diesel HDS[J]. J Fuel Chem Technol,2018,46(8):950−956. doi: 10.1016/S1872-5813(18)30038-0 [3] 张亮亮, 汪镭, 陈霄, 等. Co2Si@C催化剂的合成及其加氢脱硫性能[J]. 分子催化,2020,34(2):116−124.ZHANG Liangliang, WANG Lei, CHEN Xiao, et al. Synthesis of Co2Si@C and its catalytic performance in the hydrodesulphurization of dibenzothiophene[J]. Mol Catal,2020,34(2):116−124. [4] ASIF M, MUNEER T. Energy supply, its demand and security issues for developed and emerging economies[J]. Renewablet Sustainable Energy Rev,2007,11(7):1388−1413. doi: 10.1016/j.rser.2005.12.004 [5] TANIMU A, ALHOOSHANI K. Advanced hydrodesulfurization catalysts: A review of design and synthesis[J]. Energy Fuels,2019,33(4):2810−2838. doi: 10.1021/acs.energyfuels.9b00354 [6] WENG X, CAO L, ZHANG G, et al. Ultradeep hydrodesulfurization of diesel: Mechanisms, catalyst design strategies, and challenges[J]. Ind Eng Chem Res,2020,59(49):21261−21274. doi: 10.1021/acs.iecr.0c04049 [7] 于志庆, 黄文斌, 王晓晗, 等. B 掺杂Al2O3@C 负载CoMo 型加氢脱硫催化剂性能[J]. 化工进展,2023,42(7):3550−3560.YU Zhiqing, HUANG Wenbin, WANG Xiaohan, et al. B-doped Al2O3@C support for CoMo hydrodesulfurization catalyst and their hydrodesulfurization performance[J]. Chem Ind Eng Prog,2023,42(7):3550−3560. [8] 刘娟, 李文英, 冯杰, 等. Ni对MoS2基催化剂活性相及加氢脱氮脱硫性能的影响[J]. 燃料化学学报,2021,49(10):1513−1521.LIU Juan, LI Wenying, FENG Jie, et al. Influence of Ni on the active phase and hydrodenitrogenation and hydrodesulfurization activities of MoS2 catalysts[J]. J Fuel Chem Technol,2021,49(10):1513−1521. [9] 孙嫚, 夏少青, 戴薇薇, 等. M-PHG技术在国Ⅵ汽油质量升级改造项目中的应用[J]. 中外能源,2022,27(4):67−70.SUN Man, XIA Shaoqing, DAI Weiwei, et al. Application of M-PHG technology in national Ⅵ gasoline quality upgrading project[J]. Sino Global Energy,2022,27(4):67−70. [10] 孟欣欣, 邱泽刚, 郭兴梅, 等. 不同金属含量Ni-W催化剂的煤焦油加氢脱硫脱氮性能研究[J]. 燃料化学学报,2016,44(5):570−578.MENG Xinxin, QIU Zegang, GUO Xingmei, et al. Hydrodenitrogenation and hydrodesulfurization of coal tar on Ni-W catalysts with different metal loadings[J]. J Fuel Chem Technol,2016,44(5):570−578. [11] 孙进, 郭蓉, 陈晓贞, 等. 助剂Co对加氢处理催化剂性能的影响[J]. 石油炼制与化工,2023,54(6):32−38. doi: 10.3969/j.issn.1005-2399.2023.06.007SUN Jin, GUO Rong, CHEN Xiaozhen, et al. Effect of promoter cobalt on the performance of hydrotreating catalysts[J]. Pet Process Petrochem,2023,54(6):32−38. doi: 10.3969/j.issn.1005-2399.2023.06.007 [12] TOPSØE H. The role of Co-Mo-S type structures in hydrotreating catalysts[J]. Appl Catal A: Gen,2007,322:3−8. doi: 10.1016/j.apcata.2007.01.002 [13] TUXEN A K, FÜCHTBAUER H G, TEMEL B, et al. Atomic-scale insight into adsorption of sterically hindered dibenzothiophenes on MoS2 and Co-Mo-S hydrotreating catalysts[J]. J Catal,2012,295:146−154. doi: 10.1016/j.jcat.2012.08.004 [14] VÍT Z, GULKOVÁ D, KALUŽA L, et al. Pd-Pt catalysts on mesoporous SiO2-Al2O3 with superior activity for HDS of 4, 6-dimethyldibenzothiophene: Effect of metal loading and support composition[J]. Appl Catal B: Environ,2015,179:44−53. doi: 10.1016/j.apcatb.2015.04.057 [15] GALINDO-ORTEGA Y, INFANTES-MOLINA A, HUIRACHE-ACUÑA R, et al. Active ruthenium phosphide as selective sulfur removal catalyst of gasoline model compounds[J]. Fuel Process Technol,2020,208:106507. doi: 10.1016/j.fuproc.2020.106507 [16] MAJODINA S, TSHENTU Z. R, OGUNLAJA A S. Effect of adding chelating ligands on the catalytic performance of Rh-promoted MoS2 in the hydrodesulfurization of dibenzothiophene[J]. Catal,2021,11(11):1398. doi: 10.3390/catal11111398 [17] INFANTES-MOLINA A, ROMERO-PÉREZ A, FINOCCHIO E, et al. HDS and HDN on SBA-supported RuS2 catalysts promoted by Pt and Ir[J]. J Catal,2013,305:101−117. doi: 10.1016/j.jcat.2013.05.001 [18] 杨晓东, 关旭, 高善彬, 等. 载体性质对Pd催化剂加氢脱硫性能的影响[J]. 燃料化学学报,2017,45(8):980−985.YANG Xiaodong, GUAN Xu, GAO Shanbin, et al. Effect of support properties on the performance of supported Pd catalysts in hydrodesulfurization[J]. J Fuel Chem Technol,2017,45(8):980−985. [19] SCHACHT P, HERNÁNDEZ G, CEDEÑO L, et al. Hydrodesulfurization activity of CoMo catalysts supported on stabilized TiO2[J]. Energy Fuels,2003,17(1):81−86. doi: 10.1021/ef020144u [20] HUBAUT R. Vanadium-based sulfides as hydrotreating catalysts[J]. Appl Catal A: Gen,2007,322:121−128. doi: 10.1016/j.apcata.2007.01.020 [21] PUELLO-POLO E, GUTIÉRREZ-ALEJANDRE A, GONZÁLEZ G, et al. Relationship between sulfidation and HDS catalytic activity of activated carbon supported Mo, Fe-Mo, Co-Mo and Ni-Mo carbides[J]. Catal Lett,2010,135(3/4):212−218. doi: 10.1007/s10562-010-0303-6 [22] MÉNDEZ F J, FRANCO-LÓPEZ O E, DÍAZ G, et al. On the role of niobium in nanostructured Mo/Nb-MCM-41 and NiMo/Nb-MCM-41 catalysts for hydrodesulfurization of dibenzothiophene[J]. Fuel,2020,280:118550. doi: 10.1016/j.fuel.2020.118550 [23] XIE J, LU H, SHU G, et al. The relationship between the microstructures and catalytic behaviors of iron-oxygen precursors during direct coal liquefaction[J]. Chin J Catal,2018,39(4):857−866. doi: 10.1016/S1872-2067(17)62919-X [24] ANDERSON J S, RITTLE J, PETERS J C. Catalytic conversion of nitrogen to ammonia by an iron model complex[J]. Nature,2013,501(7465):84−87. doi: 10.1038/nature12435 [25] VAN STEEN E, CLAEYS M. Fischer-Tropsch catalysts for the biomass to liquid process[J]. Chem Eng Technol,2008,31(5):655−666. doi: 10.1002/ceat.200800067 [26] 刘化章, 李小年. Fe1−xO 基氨合成催化剂高活性机理初探[J]. 催化学报,2005,26(1):79−86.LIU Huazhang, LI Xiaonian. Study on mechanism of high activity of Fe1−xO-based catalyst for ammonia synthesis[J]. Chin J Catal,2005,26(1):79−86. [27] LI H, LIU J, LI J, et al. Promotion of the inactive iron sulfide to an efficient hydrodesulfurization catalyst[J]. ACS Catal,2017,7(7):4805−4816. doi: 10.1021/acscatal.6b03495 [28] LIU P, LI Z, LIU X, et al. Steaming drived chemical interactions of ZnClx with Y zeolite framework, its regulation to dealumination/silicon-healing as well as enhanced availability of Brønsted acidity[J]. ACS Catal,2020,10(16):9197−9214. doi: 10.1021/acscatal.0c01181 [29] EMEIS C A. Determination of integrated molar extinction coefficients for infrared absorption bands of pyridine adsorbed on solid acid catalysts[J]. J Catal,1993,141(2):347−354. doi: 10.1006/jcat.1993.1145 [30] LIU X, LIU J, LI L, et al. Hydrodesulfurization of dibenzothiophene on TiO2–x modified Fe-based catalysts: Electron transfer behavior between TiO2–x and Fe species[J]. ACS Catal,2020,10:9019−9033. doi: 10.1021/acscatal.0c01068 [31] PEÑA L, VALENCIA D, KLIMOVA T. CoMo/SBA-15 catalysts prepared with EDTA and citric acid and their performance in hydrodesulfurization of dibenzothiophene[J]. Appl Catal B: Environ,2014,147:879−887. doi: 10.1016/j.apcatb.2013.10.019 [32] HEINRICH F, SCHMIDT C, LÖFFLER E, et al. Fe-ZSM-5 catalysts for the selective reduction of NO by isobutane-The problem of the active sites[J]. J Catal,2002,212(2):157−172. doi: 10.1006/jcat.2002.3775 [33] LIANG M, KANG W, XIE K. Comparison of reduction behavior of Fe2O3, ZnO and ZnFe2O4 by TPR technique[J]. J Nat Gas Chem,2009,18(1):110−113. doi: 10.1016/S1003-9953(08)60073-0 [34] WANG J, WANG Y, XIE S, et al. Partial hydrogenation of benzene to cyclohexene on a Ru-Zn/m-ZrO2 nanocomposite catalyst[J]. Appl Catal A: Gen,2004,272(1):29−36. [35] VRINAT M L, GACHET C G, DE MOURGUES L. Catalytic hydrodesulfurization of dibenzothiophene over Y type zeolites[C]//Studies in Surface Science and Catalysis. Amsterdam: Elsevier, 1980: 219−225. [36] TANG H, LI Q, SONG Z, et al. Enhancement of desulfurization performance of nickel-based adsorbents by hydrogen reduction pretreatment[J]. Catal Commun,2011,12(12):1079−1083. doi: 10.1016/j.catcom.2011.03.022 [37] ZHANG X, ZHANG B, CHEN Y, et al. Silica improved formation of Fe(III)-S and electron-deficient effect in Fe-based catalysts to improve hydrodesulfurization[J]. Fuel,2022,307:121787. doi: 10.1016/j.fuel.2021.121787 [38] HAN W, NIE H, LONG X, et al. Effects of the support Brønsted acidity on the hydrodesulfurization and hydrodenitrogention activity of sulfided NiMo/Al2O3 catalysts[J]. Catal Today,2017,292:58−66. doi: 10.1016/j.cattod.2016.11.049 [39] JIAO J, FU J, WEI Y, et al. Al-modified dendritic mesoporous silica nanospheres-supported NiMo catalysts for the hydrodesulfurization of dibenzothiophene: Efficient accessibility of active sites and suitable metal-support interaction[J]. J Catal,2017,356:269−282. [40] FAN Y, XIAO H, SHI G, et al. Citric acid-assisted hydrothermal method for preparing NiW/USY-Al2O3 ultradeep hydrodesulfurization catalysts[J]. J Catal,2011,279(1):27−35. doi: 10.1016/j.jcat.2010.12.014 [41] HESSOU E P, JABRAOUI H, KHALIL I, et al. Ab initio screening of zeolite Y formulations for efficient adsorption of thiophene in presence of benzene[J]. Appl Surf Sci,2021,541:148515. doi: 10.1016/j.apsusc.2020.148515 [42] XIA B, CAO L, LUO K, et al. Effects of the active phase of CoMo/γ-Al2O3 catalysts modified using cerium and phosphorus on the HDS performance for FCC gasoline[J]. Energy Fuels,2019,33(5):4462−4473. doi: 10.1021/acs.energyfuels.8b04332 [43] HAN W, NIE H, LONG X, et al. Preparation of F-doped MoS2/Al2O3 catalysts as a way to understand the electronic effects of the support Brønsted acidity on HDN activity[J]. J Catal,2016,339:135−142. doi: 10.1016/j.jcat.2016.04.005 [44] WEI W, ZHANG X, LIU X, et al. Tuning effect of the zeolite Brønsted acidity on the FeZn bimetallic hydrodesulfurization catalyst[J]. Energy Fuels,2022,36(1):527−538. doi: 10.1021/acs.energyfuels.1c03142 [45] MGUNI L L, YAO Y, NKOMZWAYO T, et al. Desulphurization of diesel fuels using intermediate Lewis acids loaded on activated charcoal and alumina[J]. Chem Eng Commun,2019,206(5):572−580. doi: 10.1080/00986445.2018.1511983 [46] VILLARROEL M, BAEZA P, ESCALONA N, et al. MD//Mo and MD//W [MD=Mn, Fe, Co, Ni, Cu and Zn] promotion via spillover hydrogen in hydrodesulfurization[J]. Appl Catal A: Gen,2008,345(2):152−157. doi: 10.1016/j.apcata.2008.04.033 [47] COSTA C S, THI H D, VAN GEEM K M, et al. Assessment of acidity and the zeolite porous structure on hydrocracking of HDPE[J]. Sustainable Energy Fuels,2022,6:3611−3625. [48] SUN H, SUN H, ZHANG X, et al. Effect of divalent tin on the SnSAPO-5 molecular sieve and its modulation to alumina support to form a highly efficient NiW catalyst for deep hydrodesulfurization of 4, 6-dimethyldibenzothiophene[J]. ACS Catal,2019,9(8):6613−6623. doi: 10.1021/acscatal.9b01668 [49] LIU X, LIU J, LI L, et al. Preparation of electron-rich Fe-based catalyst via electronic structure regulation and its promotion to hydrodesulfurization of dibenzothiophene[J]. Appl Catal B: Environ,2020,269(269):118779. [50] YUE S. , WU B., CAO Z., et al. Cobalt promoted molybdenum carbide supported on γ-alumina as an efficient catalyst for hydrodesulfurization of dibenzothiophene[J]. J Cleaner Prod,2022,371:133642. doi: 10.1016/j.jclepro.2022.133642 [51] CHEN W, NIE H, LI D, et al. Effect of Mg addition on the structure and performance of sulfide Mo/Al2O3 in HDS and HDN reaction[J]. J Catal,2016,344:420−433. doi: 10.1016/j.jcat.2016.08.025 [52] SHAFIQ I, SHAFIQUE S, AKHTER P, et al. Recent developments in alumina supported hydrodesulfurization catalysts for the production of sulfur-free refinery products: A technical review[J]. Catal Rev,2022,64(1):1−86. doi: 10.1080/01614940.2020.1780824 [53] LIU X, LI L, SUN H, et al. NiW catalyst modified with C12A7-H- and its promotion to hydrogenation selectivity of hydrodesulfurization[J]. Fuel,2021,290:120037. doi: 10.1016/j.fuel.2020.120037 [54] LI L, WANG M, HUANG L, et al. Electron-donating-accepting behavior between nitrogen-doped carbon materials and Fe species and its promotion for DBT hydrodesulfurization[J]. Appl Catal B: Environ,2019,254:360−370. doi: 10.1016/j.apcatb.2019.05.011 [55] CAO Z, ZHANG X, GUO R, et al. Synergistic effect of acidity and active phases for NiMo catalysts on dibenzothiophene hydrodesulfurization performance[J]. Chem Eng J,2020,400:125886. doi: 10.1016/j.cej.2020.125886 -




 下载:
下载: