Comparative study on the catalytic performance of zeolite catalysts with different topologies in 1-methylnaphthalene isomerization-transalkylation coupling reaction
-
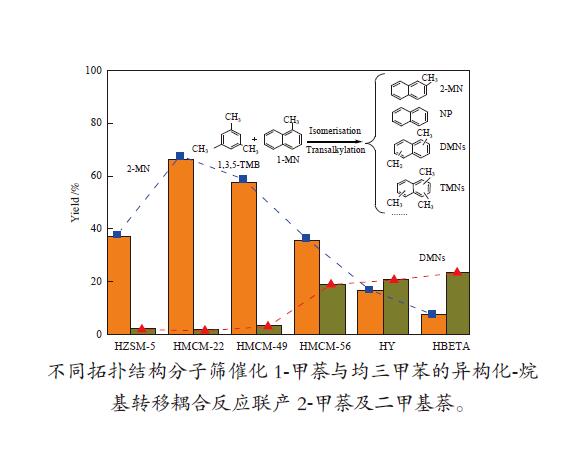
摘要: 以1-甲萘和均三甲苯为原料经固体酸催化的异构化-烷基转移耦合反应制备2-甲萘及二甲基萘。对MWW、BEA、FAU、MFI拓扑结构分子筛的催化性能进行对比研究,对反应机理进行探讨。采用XRD、BET、SEM、NH3-TPD、Py-FTIR及ICP等方法对催化剂结构物性进行了表征。与具有十元环特征孔道的MFI分子筛相比,具有十二元环特征孔道的MWW、BEA、FAU分子筛表现出更好的催化活性。BEA结构分子筛具有较高的二甲基萘选择性,而MWW结构分子筛具有较高的2-甲萘选择性并表现出优异的催化稳定性。在MWW结构分子筛中,HMCM-22的1-甲萘转化率达到70.27%,2-甲萘收率达到66.69%。而HMCM-56上则同时获得35.74%的2-甲萘收率和19.00%的二甲基萘收率。该研究为以中国丰富的碳十资源为原料制备高端聚酯单体2,6-二甲基萘开辟了颇具潜力的技术路线。Abstract: The isomerization-transalkylation coupling reaction between 1-methylnaphthalene and 1,3,5-trimethylbenzene was studied over zeolite catalysts to produce 2-methylnaphthalene and dimethylnaphthalene. The catalytic performance of zeolites with MWW, BEA, FAU and MFI topological structure was investigated and the reaction mechanism was discussed. The properties of zeolites were characterized by XRD, N2 adsorption-desorption, SEM, NH3-TPD, Py-FTIR and ICP. Compared with MFI zeolite with 10-membered ring channel, MWW, BEA and FAU zeolite with 12-membered ring channel showed better catalytic activity. BEA zeolite exhibited higher selectivity to dimethylnaphthalene, while MWW zeolites showed higher selectivity to 2-methylnaphthalene and the best catalytic stability. As a branch of MWW zeolites, HMCM-22 showed a 1-methylnaphthalene conversion of 70.27% and a 2-methylnaphthalene yield of 66.69%. It was worth noting that a relatively high yield of 2-methylnaphthalene (35.74%) and dimethylnaphthalene (19.00%) were obtained on HMCM-56 at the same time. This research has opened up a promising route using C10 aromatics as raw materials for the preparation of 2,6-dimethylnaphthalene, an important polyester monomer.
-
Key words:
- molecular sieve /
- 1-methylnaphthalene /
- isomerization /
- transalkylation /
- 2,6-dimethylnaphthalene
-
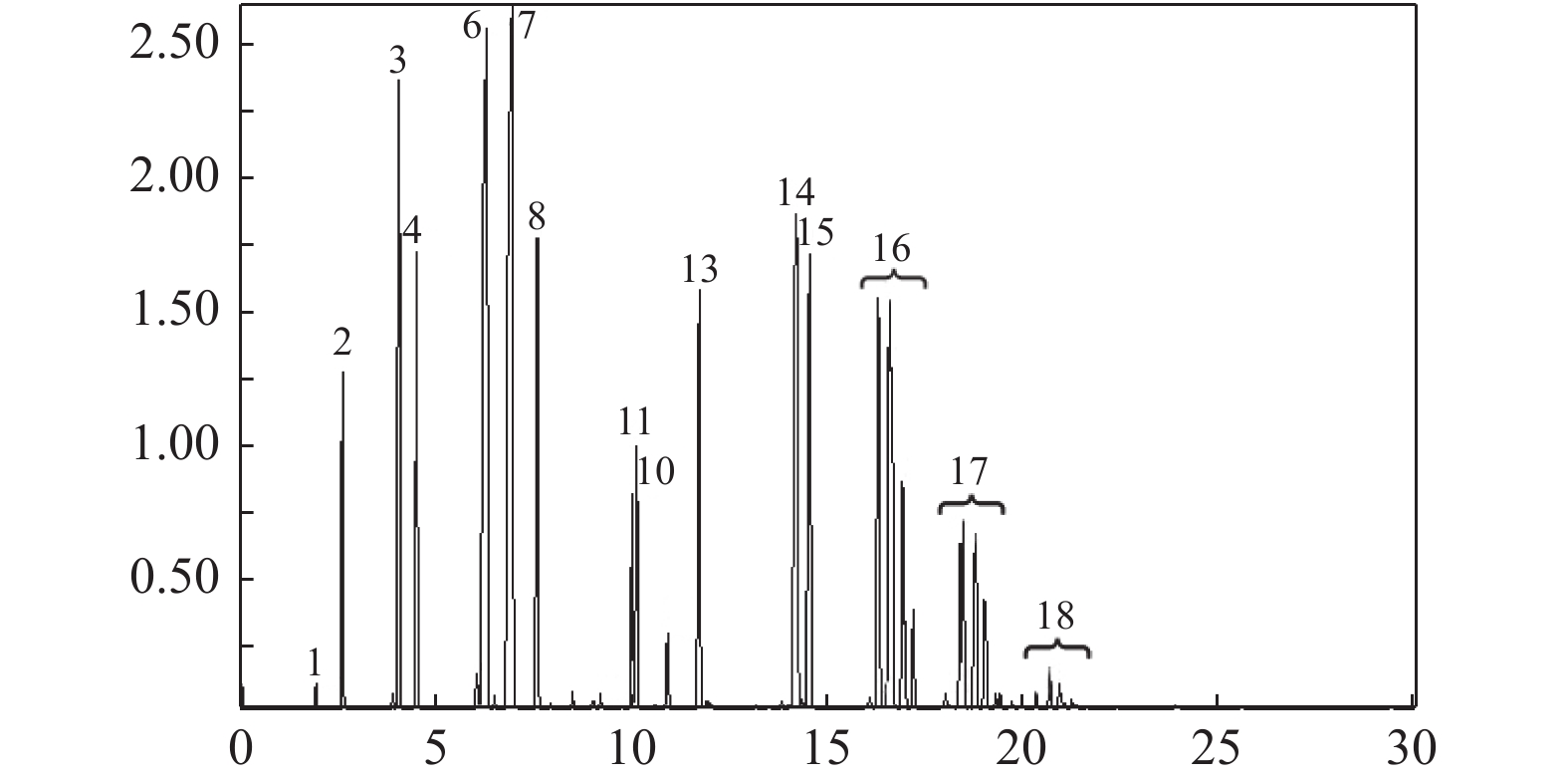
图 7 1-MN与1,3,5-TMB异构化-烷基转移耦合反应产物的质谱图
Figure 7 Mass spectrometric analysis of the coupling reaction of 1-MN and 1,3,5-TMB isomerization-transalkylation
1: benzene; 2: toluene; 3–4: dimethylbenzene; 5: 1-ethyl-2-methylbenzene; 6: 1,3,5-trimethylbenzene; 7: 1,2,3-TMB (1,2,3-trimethylbenzene); 8: 1,2,4-TMB (1,2,4-trimethylbenzene; 9–12: tetramethylbenzene; 13: naphthalene; 14: 2-methylnaphthalene; 15: 1-methylnaphthalene;16: dimethylnaphthalene; 17: trimethylnaphthalene; 18: polymethyl naphthalene
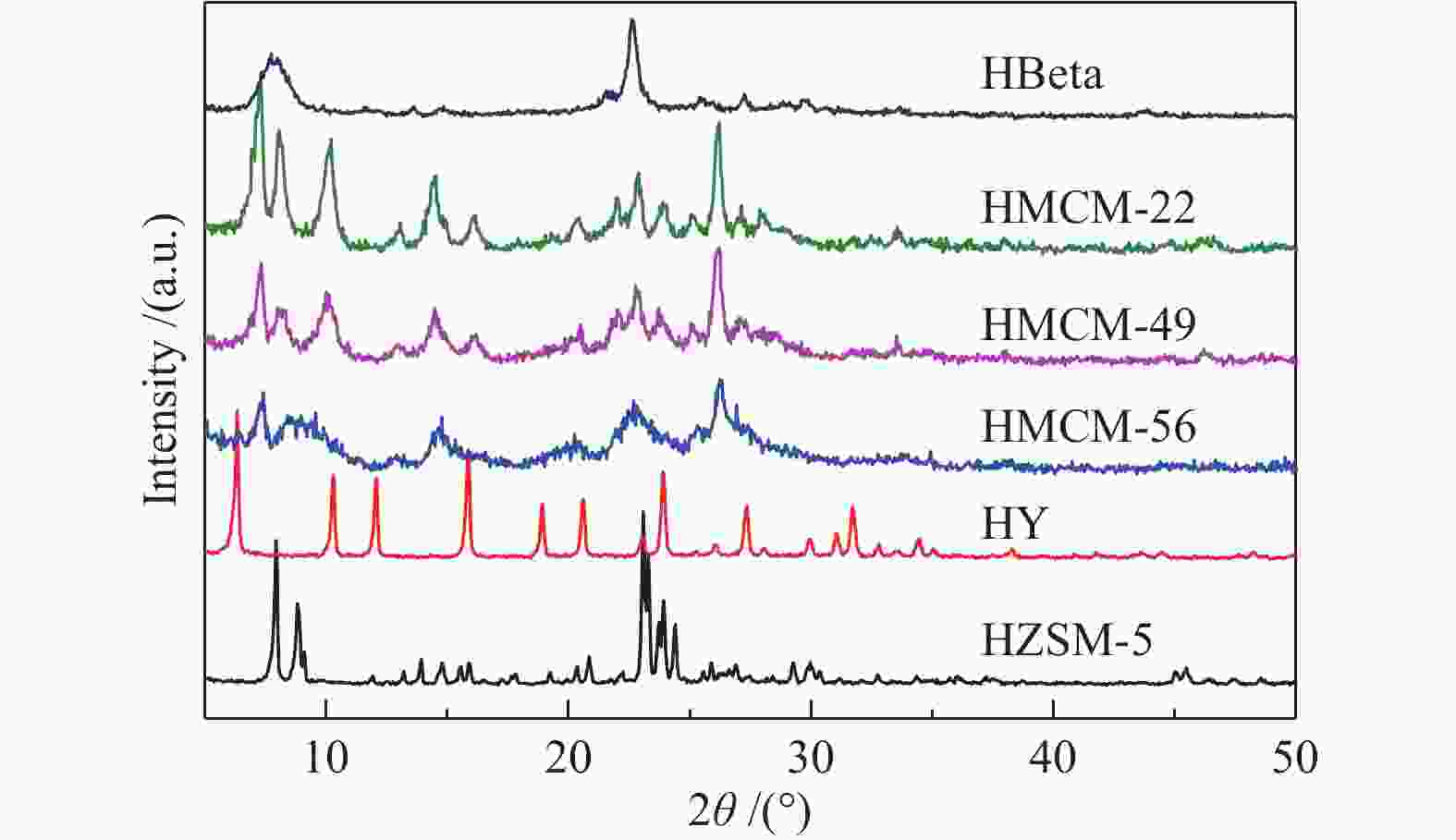
表 1 样品的孔结构参数
Table 1 Textural properties of the samples
Sample ABET/
(m2∙g−1)Aext /
(m2∙g−1)vtotal/
(cm3∙g−1)vmic/
(cm3∙g−1)vmes/
(cm3∙g−1)Aext/
ABETHZSM-5 326 87 0.24 0.09 0.10 0.48 HBeta 580 160 0.50 0.19 0.32 0.27 HY 635 138 0.47 0.40 0.08 0.21 HMCM-22 444 106 0.68 0.14 0.56 0.24 HMCM-49 447 86 0.76 0.15 0.64 0.19 HMCM-56 499 177 1.32 0.13 1.23 0.36 表 2 元素组成、NH3-TPD和Py-FTIR测试
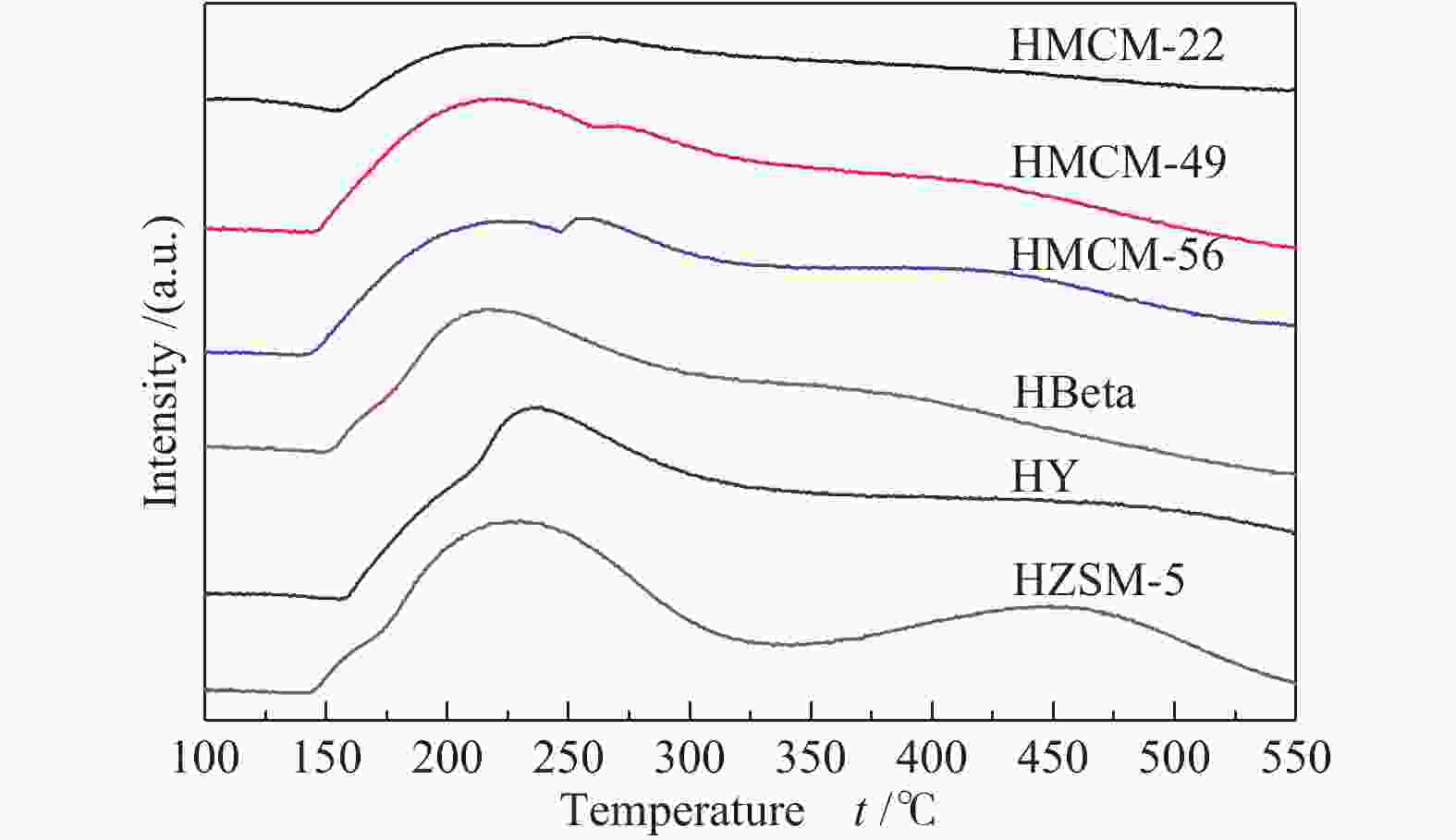
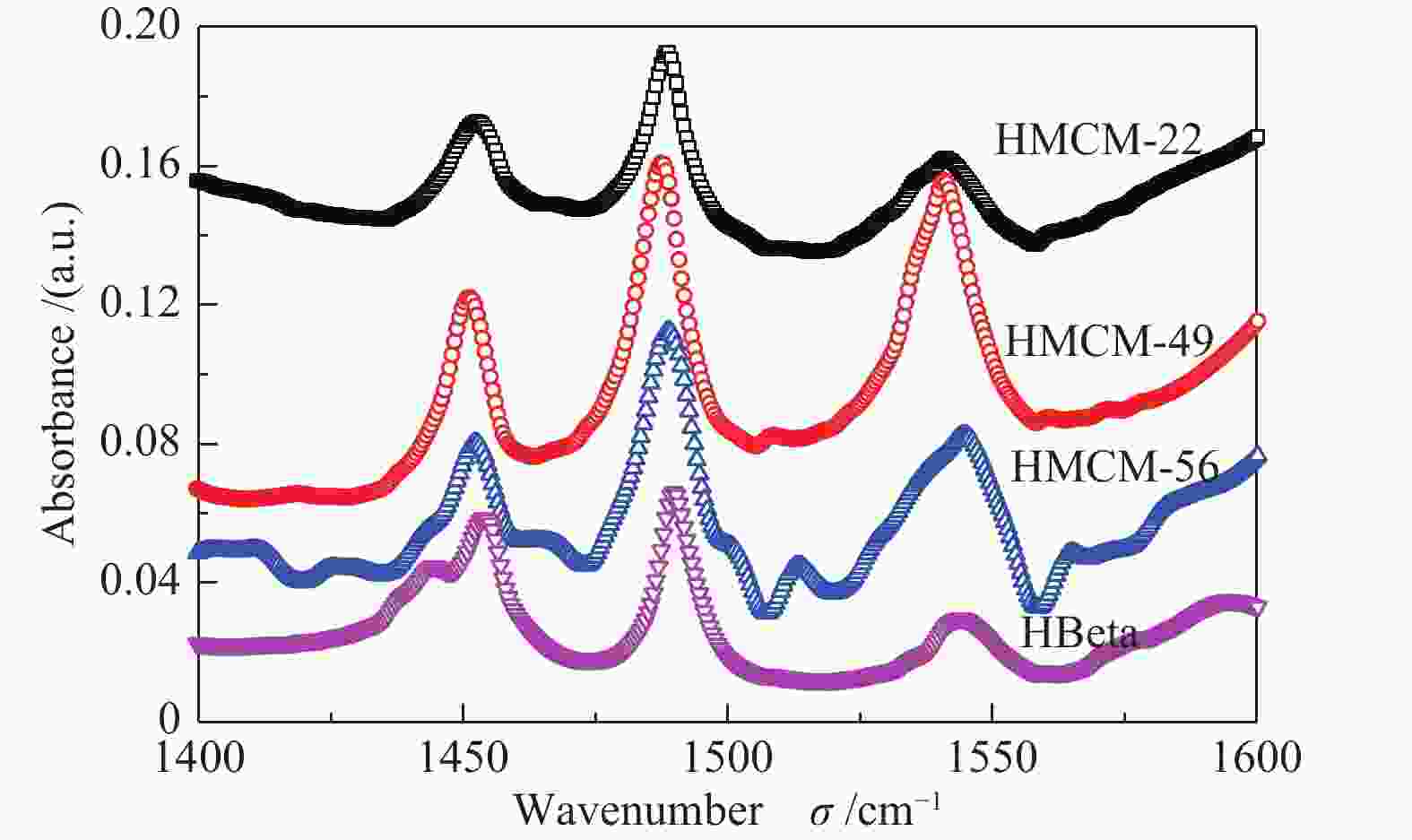
Table 2 Element composition, NH3-TPD and Py-FTIR results
Sample SiO2/Al2O3a Desorption peak area of NH3-TPDb Rc Acidity /(μmol∙g−1) Brønsted/Lewis A1(100−250 ℃) A2(250−400 ℃) A3(400−550 ℃) Brønsted Lewis HZSM-5 24.2 1345 570 1039 0.5 − − − HY 5.1 886 1383 600 0.7 − − − HBeta 17.8 805 1692 − 0.7 63 118 0.5 HMCM-22 13.1 368 906 − 0.7 124 61 2.0 HMCM-49 9.6 958 1673 − 0.6 287 103 2.8 HMCM-56 11.8 1227 1230 − 0.5 209 67 3.1 a: measured by ICP method; b: calculated by Gaussian fitting method; c: (A2 + A3) to (A1 + A2 + A3) ratio 表 3 分子筛催化异构化-烷基转移耦合反应催化性能a
Table 3 Catalytic performance of isomerization-transalkylation of 1-MN with 1,3,5-TMB over zeolites a
Catalysts HZSM-5 HMCM-22 HMCM-49 HMCM-56 HBeta HY Conversion x/% 1-MN 77.56 70.27 78.04 80.70 93.34 89.71 1,3,5-TMB 28.14 63.53 69.58 76.88 80.63 50.93 Distributionb s/% 2-MN 91.16 94.46 89.28 55.54 13.74 29.00 DMNs 5.56 2.53 5.28 29.52 42.76 35.95 TMNs 1.20 2.14 1.85 7.39 32.53 15.63 NP 2.08 0.87 3.59 7.55 10.97 19.42 Yieldc w/% 2-MN 37.05 66.34 57.66 35.74 7.53 16.70 DMNs 2.26 1.82 3.41 19.00 23.41 20.69 2,6-DMN/DMNs 0.55 0.37 0.41 0.35 0.38 0.34 Yieldd w/% 2-MN 5.58 66.69 55.95 52.69 53.18 52.29 DMNs 2.04 0.93 2.46 9.23 5.32 6.15 2,6-DMN/DMNs 0.50 0.46 0.38 0.36 0.41 0.37 a: t = 400 ℃, WHSV1-MN = 0.5 h−1, n1-MN/nTMB = 1/3, p = 0.2 MPa, T.O.S = 1 h
b: Mole of specific product×100/mole of 1-MN derivatives, including NP (naphthalene), 2-MN (2-methylnaphthalene), DMNs (dimethyl naphthalene) and TMNs (trimethyl naphthalene)
c: Mole of 2-MN (or DMNs) in product×100/mole of 1-MN in raw materials,T.O.S = 1 h
d: T.O.S = 6 h -
[1] 唐文秀. 从炼焦洗油中分离提纯β-甲基萘的研究[J]. 石油化工应用,2018,37(10):97−100. doi: 10.3969/j.issn.1673-5285.2018.10.022TANG Wen-xiu. Study on separation and purification of β-methyl naphthalene from coking wash oil[J]. Chin Petroch Indus App,2018,37(10):97−100. doi: 10.3969/j.issn.1673-5285.2018.10.022 [2] 田苗, 索隆宁, 田红, 魏元博. β-甲基萘氧化合成维生素K3(1,4-甲萘醌)的研究进展[J]. 云南化工,2019,46(8):51−52. doi: 10.3969/j.issn.1004-275X.2019.08.018TIAN Miao, SUO Long-ning, TIAN Hong, WEI Yuan-bo. Research progress of β-methylnaphthalene oxidation to synthesize vitamin K3 (1,4-menadione)[J]. Yunnan Chem Technol,2019,46(8):51−52. doi: 10.3969/j.issn.1004-275X.2019.08.018 [3] LI C, LI L, WU W, DONGSHENG W, TOKTAREV A V, KIKHTYANIN O V, ECHEVSKII G V. Highly selective synthesis of 2,6-dimethylnaphthalene over alkaline treated ZSM-12 zeolite[J]. Procedia Eng,2011,18:200−205. doi: 10.1016/j.proeng.2011.11.032 [4] NIE X, JANIK M J, GUO X, WANG X, SONG C, LIU H. Shape-selective methylation of 2-methylnaphthalene with methanol over H-ZSM–5 zeolite: A computational study[J]. J Phys Chem C,2015,116(6):4071−4082. [5] ZHANG C, GUO X W, SONG C S, ZHAO S Q, WANG X S. Effects of steam and TEOS modification on HZSM-5 zeolite for 2,6-dimethylnaphthalene synthesis by methylation of 2-methylnaphthalene with methanol[J]. Catal Today,2010,149(1/2):196−201. doi: 10.1016/j.cattod.2009.04.015 [6] SANHOOB M A, MURAZA O, TAGO T, TANIGUCHI T, WATANABE G, MASUDA T. Development of mesoporous ZSM-12 zeolite and its application in alkylation of 2-methylnaphthalene[J]. Res Chem Intermed,2016,42(7):1−12. [7] 栾珊, 靳立军, 郭学华, 于泳, 胡浩权, 王亚涛. 介孔ZSM-5沸石的制备及在2-甲基萘甲基化反应中的应用[J]. 石油学报(石油加工),2014,30(2):204−204.LUAN Shan, ZHAN Li-jun, GUO Xue-hua, YU Yong, HU Hao-quan, WANG Ya-tao. Preparation of mesoporous ZSM-5 zeolite and its application in 2-methylnaphthalene methylation reaction[J]. Acta Pet Sin (Pet Process Sec),2014,30(2):204−204. [8] 吴伟, 吴维果, 李凌飞, 杨巍, 武光. 氟硅酸铵改性的HZSM-12分子筛催化合成2,6–二甲基萘[J]. 石油学报(石油加工),2010,26(2):189−194.WU W, LI L, YANG W, WU G. Shape-selective synthesis of 2,6-dimethylnaphthalene over (NH4)2SiF6-modified HZSM-12 Zeolite[J]. Acta Pet Sin (Pet Process Sect),2010,26(2):189−194. [9] 王潇潇, 刘振民, 温健, 张伟, 赵亮富. 水蒸气处理改性的SAPO-11分子筛催化合成2,6-二甲基萘的研究[J]. 分子催化,2015,29(4):331−338.WANG Xiao-xiao, LIU Zhen-min, WEN Jian, ZHANG Wei, ZHAO Liang-fu. Study on catalytic synthesis of 2,6-dimethylnaphthalene over SAPO-11 zeolites modified by steam[J]. J Mol Catal (China),2015,29(4):331−338. [10] FATHIH G, SHER F, KARADUMAN A. Catalytic performance of Cu- and Zr-modified beta zeolite catalysts in the methylation of 2-methylnaphthalene[J]. Pet Sci,2018,16:161−172. [11] 王亚涛, 张新异, 房承宣, 靳立军, 郭学华, 胡浩权. 中空ZSM-5分子筛的制备及其在2-甲基萘烷基化合成2,6-二甲基萘中的应用[J]. 石油化工,2012,41(12):1351−1356. doi: 10.3969/j.issn.1000-8144.2012.12.003WANG Ya-tao, ZHANG Xin-yi, FANG Chen-xuan, JIN Li-jun, HU Hao-quan. Preparation of hollow ZSM-5 and its application in methylation of 2-methylnaphthalene to 2,6-dimethylnaphthalene[J]. Chin Petrochem Technol,2012,41(12):1351−1356. doi: 10.3969/j.issn.1000-8144.2012.12.003 [12] LI H J, GONG Q, LIAN H, DING L F, HU Z H, ZHU Z R. Transalkylation of C10 aromatics with 2-methylnaphthalene for 2,6-dimethylnaphthalene synthesis: High-efficiently shape-selective & synergistic catalysis over a multifunctional SiO2 -Mo-HBeta catalyst[J]. J Catal,2019,378:144−152. doi: 10.1016/j.jcat.2019.08.030 [13] LI H J, GONG Q, LIAN H, HU Z H, ZHU Z R. New process for 2,6-dimethylnaphthalene synthesis by using C10 aromatics as solvent and transmethylation-agentia: High-efficiency and peculiar subarea-catalysis over shape-selective ZSM-5/Beta[J]. Ind Eng Chem Res,2019,58:12593−12601. doi: 10.1021/acs.iecr.9b01596 [14] FERINO I, MONACI R, ROMBI E. Microcalorimetric investigation of mordenite and Y zeolites for 1-methylnaphthalene isomerisation[J]. J Chem Soc, Faraday Trans,1998,94(17):2647−2652. doi: 10.1039/a803931c [15] 栗同林, 刘希尧, 王祥生. β沸石对甲基萘歧化反应的择形性[J]. 催化学报,1997,18(3):48−51.LI Tong-lin, LIU Xi-yao, WANG Xiang-sheng. Shape selectivity for the disproportionation of methylnaphthalene over zeolite Hβ[J]. Chin J Catal,1997,18(3):48−51. [16] 董伟峰, 赵忠奎, 乔卫红, 王桂茹, 李宗石. Hβ沸石催化剂催化α-甲基萘的异构化反应[J]. 石油化工,2004,33(9):820−823. doi: 10.3321/j.issn:1000-8144.2004.09.005DONG Wei-feng, ZHAO Zhong-kui, QIAO Wei-hong, WANG Gui-ru, LI Zong-shi. Isomerization of α-methylnaphthalene on Hβ zeolite catalysts[J]. Chin Petrochem Technol,2004,33(9):820−823. doi: 10.3321/j.issn:1000-8144.2004.09.005 [17] 孙昊. 1-甲基萘定向转化反应的HBEA分子筛可控修饰及机理研究[D]. 南京, 南京师范大学, 2017.SUN Hao, Study on the controllable modification and mechanism of HBEA molecular sieve for the directed conversion of 1-Methylnaphthalene[D]. Nanjing: Nanjing Normal University, 2017. [18] JIN L J, FANG Y M, HU H Q. Selective synthesis of 2,6-dimethylnaphthalene by methylation of 2-methylnaphtha[J]. Catal Commun,2006,7:255−259. doi: 10.1016/j.catcom.2005.11.012 [19] ROTH W J, ČEJKI J, MILLINI R, MONTANARI E, GIL B, KUBU M. Swelling and interlayer chemistry of layered MWW zeolites MCM-22 and MCM-56 with high Al content[J]. Chem Mater,2015,27(13):4620−4629. doi: 10.1021/acs.chemmater.5b01030 [20] OSTROUMOVA V A, MAKSIMOV A L. MWW-Type zeolites: MCM-22, MCM-36, MCM-49, and MCM-56 (A Review)[J]. Pet Chem,2019,59(8):788−801. doi: 10.1134/S0965544119080140 [21] 李峰, 王红艳, 陈树伟, 李文林, 崔杏雨, 李瑞丰. 不同结构Zr-Beta分子筛制备及MPV反应性能研究[J]. 应用化工,2020,49(4):867−871. doi: 10.3969/j.issn.1671-3206.2020.04.017LI Feng, WANG Hong-yan, CHEN Shu-wei. Preparation of Zr-Beta molecular sieves with different structures and research on MPV reaction performance[J]. App Chem Ind,2020,49(4):867−871. doi: 10.3969/j.issn.1671-3206.2020.04.017 [22] 韩琪, 李海岩, 杨英, 刘百军. 超稳Y型分子筛的骨架硅铝比对甘油气相脱水反应的影响[J]. 化工进展,2019,38(6):2791−2795.HAN Qi, LI Hai-yan, YANG Ying, LIU Bai-jun. Influence of the SiO2/Al2O3 ratio on the performance of USY zeolites for the gas-phase dehydration of glycerol to acrolein[J]. Chem Ind Eng Prog (China),2019,38(6):2791−2795. [23] FENG R, YAN X, HU X, YAN Z, LIN J, LI Z, HOU K, ROOD M J. Surface dealumination of micro-sized ZSM-5 for improving propylene selectivity and catalyst lifetime in methanol to propylene (MTP) reaction[J]. Catal Commun,2018,109:1−5. doi: 10.1016/j.catcom.2018.02.005 [24] 张钰, 吴淑杰, 杨胥微, 贾明君, 王丽霞, 张祚望, 吴通好, 孙家锺. 脱铝MCM-49分子筛的结构、酸性及苯与丙烯液相烷基化催化性能研究[J]. 高等学校化学学报,2007,28(7):1319−1324. doi: 10.3321/j.issn:0251-0790.2007.07.021ZHANG Yu, WU Shu-jie, YANG Xu-wei, JIA Ming-jun, WANG Li-xia, ZHANG Zuo-wang, WU Tong-hao, SUN Jia-zhong. Structure, acid properties and catalysis performance of dealuminated MCM-49 zeolites for the alkylation of benzene with propylene[J]. Chem J Chin Univ,2007,28(7):1319−1324. doi: 10.3321/j.issn:0251-0790.2007.07.021 [25] EMEIS C A. Determination of integrated molar extinction coefficients for infrared absorption bands of pyridine adsorbed on solid acid catalysts[J]. J Catal,1993,141:347−354. doi: 10.1006/jcat.1993.1145 [26] ANUNZIATA O A, PIERELLA L B. Transalkylation of naphthalene with mesitylene over HZSM-11 zeolite[J]. Catal Lett,1997,44(3):259−263. [27] MENG F, SHANG Q, HUA D, CHEN L, SUN L, YANG S, XIE X, ZHAO B, ZHANG X. Naphthalene and alkyl naphthalene from catalytic fast pyrolysis of biomass over ZSM-5 aggregates[J]. J Biobased Mater Bioenergy,2020,14:195−202. doi: 10.1166/jbmb.2020.1943 [28] ALMULLA F M, ALI S A, ALDOSSARY M R, ALNAIMI E I, JUMAH A B, GARFORTH A A. Transalkylation of 1,2,4-trimethylbenzene with toluene over large pore zeolites: Role of pore structure and acidity[J]. Appl Catal A: Gen,2020,608:117886−117897. doi: 10.1016/j.apcata.2020.117886 [29] COLÓN G, FERINO I, ROMBI E, MAGNOUX P, GUISNET M. Evidence of transalkylation during liquid-phase isopropylation of naphthalene[J]. React Kinet Catal Lett,1998,63(1):3−8. doi: 10.1007/BF02475422 [30] TAKEUCHI G, SHIMOURA Y, HARA T. Selective transalkylation of naphthalene and ethylnaphthalene over solid acid catalysts[J]. Catal Lett,1996,41:195−197. doi: 10.1007/BF00811490 -




 下载:
下载: