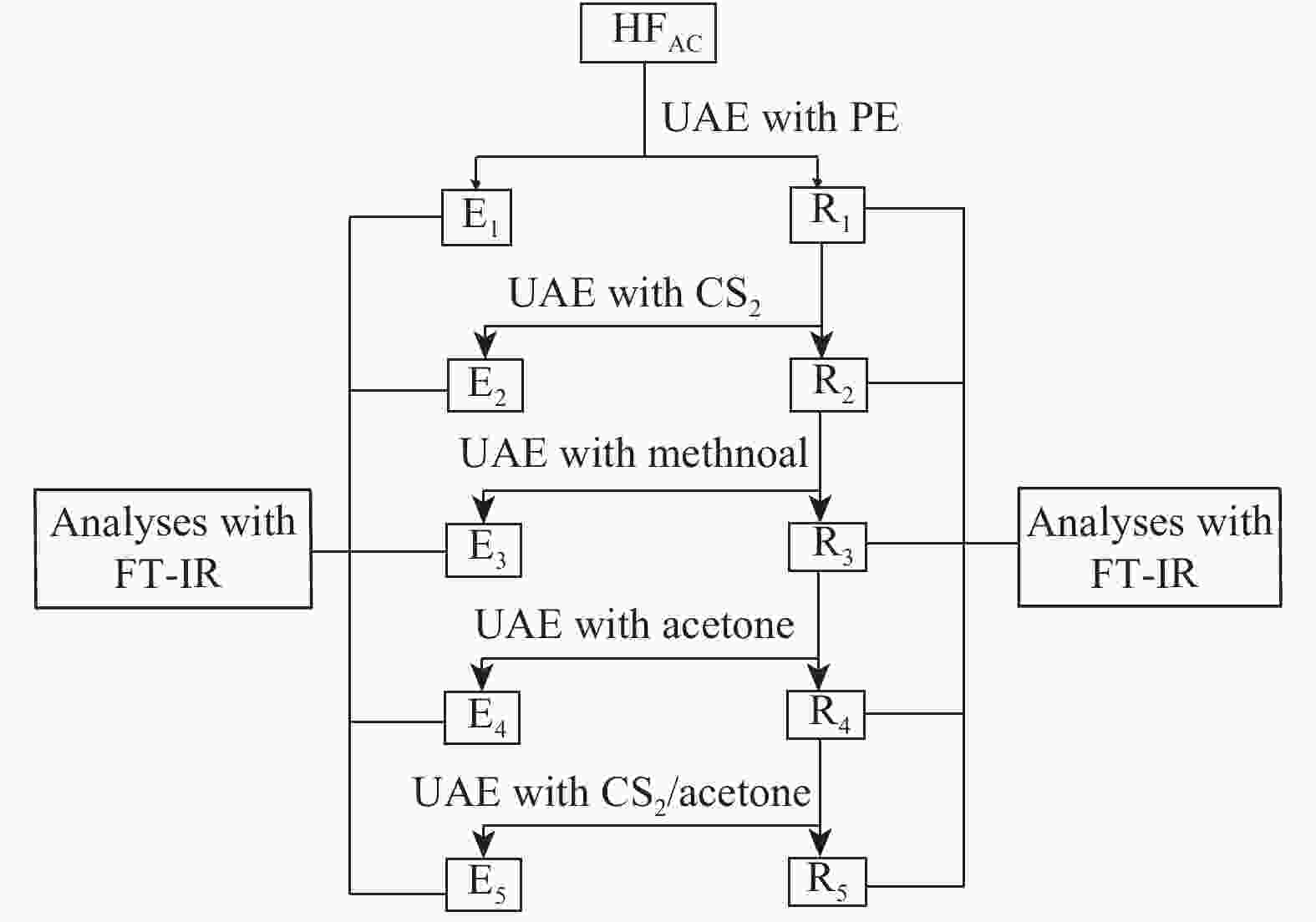
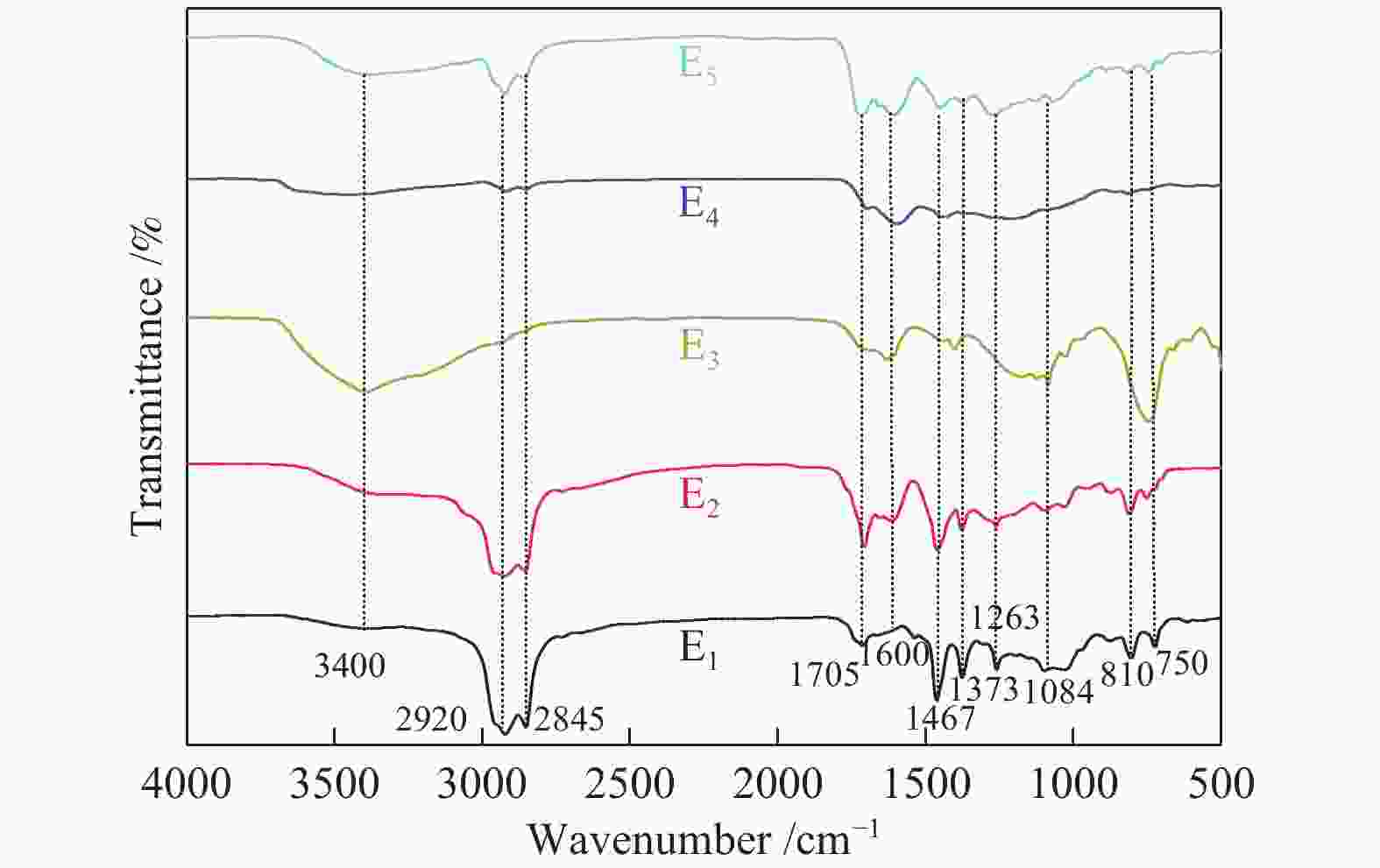
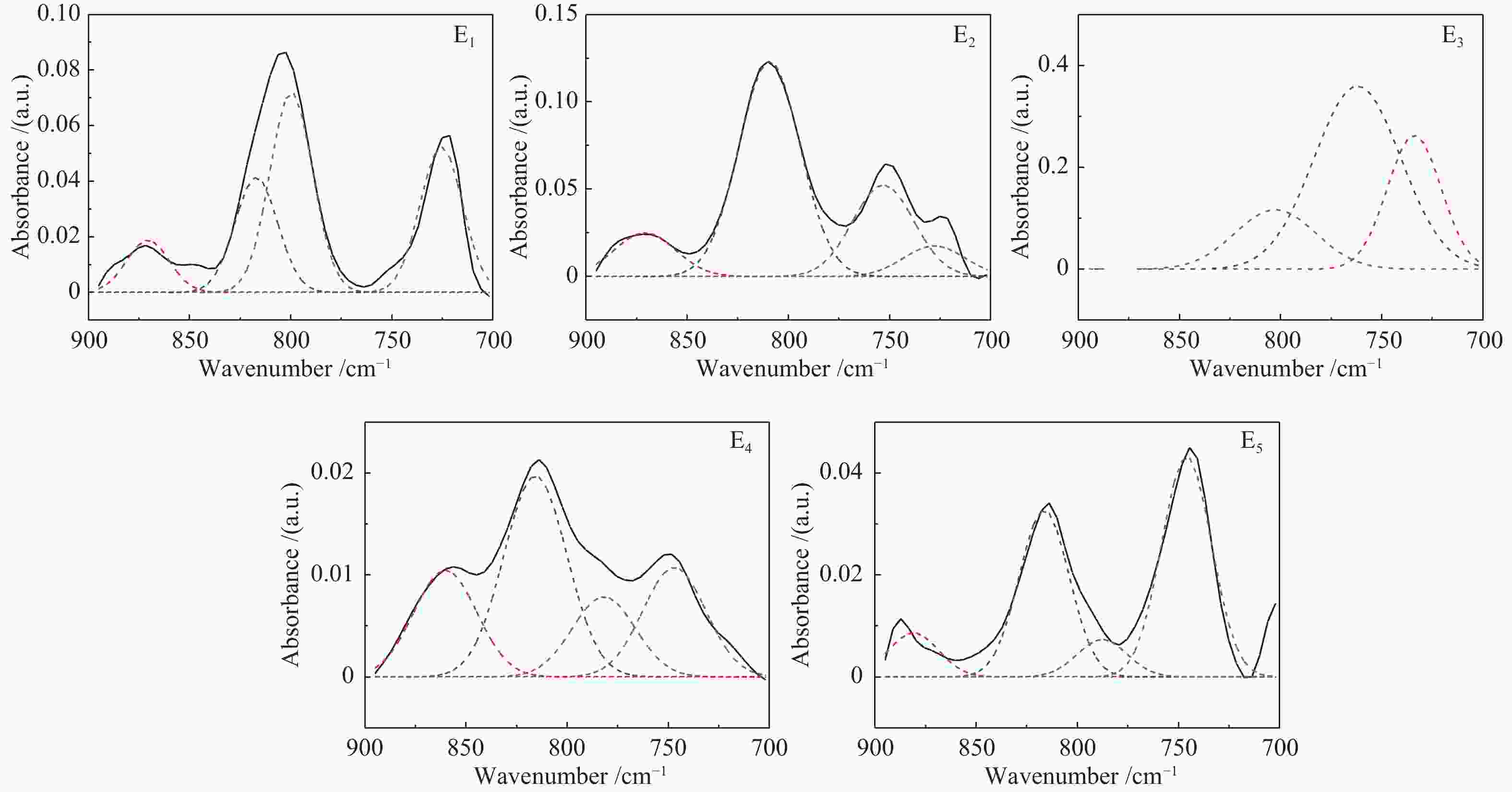
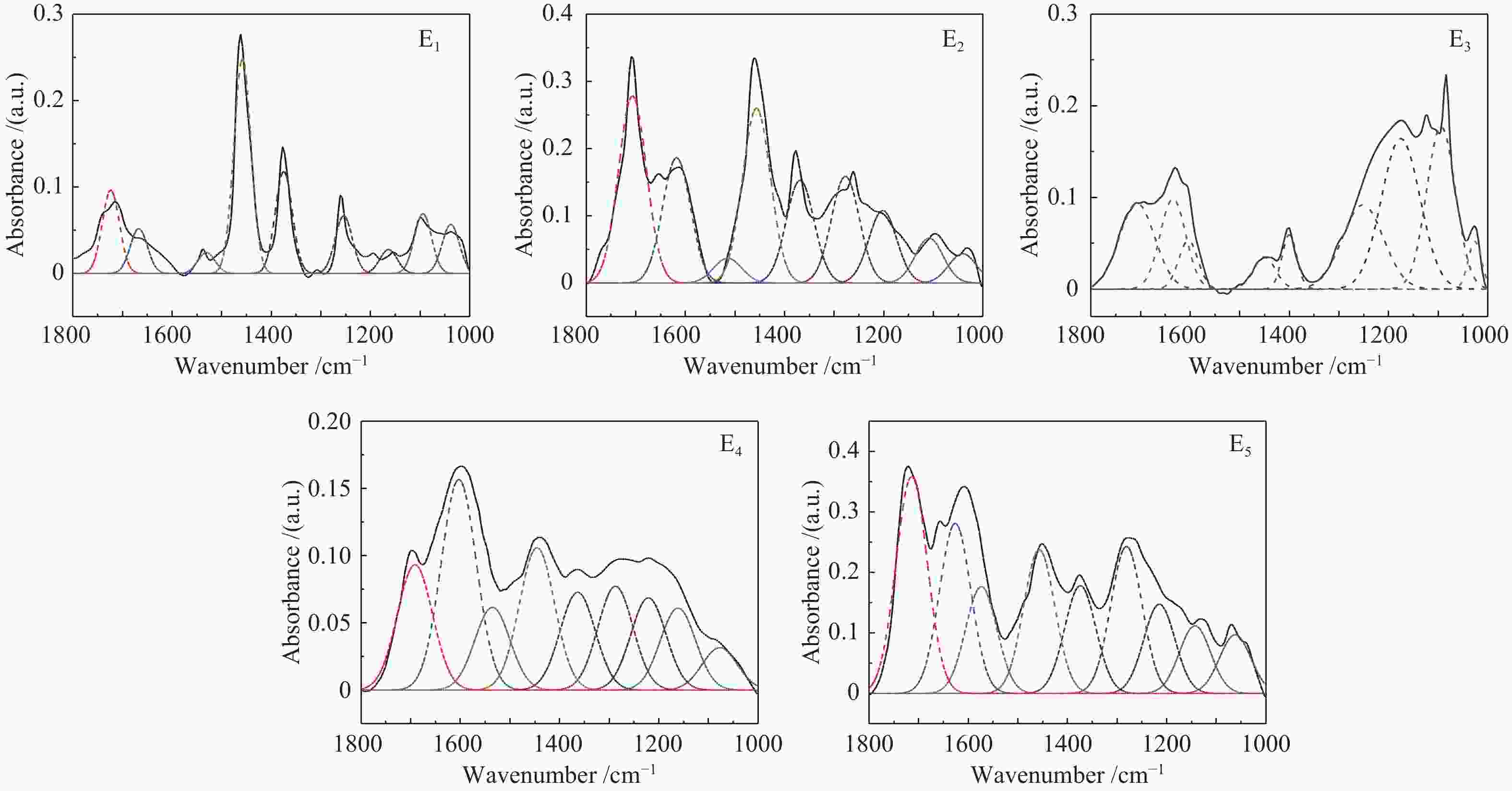
Functional groups of sequential extracts and corresponding residues from Hefeng sub-bituminous coal based on FT-IR analysis
-
摘要: 以酸洗和丰次烟煤为研究对象,依次采用石油醚、二硫化碳、甲醇、丙酮和丙酮/二硫化碳对煤样进行超声五级萃取,每级萃取物和萃余物分别记为Ei和Ri(i=1、2、3、4、5)。通过对Ei及Ri进行FT-IR表征,借助分段分峰拟合手段分析了萃取产物的分子结构。结果表明,五级萃取物的羟基氢键结构均以自缔合羟基氢键为主;在脂肪类物质中,除第三级萃取物E3以脂肪族−CH3和不对称的−CH2伸缩振动为主之外,其余几级萃取物均以对称和不对称的−CH2伸缩振动为主;E1主要以脂肪链末端−CH3的对称弯曲振动和−CH3、−CH2的不对称变形振动为主,说明石油醚主要将煤样中易于解离的化学键断裂;五级萃取中CS2溶解了更大比例的含脂肪侧链的芳香结构。各级萃余物所含官能团种类相同,说明煤样主体结构并未因逐级萃取而发生改变。萃取对残渣中芳香结构和羟基氢键结构影响较大。其中,酸洗煤样以苯环二取代为主,而萃取后以苯环四取代为主。萃取前,酸洗煤样中羟基氢键结构以羟基醚氢键为主,而萃取后转变成以自缔合羟基氢键为主。另外,分级萃取对含氧官能团和脂肪类官能团影响较小。对比红外结构参数发现,E1、E3及R5有较高的芳环缩合度,E4的脂肪直链较长、支链较少。Abstract: Petroleum ether, carbon disulfide, methanol, acetone, and acetone/carbon disulfide were selected as solvents for ultrasonic-assisted extraction of acid-washed Hefeng sub-bituminous coal. The extract and residue were identified as Ei and Ri (i = 1, 2, 3, 4, 5) for each stage. By FT-IR characterization of Ei and Ri, molecular structure of the extracted product was analyzed by means of segmented peak fitting. The results show that the hydroxy hydrogen bond in the fifth-order extract is dominated by self-associating hydroxy hydrogen bond; in aliphatic substances, only E3 was dominated by aliphatic −CH3 and asymmetric −CH2 stretching vibration, while the other extracts were dominated by symmetric and asymmetric −CH2 stretching vibration. E1 is dominated by symmetric bending vibration of aliphatic chain terminal −CH3 and asymmetric deformation vibration of −CH3 and −CH2, indicating that petroleum ether mainly break the easily dissociated chemical bonds in coal samples; CS2 dissolve a higher proportion of aromatic structure containing aliphatic side chains. The functional groups contained in the five residues are the same, indicating that the main structure of the coal sample is not changed due to the stepwise extraction. Extraction has an obvious influence on aromatic structure and hydroxy hydrogen bond in the residue. Aromatic structures change from di-substituted benzene dominant to tetra-substituted benzene dominant. Before extraction, the hydroxyl hydrogen bond in the acid-washed coal sample was dominated by hydroxyl ether hydrogen bonds, and after extraction, it was transformed into self-associated hydroxyl ones. In addition, sequential extraction has little effect on oxygen-containing functional groups and aliphatic functional groups. By comparing structural parameters, it is found that E1, E3, and R5 has a higher degree of aromatic condensation, and E4 has a longer straight-chain and less branched chain.
-
Key words:
- coal structure /
- FT-IR spectroscopy /
- functional groups /
- extract residue
-
表 1 煤样的工业分析和元素分析
Table 1 Proximate and ultimate analyses of the coal samples
Sample Proximate analysis w/% Ultimate analysis wdaf/% H/C Mad Ad Vdaf FCdaf C H N S O* HF 5.88 21.18 42.81 57.19 74.91 5.65 1.50 0.37 17.57 0.91 HFAC 1.52 1.84 44.37 55.63 73.05 5.33 1.50 0.38 19.74 0.88 表 2 五种样品红外光谱分峰拟合各吸收峰面积比例
Table 2 Parameters of FT-IR spectrum for five samples by curve-fitting
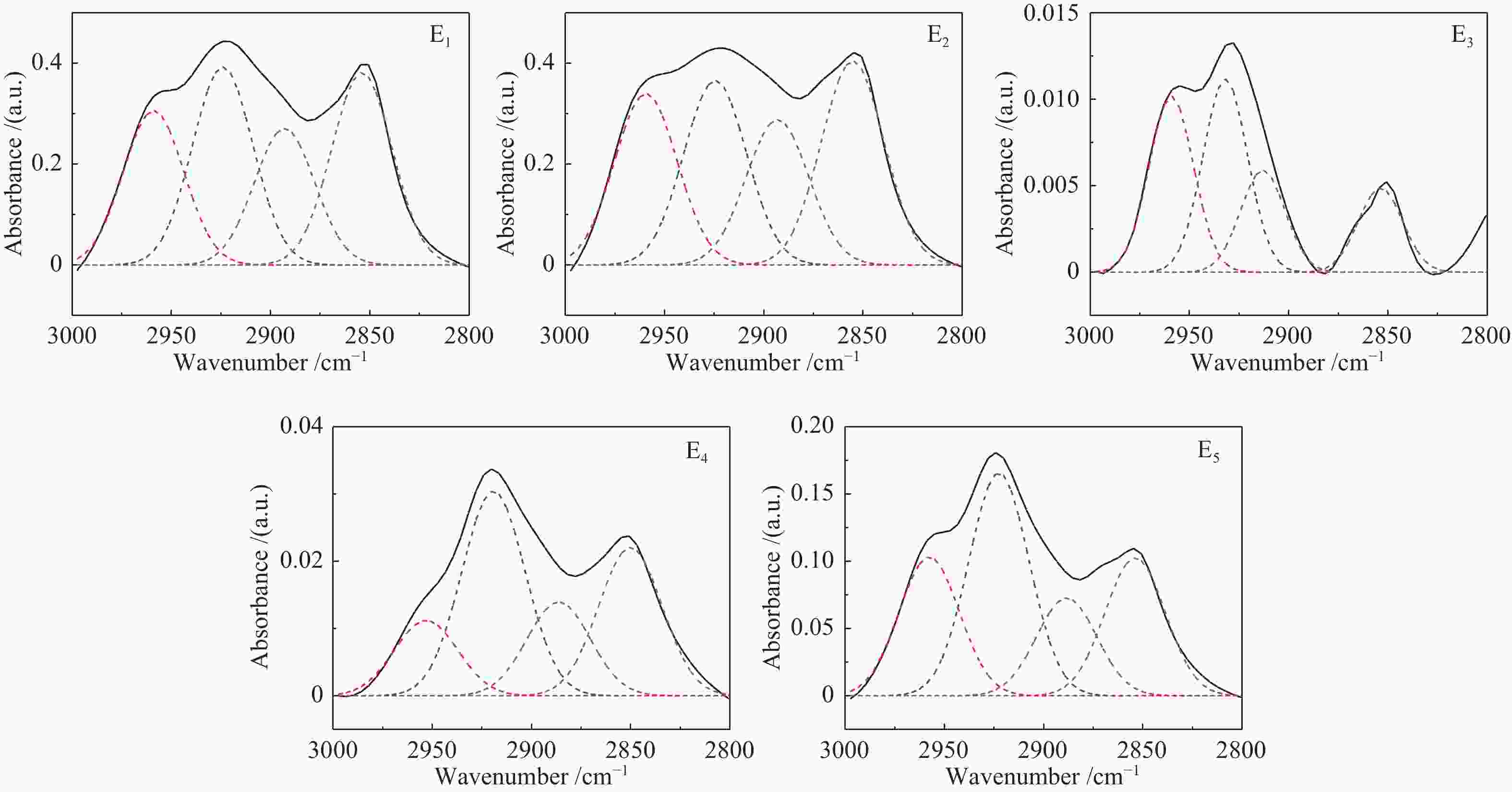
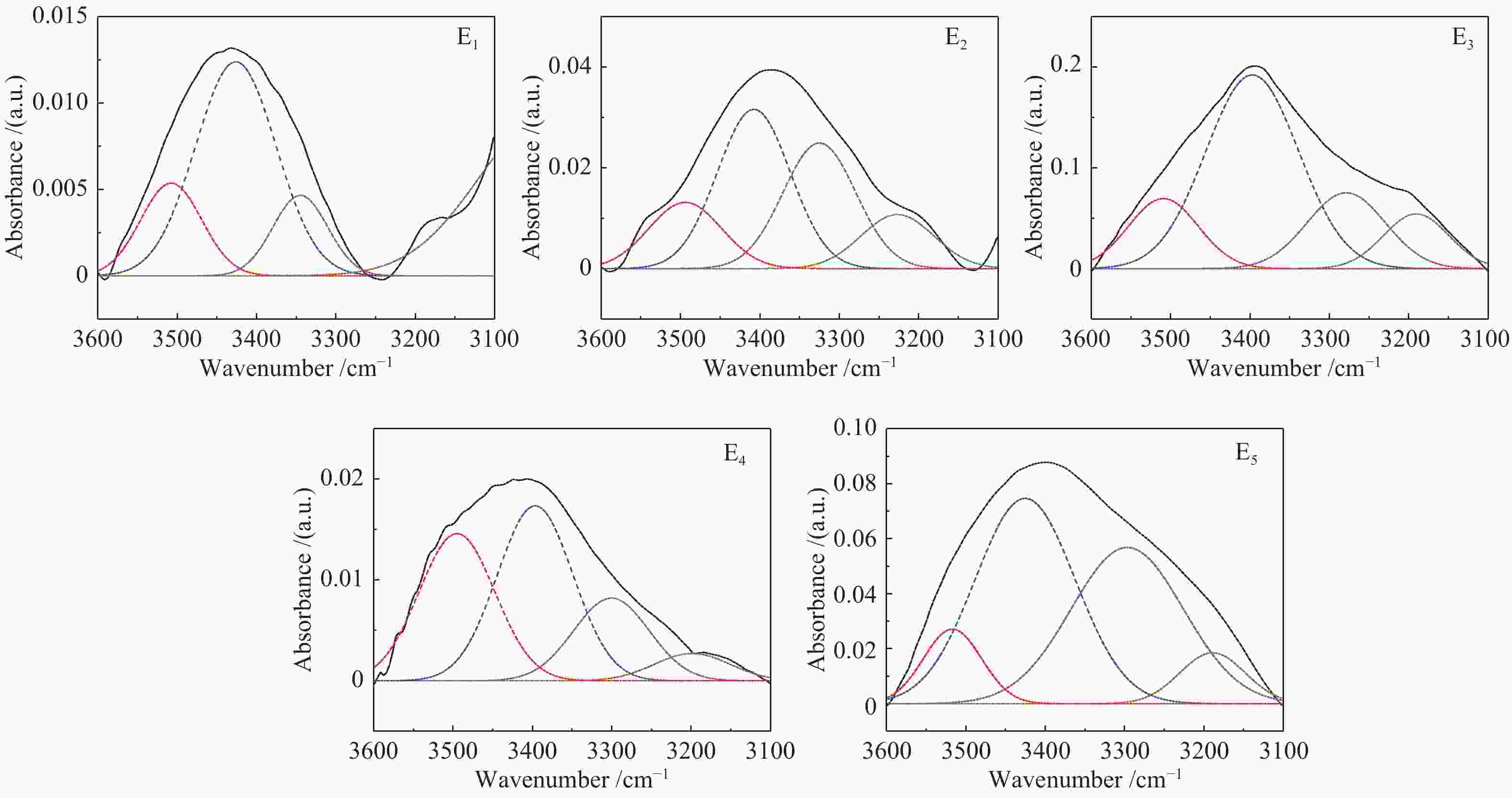
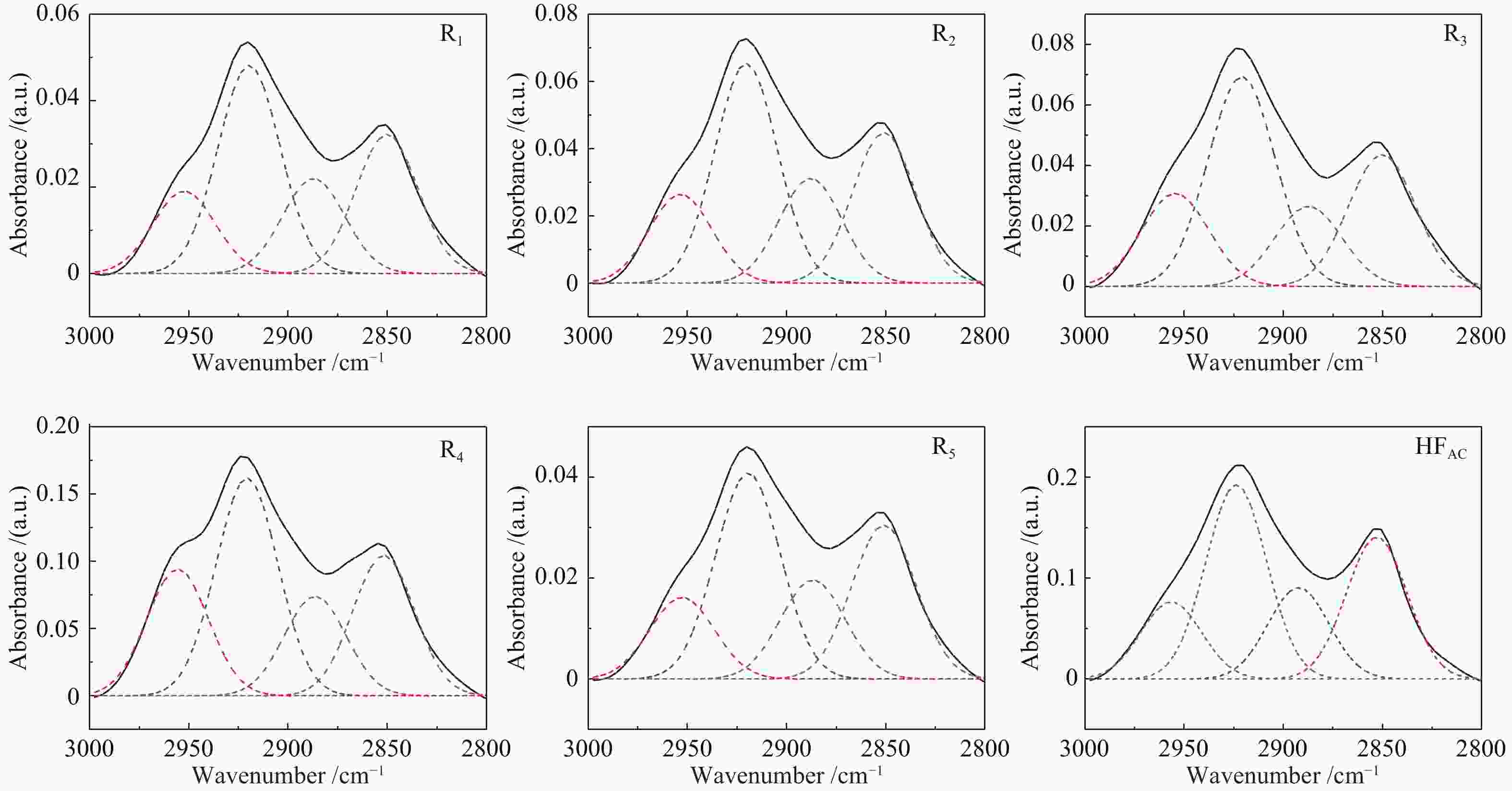
Band position/cm−1 Functional groups Area percentage/% E1 E2 E3 E4 E5 900−860 five adjacent H deformation 10.13 10.95 0 21.24 8.28 860−810 four adjacent H deformations 22.54 57.05 17.21 40.56 36.04 810−750 three adjacent H deformations 39.22 24.24 56.95 16.18 8.10 750−720 two adjacent H deformations 28.11 7.77 25.84 22.03 47.59 1710 carboxylic acids C=O 11.48 21.61 13.59 12.78 19.54 1650 conjugated C=O 4.73 14.48 10.34 21.51 15.39 1500 aromatic C=C 1.26 2.86 4.21 8.46 9.67 1436 asymmetric −CH3, −CH2 36.56 20.20 3.18 14.56 13.01 1371 CH3-Ar, R 16.80 11.88 3.55 10.00 9.75 1269 symmetric deformation −CH3 8.95 12.32 15.45 10.61 13.30 1165 C−O phenols 2.54 8.37 25.91 9.43 8.07 1110 grease C−O 9.53 5.19 20.94 8.39 6.12 1035 alkyl ethers 7.84 3.09 2.84 4.26 5.16 2950 aliphatic −CH3 22.56 24.16 31.67 14.42 23.15 2920 asymmetric aliphatic −CH2 29.12 26.21 34.80 39.21 37.34 2890 aliphatic −CH 20.06 20.65 18.36 17.99 16.40 2850 symmetric aliphatic −CH2 28.27 28.98 15.17 28.39 23.10 3600−3500 OH−π 17.43 16.20 14.86 33.78 9.41 3500−3350 self-associated OH 53.67 39.40 55.80 40.73 44.94 3350−3260 OH-ether O 13.72 31.07 18.41 19.25 38.44 3260−3170 cyclic OH 15.19 13.33 10.93 6.24 7.21 表 3 五级萃取物的结构参数
Table 3 Structural parameters of five samples derived from the FT-IR spectrum
Sample DOC A(CH2)/A(CH3) E1 16.19 1.28 E2 3.24 1.08 E3 13.03 1.06 E4 0.90 2.71 E5 0.34 1.61 表 4 六种样品红外光谱分峰拟合各吸收峰面积比例
Table 4 Parameters of FT-IR spectrum for six samples by curve-fitting
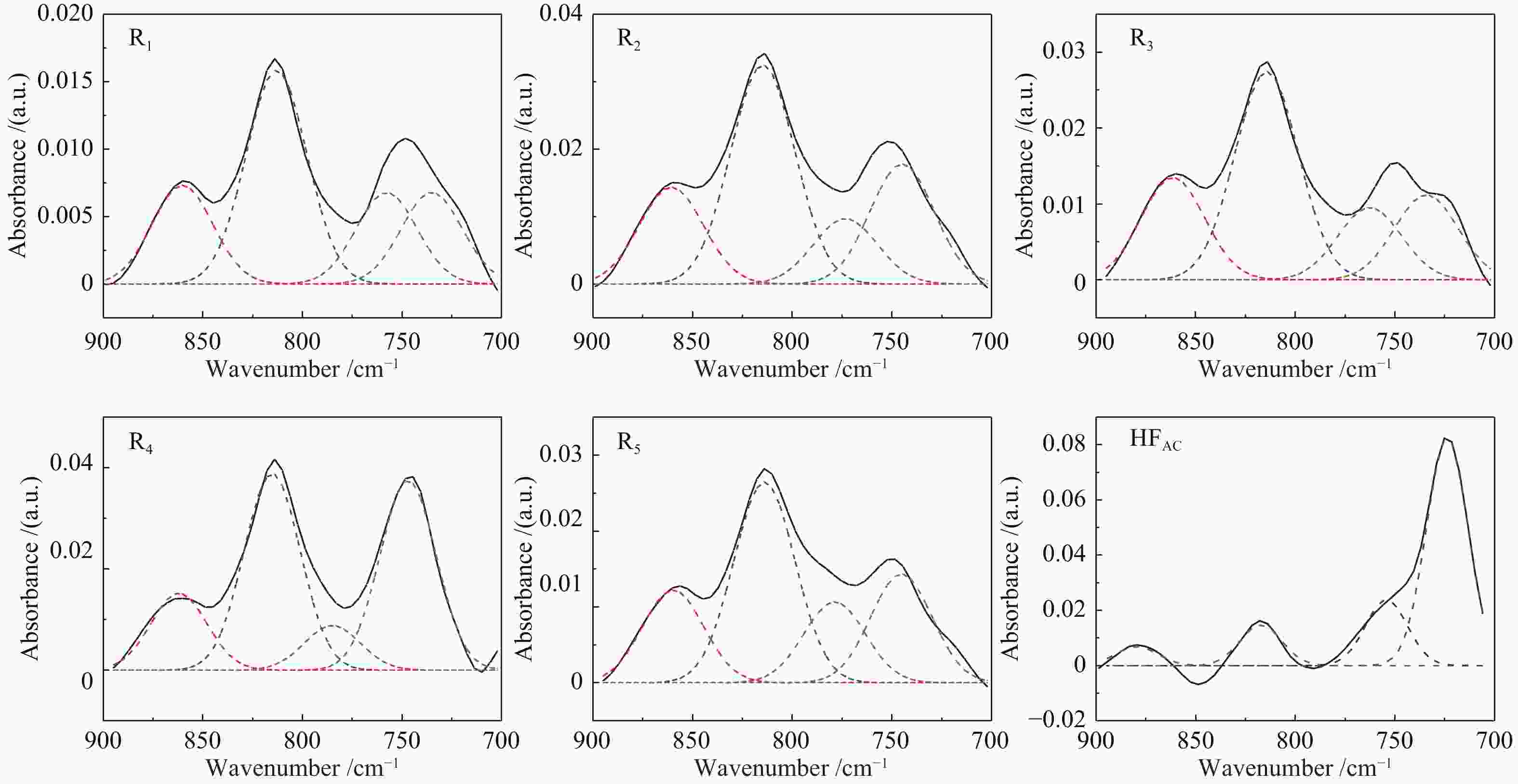
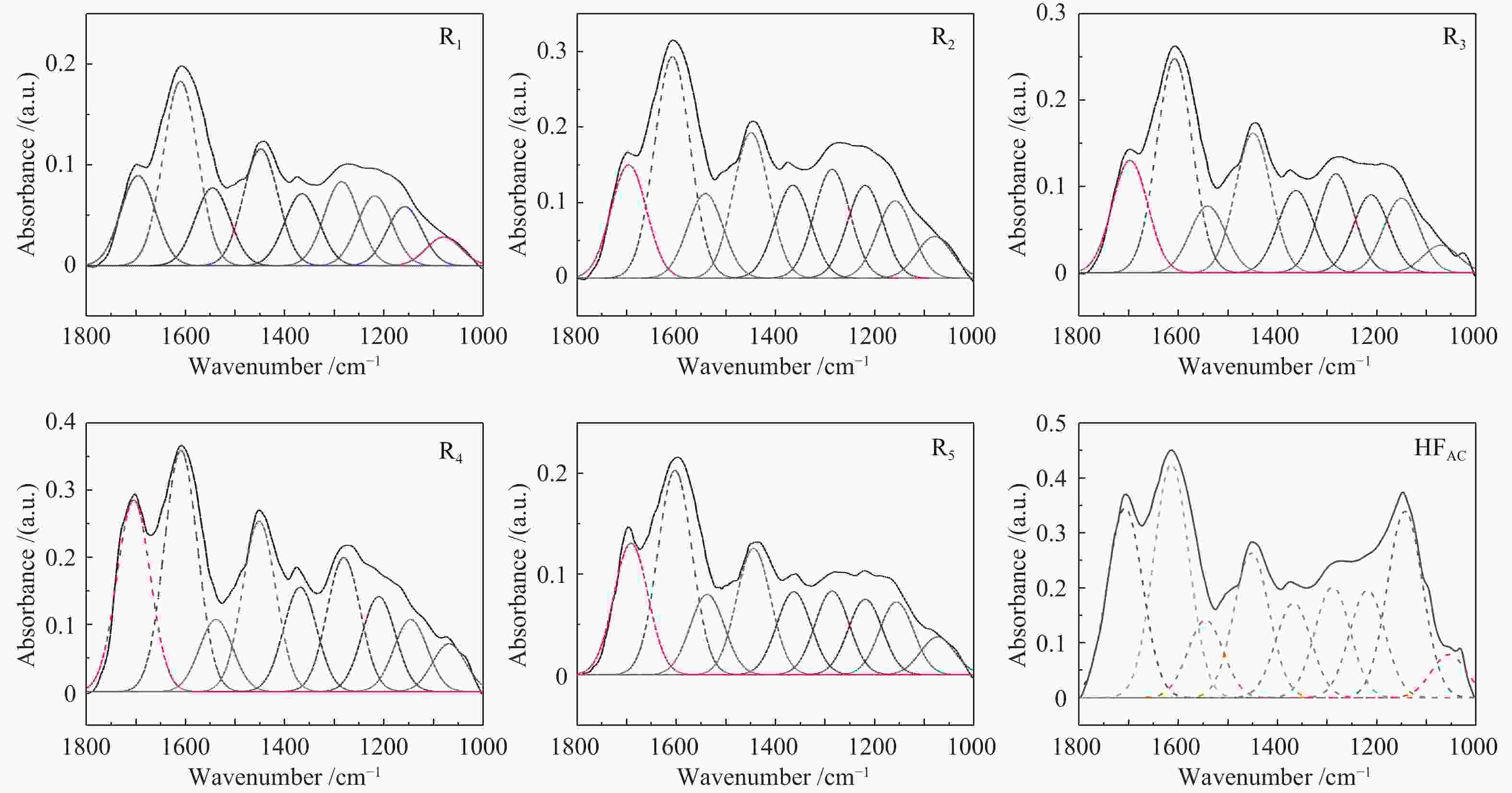
Band position/cm−1 Functional groups Area percentage/% HFAC R1 R2 R3 R4 R5 900−860 five adjacent H deformation 5.41 19.88 19.19 21.66 15.01 19.02 860−810 four adjacent H deformations 11.52 43.34 43.82 44.84 38.78 41.66 810−750 three adjacent H deformations 18.73 18.51 13.08 15.62 8.81 16.81 750−720 two adjacent H deformations 64.34 18.27 23.91 17.89 37.4 22.51 1710 carboxylic acids C=O 15.94 11.49 11.57 12.56 16.96 14.68 1650 conjugated C=O 19.67 23.57 22.66 23.98 21.41 22.85 1500 aromatic C=C 6.54 9.96 8.65 7.48 6.40 9.00 1436 asymmetric −CH3, −CH2 12.18 14.95 14.89 15.64 15.12 14.14 1371 CH3-Ar, R 7.98 9.2 9.51 9.21 9.26 9.27 1269 symmetric deformation −CH3 9.33 10.75 11.14 11.07 11.90 9.35 1165 C−O phenols 9.05 8.93 9.48 8.71 8.43 8.42 1110 grease C−O 15.69 7.55 7.91 8.36 6.39 8.14 1035 alkyl ethers 3.62 3.61 4.19 2.99 4.13 4.13 2950 aliphatic −CH3 19.06 15.68 15.71 17.99 21.61 15.06 2920 asymmetric aliphatic −CH2 35.83 39.69 39.00 40.82 37.31 38.15 2890 aliphatic −CH 24.95 18.11 18.60 15.59 17.04 18.34 2850 symmetric aliphatic −CH2 20.16 26.51 26.69 25.61 24.04 28.44 3600−3500 OH−π 30.53 29.18 26.76 26.23 7.42 31.85 3500−3350 self-associated OH 36.26 40.13 34.76 31.00 41.18 38.01 3350−3260 OH-ether O 20.06 18.75 22.95 24.68 42.82 18.08 3260−3170 cyclic OH 13.15 11.94 15.53 18.09 8.58 12.06 表 5 酸洗煤及五级萃余物的结构参数
Table 5 Structural parameters of six samples derived from the FT-IR spectrum
Sample I DOC A(CH2)/A(CH3) $ 'C' $ HFAC 0.17 0.09 2.53 0.84 R1 0.30 0.08 2.54 0.81 R2 0.46 0.11 2.49 0.82 R3 0.35 0.11 2.28 0.84 R4 0.19 0.10 1.72 0.86 R5 0.60 0.14 2.55 0.83 -
[1] KONG J, WEI X Y, LI Z K, YAN H L, ZONG Z M. Identification of organonitrogen and organooxygen compounds in the extraction residue from Buliangou subbituminous coal by FTICRMS[J]. Fuel,2015,171:151−8. [2] ZHANG Z Q, KANG Q N, WEI S, YUN T, YAN G C, YAN K F. Large scale molecular model construction of Xishan bituminous coal[J]. Energy Fuels,2017,31(2):1310−1317. doi: 10.1021/acs.energyfuels.6b02623 [3] MATHEWS J P, CHAFFEE A L. The molecular representations of coal-A review[J]. Fuel,2012,96:1−14. doi: 10.1016/j.fuel.2011.11.025 [4] RATHSACK P, KROLL M M, OTTO M. Analysis of high molecular compounds in pyrolysis liquids from a German brown coal by FT-ICR-MS[J]. Fuel,2014,115:461−468. doi: 10.1016/j.fuel.2013.07.064 [5] HE X Q, MO W L, MA Y Y, MA F Y, WANG Q, FAN X, WEI X Y. Effect of swelling treatment by organic solvent on the structure and pyrolysis performance of the direct coal liquefaction residue[J]. Energy Fuels,2020,34(7):8685−8696. doi: 10.1021/acs.energyfuels.0c01031 [6] WANG S Q, TANG Y G, SCHOBERT H H, GUO Y N, GAO W C, LU X K. FTIR and simultaneous TG/MS/FTIR study of Late Permian coals from Southern China[J]. J Anal Appl Pyrolysis,2013,100:75−80. doi: 10.1016/j.jaap.2012.11.021 [7] LI F J, WEI X Y, FAN M H, ZONG Z M. Separation and structural characterization of the value-added chemicals from mild degradation of lignites: A review[J]. Appl Energy,2016,170:415−436. doi: 10.1016/j.apenergy.2016.02.131 [8] LU H Y, WEI X Y, YU R, PENG Y L. Sequential thermal dissolution of Huolinguole lignite in methanol and ethanol[J]. Energy Fuels,2011,25(6):2741−2745. doi: 10.1021/ef101734f [9] ZHAO X Y, ZONG Z M, CAO J P, MA Y M, HAN L, LIU G F, ZHAO W, LI W Y, XIE K C, BAI X F, WEI X Y. Difference in chemical composition of carbon disulfide-extractable fraction between vitrinite and inertinite from Shenfu-Dongsheng and Pingshuo coals[J]. Fuel,2008,87(5):565−575. [10] ZHOU G L, WU J J, MIAO Z Y, HU X L, LI X, SHI X, CAI Z D. Effects of process parameters on pore structure of semi-coke prepared by solid heat carrier with dry distillation[J]. Int J Min Sci Technol,2013,23(3):423−428. doi: 10.1016/j.ijmst.2013.05.003 [11] OENAL Y, CEYLAN K. Low temperature extractability and solvent swelling of Turkish lignites[J]. Fuel Process Technol,1997,53(1):81−97. [12] DING M, ZHAO Y P, DOU Y Q, WEI X Y, FAN XING, CAO J P, WANG Y L, ZONG Z M. Sequential extraction and thermal dissolution of Shengli lignite[J]. Fuel Process Technol,2015,135:20−24. doi: 10.1016/j.fuproc.2014.09.031 [13] LI P, ZONG Z M, LIU F J, WANG Y G, WEI X Y, FAN X, ZHAO Y P, ZHAO W. Sequential extraction and characterization of liquefaction residue from Shenmu-Fugu subbituminous coal[J]. Fuel Process Technol,2015,136:1−7. doi: 10.1016/j.fuproc.2014.04.013 [14] TAKANOHASHI T, XIAO F J, YOSHIDA T, SAITO I. Difference in extraction yields between CS2/NMP and NMP for upper freeport coal[J]. Energy Fuels,2003,17(1):255−256. doi: 10.1021/ef020141h [15] LIU F J, WEI X Y, GUI J, WANG Y G, LI P, ZONG Z M. Characterization of biomarkers and structural features of condensed aromatics in Xianfeng lignite[J]. Energy Fuels,2013,27:7369−7378. [16] 郭振兴, 高春楠, 张春明, 贾利国, 张跃弟, 孙昊, 江春, 秦志宏. 淮北煤两级分次萃取物的红外光谱分析[J]. 煤化工,2006,34(6):32−35. doi: 10.3969/j.issn.1005-9598.2006.06.010GUO Zhen-xing, GAO Chun-nan, ZHANG Chun-ming, JIA Li-guo, ZHANG Yue-di, SUN Hao, JIANG Chun, QIN Zhi-hong. FTIR analysis of extracts from Huaibei coal by fractional extraction[J]. Coal Chem Ind,2006,34(6):32−35. doi: 10.3969/j.issn.1005-9598.2006.06.010 [17] 江春, 孙昊, 张迪, 胡卫新, 秦志宏. FTIR法对准格尔煤两级分次萃取物的分析[J]. 煤炭学报,2007,32(1):85−89. doi: 10.3321/j.issn:0253-9993.2007.01.019JIANG Chun, SUN Hao, ZHANG Di, HU Wei-xin, QIN Zhi-hong. FTIR analysis of fractional extracts from Zhunge’ er bituminous coal[J]. J China Coal Soc,2007,32(1):85−89. doi: 10.3321/j.issn:0253-9993.2007.01.019 [18] 张硕, 张小东, 丁哲, 郝宗超, 赵家攀. 不同溶剂抽提下高阶煤的化学组成和结构变化[J]. 煤炭转化,2016,39(3):1−5. doi: 10.3969/j.issn.1004-4248.2016.03.001ZHANG Suo, ZHANG Xiao-dong, DING Zhe, HAO Zong-chao, ZHAO Jia-pan. Chemical composition and structure change of high rank coal extracted by different solvents[J]. Coal Convers,2016,39(3):1−5. doi: 10.3969/j.issn.1004-4248.2016.03.001 [19] 王晓华, 魏贤勇, 宗志敏. 溶剂分级萃取法研究平朔煤的化学组成特征[J]. 煤炭转化,2006,29(2):4−7. doi: 10.3969/j.issn.1004-4248.2006.02.002WANG Xiao-hua, WEI Xian-yong, ZONG Zhi-min. Study on chemical constituent characteristic of fractionated extraction from Pingshuo coal[J]. Coal Convers,2006,29(2):4−7. doi: 10.3969/j.issn.1004-4248.2006.02.002 [20] LI J, FENG J, LI W Y, CHANG H Z, XIE K C. Effect of hydrogen bond on coal extraction by in-situ vacuum FTIR[J]. Energy Sources, Part A,2009,31:1660−1665. doi: 10.1080/15567030903021970 [21] TAHMASEBI A, JIANG Y, YU J L, LI X C. Solvent extraction of Chinese lignite and chemical structure changes of the residue during H2O2 oxidation[J]. Fuel Process Technol,2015,129:213−221. doi: 10.1016/j.fuproc.2014.09.024 [22] 余坤坤, 张小东, 张硕, 杜志刚. 不同煤岩成分的焦煤萃取后FTIR特征研究[J]. 光谱学与光谱分析,2018,38(10):3077−3083.YU Kun-kun, ZHANG Xiao-dong, ZHANG Shuo, DU Zhi-gang. The FTIR characteristics of extracted coking coal in different macrolithotype[J]. Spectrosc Spectral Anal,2018,38(10):3077−3083. [23] WANG Y N, WEI X Y, LI Z K, YAN H L, WANG T M, ZHANG Y Y, ZONG Z M, MA F Y, LIU J M. Extraction and thermal dissolution of Piliqing subbituminous coal[J]. Fuel,2017,200:282−289. [24] 张祥成, 孟永彪, 马凤云, 刘景梅. 酸洗处理对新疆和丰煤结构的影响[J]. 河南科技大学学报(自然科学版),2019,40(6):89−94+10.ZHANG Xiang-cheng, MENG Yong-biao, MA Feng-yun, LIU Jing-mei. Effect of acid-washing treatment on structure of Xingjiang Hefeng coal[J]. J Henan Univ Sci Technol, Nat Sci,2019,40(6):89−94+10. [25] MA Y Y, MA F Y, MO W L, WANG Q. Five-stage sequential extraction of Hefeng coal and direct liquefaction performance of the extraction residue[J]. Fuel,2019,266:117039. [26] LIU L L, YUAN Y, KUMAR S, WANG Z H, HE Y, LV Y, LIU J Z, CEN K F. Catalytic effect of metal chlorides on coal pyrolysis and gasification part II. Effects of acid washing on coal characteristics[J]. Thermochim Acta,2018,666:41−50. doi: 10.1016/j.tca.2018.06.001 [27] QIN Z H, CHEN H, YAN Y J, LI C S, RONG L M, YANG X Q. FTIR quantitative analysis upon solubility of carbon disulfide/N-methyl-2-pyrrolidinone mixed solvent to coal petrographic constituents[J]. Fuel Process Technol,2015,133:14−19. doi: 10.1016/j.fuproc.2015.01.001 [28] ZHAO Y, QIU P H, CHEN G, PEI J T, SUN S Z, LIU L, LIU H P. Selective enrichment of chemical structure during first grinding of Zhundong coal and its effect on pyrolysis reactivity[J]. Fuel,2017,189:46−56. doi: 10.1016/j.fuel.2016.10.083 [29] HE X Q, LIU X F, NIE B S, SONG D Z. FTIR and Raman spectroscopy characterization of functional groups in various rank coals[J]. Fuel,2017,206:555−563. doi: 10.1016/j.fuel.2017.05.101 [30] WANG S Q, TANG Y G, SCHOBERT H H, GUO Y N, SU Y F. FTIR and 13C-NMR investigation of coal component of late Permian coals from southern China[J]. Energy Fuels,2011,25:5672−5677. doi: 10.1021/ef201196v [31] 程雪云, 李梅, 孙功成, 金权, 李佳佳, 徐荣声. 烟煤和褐煤分级萃取物的GC/MS分析[J]. 煤炭转化,2020,43(2):1−10.CHENG Xue-yun, LI Mei, SUN Gong-cheng, JIN Quan, LI Jia-jia, XU Rong-sheng. GC/MS analysis of bituminous coal and lignite extracts by different solvents[J]. Coal Convers,2020,43(2):1−10. [32] 杜姣姣, 赵云鹏, 田由甲, 张迪. 白音华褐煤可溶有机质的组成和结构特征[J]. 燃料化学学报,2017,45(1):9−14. doi: 10.3969/j.issn.0253-2409.2017.01.002DU Jiao-jiao, ZHAO Yun-peng, TIAN You-jia, ZHANG Di. Composition and structural characteristics of soluble organic species in Baiyinhua lignite[J]. J Fuel Chem Technol,2017,45(1):9−14. doi: 10.3969/j.issn.0253-2409.2017.01.002 -




 下载:
下载: