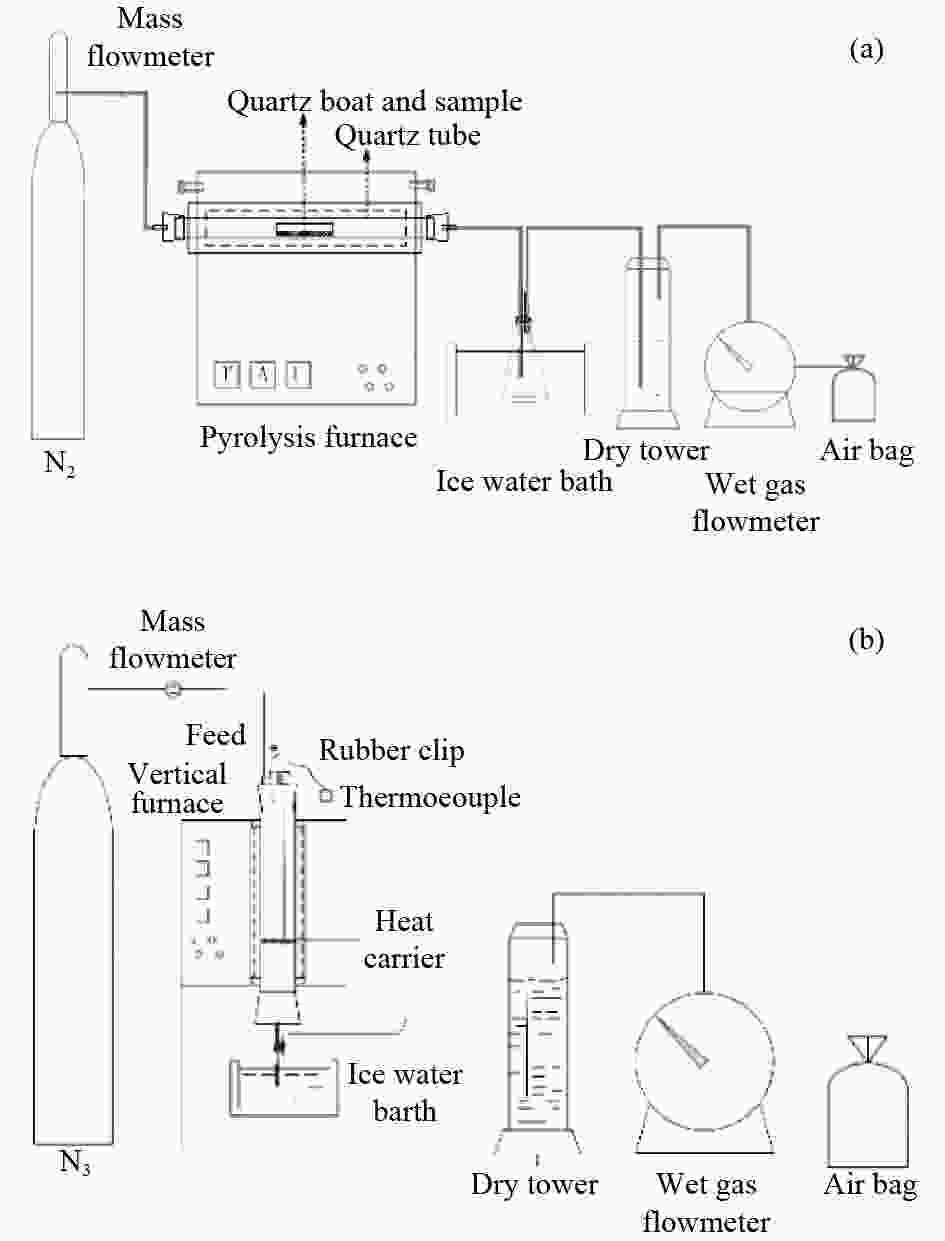
Volatility of cadmium during pyrolysis of typical low rank coals
-
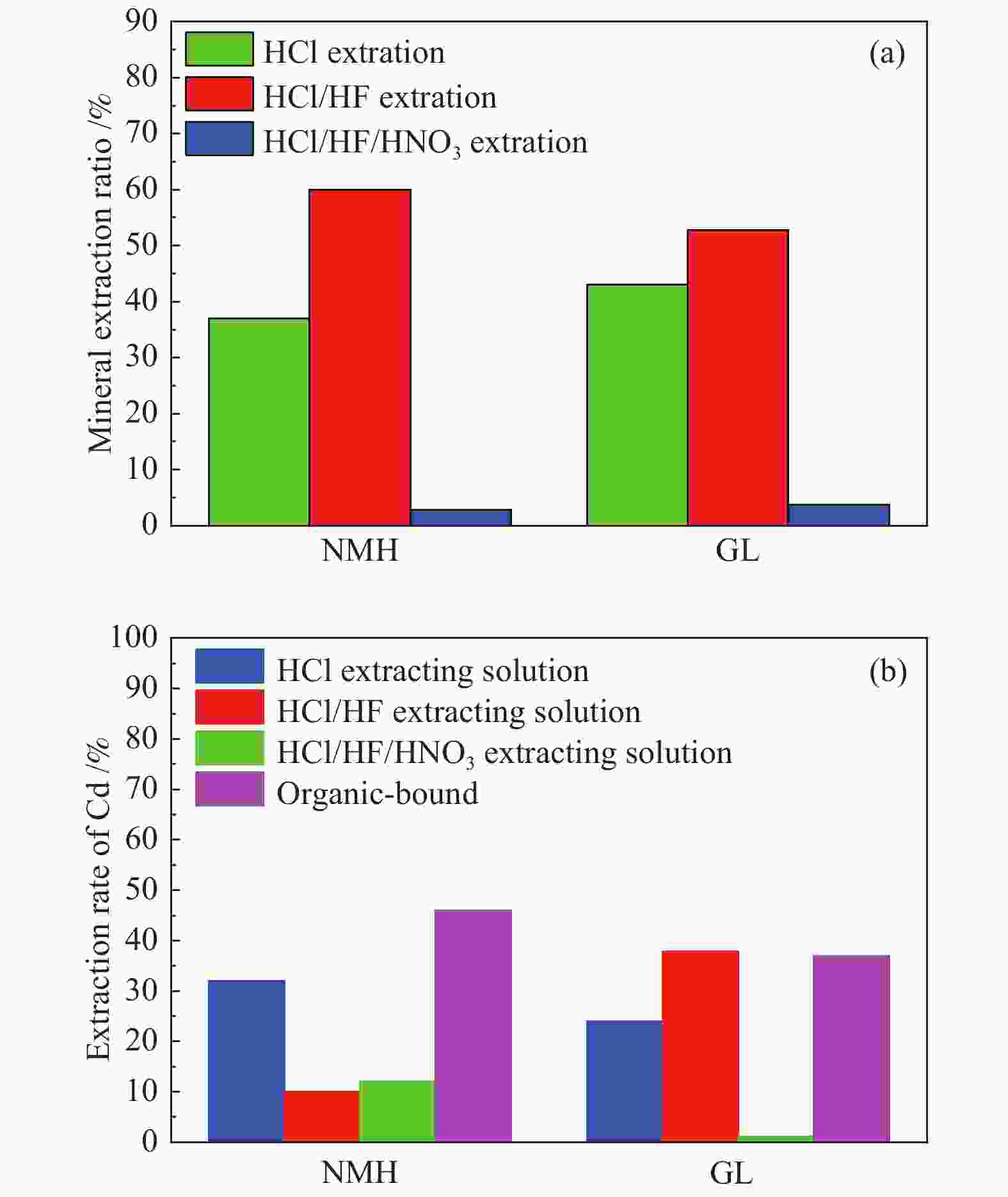
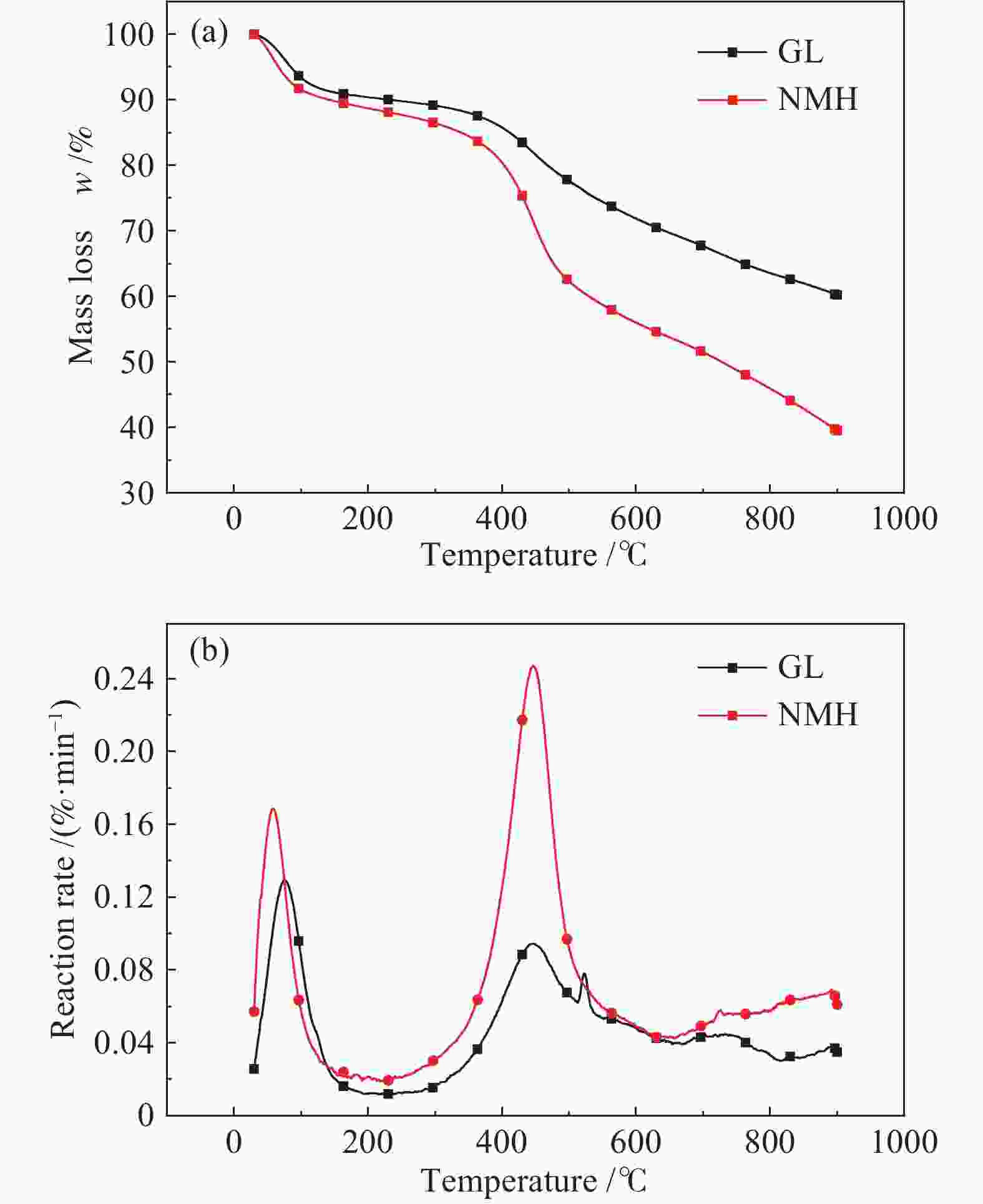
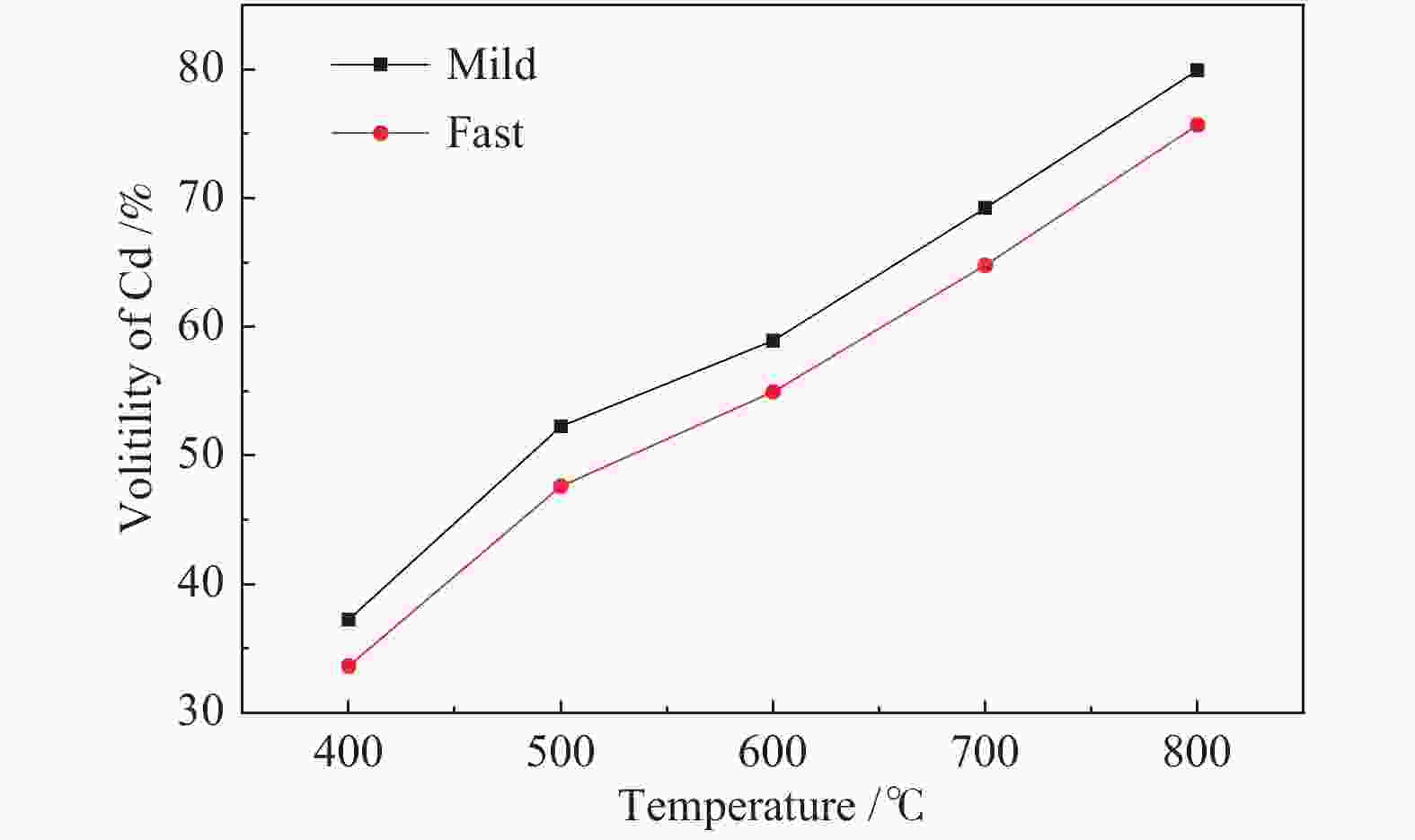
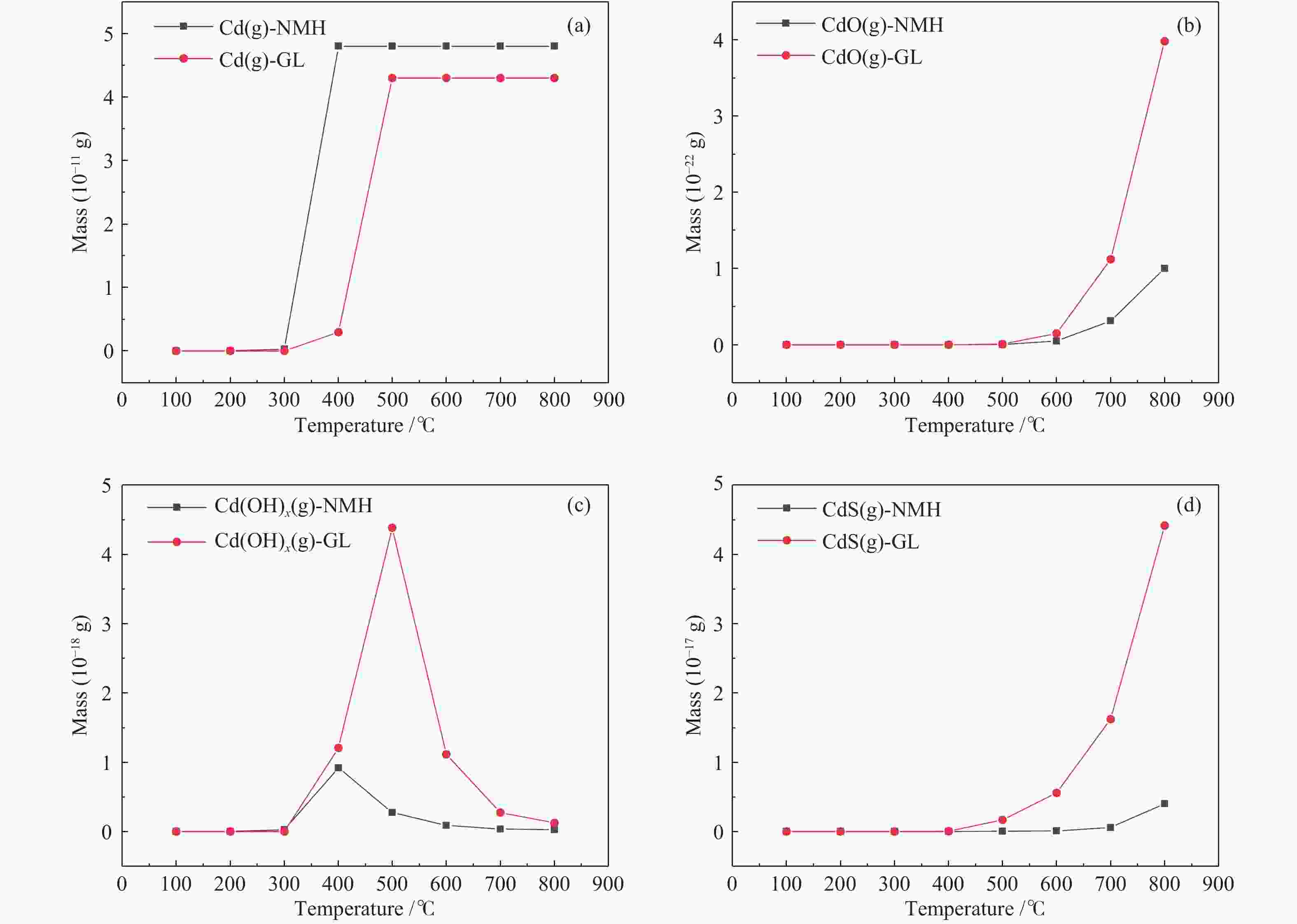
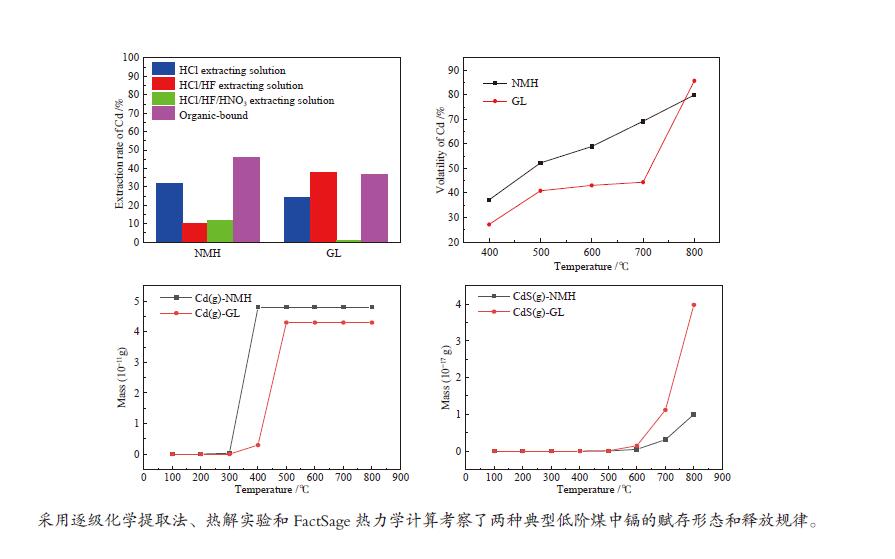
摘要: 将新疆淖毛湖煤(NMH)和内蒙高硫煤(GL)逐级化学提取,考察镉(Cd)在原煤中的赋存形态分布;分别在400−800 ℃、卧式/立式管式炉对原煤热解,考察赋存形态、热解终温、升温速率和停留时间对Cd释放规律的影响;利用FactSage软件模拟在理想条件下Cd的迁移和转化。结果表明,NMH和GL中Cd的赋存形态分布分别为有机质结合态(46%与37%)、碳酸盐结合态(32%与24%)、二硫化物结合态(12%与1%)、铝硅酸盐结合态(10%与38%)。Cd的赋存形态分布严重影响其挥发行为,有机质结合态易在低温区挥发,而碳酸盐、铝硅酸盐和硫化物结合态需在中高温区挥发;降低热解速率与延长停留时间有助于Cd的释放。FactSage模拟表明,NMH和GL中Cd的气态产物主要有Cd、CdO、Cd(OH)x和CdS,Cd在两种煤中挥发行为的差异主要是由热解温度、赋存形态分布以及煤阶决定,模拟与实验结果吻合较好。
-
关键词:
- 热解 /
- 赋存形态分布 /
- 释放规律 /
- 重金属镉 /
- FactSage模拟
Abstract: The distribution of cadmium (Cd) occurrence modes in a Naomaohu coal (NMH) and an Inner Mongolia high sulfur coal (GL) was investigated using the sequential chemical extraction method. The influence of the occurrence modes, final pyrolysis temperature, heating rate and residence time on the release behavior of Cd during the pyrolysis of two raw coals at 400-800 ℃ in a horizontal / vertical tubular furnace was examined respectively. The simulation of the migration and transformation of Cd under ideal conditions by FactSage software was carried out. The results show that Cd in NMH and GL is present in an organic matter state by 46% and 37%, a carbonate state by 32% and 24%, a disulfide state by 12% and 1%, and an aluminosilicate state by 10% and 38%, respectively. The distribution of occurrence modes seriously affects the volatilization behavior of Cd, in which the Cd in the organic matter state is easy to volatilize at low temperature, while those in the carbonate, aluminosilicate and sulfide state can volatilize at medium and high temperature; and reducing pyrolysis rate and extending residence time are conducive to the release of Cd. The FactSage simulation shows that Cd, CdO, Cd(OH)x and CdS are the major gaseous products of Cd during the pyrolysis of NMH and GL coal, and the difference of volatilization behavior of Cd in two kinds of coal is mainly determined by pyrolysis temperature, distribution of occurrence modes and coal rank, which are in good agreement with the experimental results.-
Key words:
- pyrolysis /
- distribution of occurrence modes /
- release law /
- heavy metal Cd /
- FactSage simulation
-
表 1 原煤基本性质
Table 1 Basic properties of NMH and GL raw coal
Sample NMH GL Proximate analysis w/% Mad 9.16 10.13 Ad 9.95 12.13 Vdaf 52.76 38.49 FCdaf 47.24 61.51 Ultimate analysis w/% Cdaf 77.41 76.72 Hdaf 6.60 5.07 Odaf (calculated by difference) 13.22 15.84 Ndaf 2.16 0.81 St,d 0.55 1.77 Ash composition wd/% SiO2 39.35 32.57 Al2O3 16.64 14.03 Fe2O3 5.88 10.11 CaO 23.30 17.00 Na2O 2.95 0.57 MgO 1.23 6.43 K2O 0.38 0.42 SO3 8.83 17.84 Concentration of Cd/(μg·g−1) 0.048 0.043 表 2 逐级化学提取步骤
Table 2 Sequential chemical extraction procedure
Step Reagent Extraction time/h Temperature/℃ Extraction mode 1 5 mol/L HCl 6 60 bound to carbonate 2 40% HF 6 60 bound to aluminosilicate 3 5 mol/L HNO3 6 45 bound to disulfide 4 30% H2O2 6 85 bound to organic mater 表 3 计算时输入的参数
Table 3 Parameters for calculation
Major element Mass/g Ash component Mass/g Trace element Mass/g NMH C 633.2 CaO 34 Cd $ 4.8\; \times \;10^{-5} $ H 54 Na2O 3 O 108.1 SiO2 29.7 N 17.7 Fe2O3 9.9 S 5 Al2O3 13 MgO 1.2 GL C 767.2 CaO 20.6 Cd $ 4.3\; \times\;10^{-5} $ H 50.7 Na2O 0.7 O 158.4 SiO2 39.5 N 8.1 Fe2O3 12.3 S 17.7 Al2O3 17 MgO 7.8 -
[1] 徐彦辉. 准东煤热解过程中无机钠的转化及对热解产物的影响[D]. 哈尔滨: 哈尔滨工业大学, 2015.XU Yan-jun. The transformation of inorganic sodium and it's effect on the products during the pyrolysis of Zhundong coal[D]. Harbin: Harbin Institute of Technology, 2015. [2] 党钾涛, 解强. 煤中有害微量元素及其在加工转化中的行为研究进展[J]. 现代化工,2016,36(7):59−63.DANG Jia-tao, XIE Qiang. Hazardous trace elements in coal and their bahaviors during processing and utilization[J]. Mod Chem Ind,2016,36(7):59−63. [3] ZHOU L, ZHANG G, REINMÖLLER M, MEYER B. Effect of inherent mineral matter on the co-pyrolysis of highly reactive brown coal and wheat straw[J]. Fuel,2019,239:1194−1203. doi: 10.1016/j.fuel.2018.11.114 [4] 陈高琪, 张铭, 纪律, 马军. 青海低变质煤低温干馏试验研究[J]. 化学工程与技术,2012,2(4):122−125.CHEN Gao-qi, ZHANG Ming, JI Lv, MA Jun. The research for low temperature carbonization of low metamorphic coal in Qinghai[J]. Hans J Chem Eng Technol,2012,2(4):122−125. [5] 梁鹏, 曲旋, 毕继诚. 炉前煤低温干馏的工艺研究[J]. 燃料化学学报,2008,36(4):401−405.LIANG Peng, QU Xuan, BI Ji-cheng. Study on the low temperature coal pyrolysis by solid heat carrier in a moving bed pyrolyzer[J]. J Fuel Chem Technol,2008,36(4):401−405. [6] 胡浩权. 煤直接转化制高品质液体燃料和化学品[J]. 化工进展,2016,35(12):4096−4098.HU Hao-quan. Coal direct conversion to high quality liquid fuels and chemicals[J]. Chem Ind Eng Prog,2016,35(12):4096−4098. [7] PENG H, WANG B, YANG F, CHEN F. Study on the environmental effects of heavy metals in coal gangue and coal combustion by ReCiPe2016 for life cycle impact assessment[J]. J Fuel Chem Technol,2020,48(11):1402−1408. doi: 10.1016/S1872-5813(20)30090-6 [8] SONG Q, ZHAO H, JIA J, YANG L, LV W, BAO J, SHU X, GU Q, ZHANG P. Pyrolysis of municipal solid waste with iron-based additives: A study on the kinetic, product distribution and catalytic mechanisms[J]. J Clean Prod,2020,258:120682. doi: 10.1016/j.jclepro.2020.120682 [9] ZHOU L, GUO H, WANG X, CHU M, ZHANG G, ZHANG L. Effect of occurrence mode of heavy metal elements in a low rank coal on volatility during pyrolysis[J]. Int J Coal Sci Technol,2019,6(2):235−246. doi: 10.1007/s40789-019-0251-8 [10] ZAJUSZ-ZUBEK E, KONIECZYŃSKI J. Dynamics of trace elements release in a coal pyrolysis process☆[J]. Fuel,2003,82(10):1281−1290. doi: 10.1016/S0016-2361(03)00031-0 [11] GUO R, YANG J, LIU Z. Behavior of trace elements during pyrolysis of coal in a simulated drop-tube reactor[J]. Fuel,2004,83(6):639−643. doi: 10.1016/j.fuel.2003.08.021 [12] WEI X, ZHANG G, CAI Y, LI L, LI H. The volatilization of trace elements during oxidative pyrolysis of a coal from an endemic arsenosis area in southwest Guizhou, China[J]. J Anal Appl Pyrolysis,2012,98(98):184−193. [13] GUO R, YANG J, LIU D, LIU Z. Transformation behavior of trace elements during coal pyrolysis[J]. Fuel Process Technol,2002,77(25):137−143. [14] SCACCIA S, MECOZZI R. Trace Cd, Co, and Pb elements distribution during Sulcis coal pyrolysis: GFAAS determination with slurry sampling technique[J]. Microchem J,2012,100:48−54. [15] 周玲妹, 郭豪, 初茉, 畅志兵, 王晓兵, 张冠军, 龚永强, 曲洋. 低阶煤中镉的赋存对其在热解中释放的影响[J]. 煤炭学报,2019,44(1):323−331.ZHOU Ling-mei, GUO Hao, CHU Mo, CHANG Zhi-bing, WANG Xiao-bing, ZHANG Guan-jun, GONG Yong-qiang, QU Yang. Effects of occurrence mode of Cd in low rank coal on its volatility during pyrolysis process[J]. J China Coal Soc,2019,44(1):323−331. [16] 魏晓飞. 黔西南高砷煤燃烧过程中有害元素释放规律的研究[D]. 北京: 中国科学院大学, 2012.WEI Xiao-fei. The characteristics of hazardous element release during combustion of the high arsenic coal from southwestern Guizhou, China[D]. Beijing: University of Chinese Academy of Sciences, 2012. [17] CHEN C, LUO Z, YU C. Release and transformation mechanisms of trace elements during biomass combustion[J]. J Hazard Mater,2019,380:120857. doi: 10.1016/j.jhazmat.2019.120857 [18] 刘瑞卿. 煤转化过程中砷、硒、铅的热稳定性及转化行为研究[D]. 太原: 中国科学院山西煤炭化学研究所, 2009.LIU Rui-qing. Study on thermal stability and transformation behavior of As, Se and Pb during coal conversion[D]. Taiyuan: Shanxi Institute of coal chemistry, Chinese Academy of Sciences, 2009. [19] 陈萍, 旷红伟, 唐修义. 煤中砷的分布和赋存规律研究[J]. 煤炭学报,2002,(3):259−263.CHEN Ping, KUANG Hong-wei, TANG Xiu-yi. Distribution and occurrence modes of arsenic in coal[J]. J China Coal Soc,2002,(3):259−263. [20] 郭欣, 郑楚光, 刘迎晖, 刘晶, 陆晓华. 煤中汞, 砷, 硒赋存形态的研究[J]. 工程热物理学报,2001,(6):763−766.GUO Xin, ZHENG Chu-guang, LIU Ying-hui, LIU Jing, LU Xiao-hua. The study on the modes of occurrence of mercury, arsenic and selenium in coal[J]. J Eng Therm,2001,(6):763−766. [21] SDAI S, WANG P, WARD C R, TANG Y, SONG X, JIANG J, HOWER J C, LI T, SEREDIN V V, WAGNER N J, JIANG Y, WANG X, LIU J. Elemental and mineralogical anomalies in the coal-hosted Ge ore deposit of Lincang, Yunnan, southwestern China: Key role of N2-CO2-mixed hydrothermal solutions[J]. Int J Coal Geol,2015,152:19−46. doi: 10.1016/j.coal.2014.11.006 [22] LIU J, YANG Z, YAN X, JI D, YANG Y, HU L. Modes of occurrence of highly-elevated trace elements in superhigh-organic-sulfur coals[J]. Fuel,2015,156(4):190−197. [23] 赵晶, 关腾, 李姣龙, 王金喜. 平朔矿区9~#煤中镉、铬和铊的含量分布及赋存状态[J]. 河北工程大学学报(自然科学版),2011,28(4):56−59.ZHAO Jing, GUANG Teng, LI Jiao-long, WANG Jin-xi. The distribution and occurrence state of Cd, Cr, and Tl of 9# coal in Pingshuo mine area[J]. J Heibei Univ Eng: Nat Sci Ed,2011,28(4):56−59. [24] GUO R, YANG J, LIU D, LIU Z. The fate of As, Pb, Cd, Cr and Mn in a coal during pyrolysis[J]. J Anal Appl Pyrolysis,2003,70(2):555−562. doi: 10.1016/S0165-2370(03)00025-1 [25] 于敦喜, 徐明厚, 姚洪, 刘小伟, 周科. 煤燃烧中无机矿物向颗粒物的转化规律[J]. 工程热物理学报,2008,(3):507−510.YU Xi-dun, XU Ming-hou, YAO Hong, LIU Xiao-wei, ZHOU Ke. Transformation of inorganic minerals to particulate matter in coal combustion[J]. J Eng Therm,2008,(3):507−510. [26] ZHANG G J. Mineral matter behavior during co-gasification of coal and biomass[D]. Freiberg: Technische Universität Bergakademie Freiberg, 2014. [27] DANG J, XIE Q, LIANG D, WANG X, HE D, CAO J. The fate of trace elements in yanshan coal during fast pyrolysis[J]. Minerals, 2016, 6(2): 35. [28] MENG D, WANG T, XU J, CHEN X. A numerical study on the pyrolysis of large coal particles: Heat transfer and volatile evolution[J]. Fuel,2019,254:115668. doi: 10.1016/j.fuel.2019.115668 [29] 郭瑞霞. 煤热解过程中若干微量有害元素转化行为的研究[D]. 太原: 中国科学院山西煤炭化学研究所, 2003.GUO Rui-xia. Study on behavior of several trace elements during pyrolysis of coals[D]. Taiyuan: Shanxi Institute of Coal Chemistry, Chinese Academy of Sciences, 2003. -




 下载:
下载: