Hydrogenation of naphthalene to decalin catalyzed by Pt supported on WO3 of different crystallinity at low temperature
-
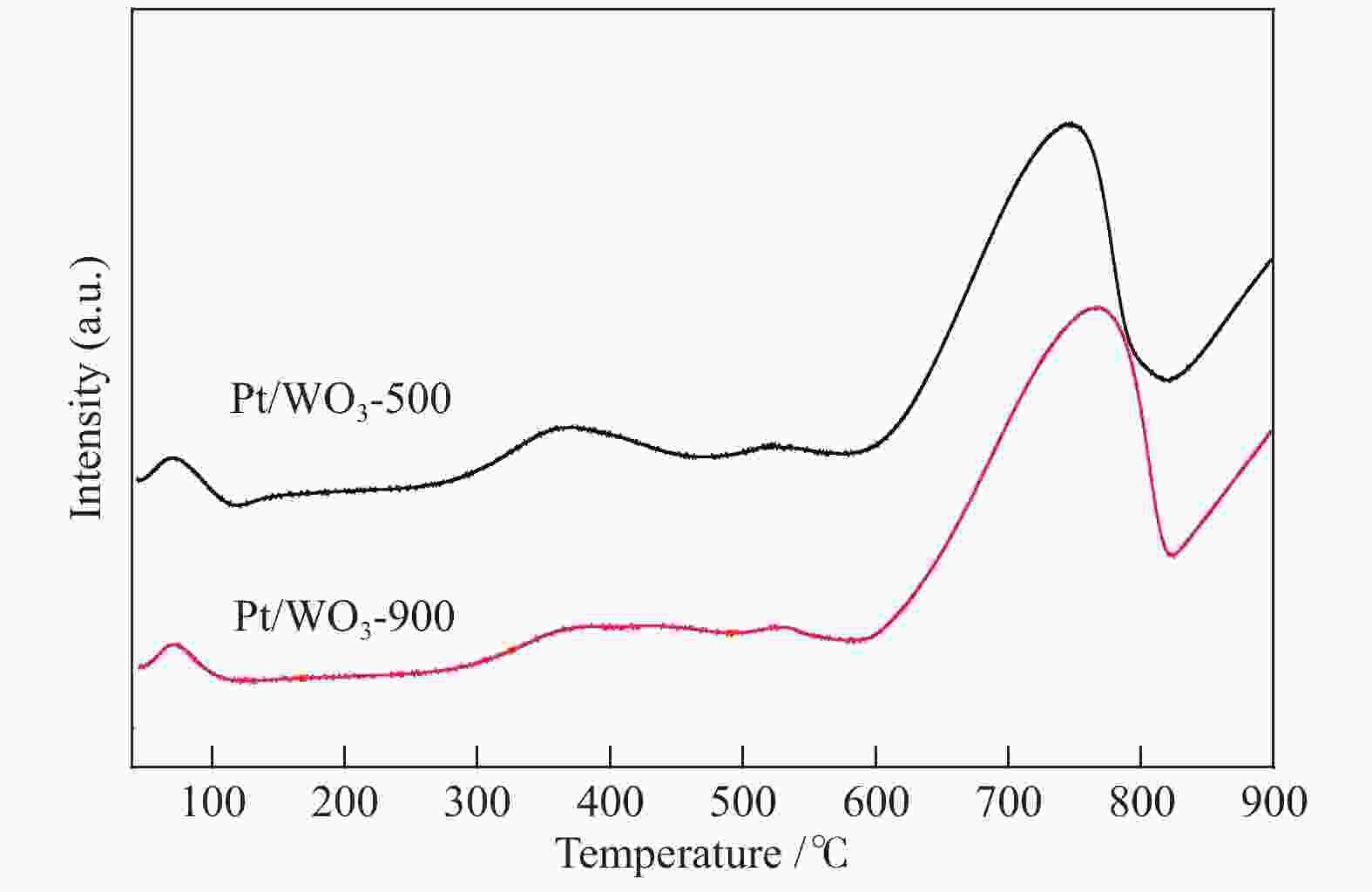
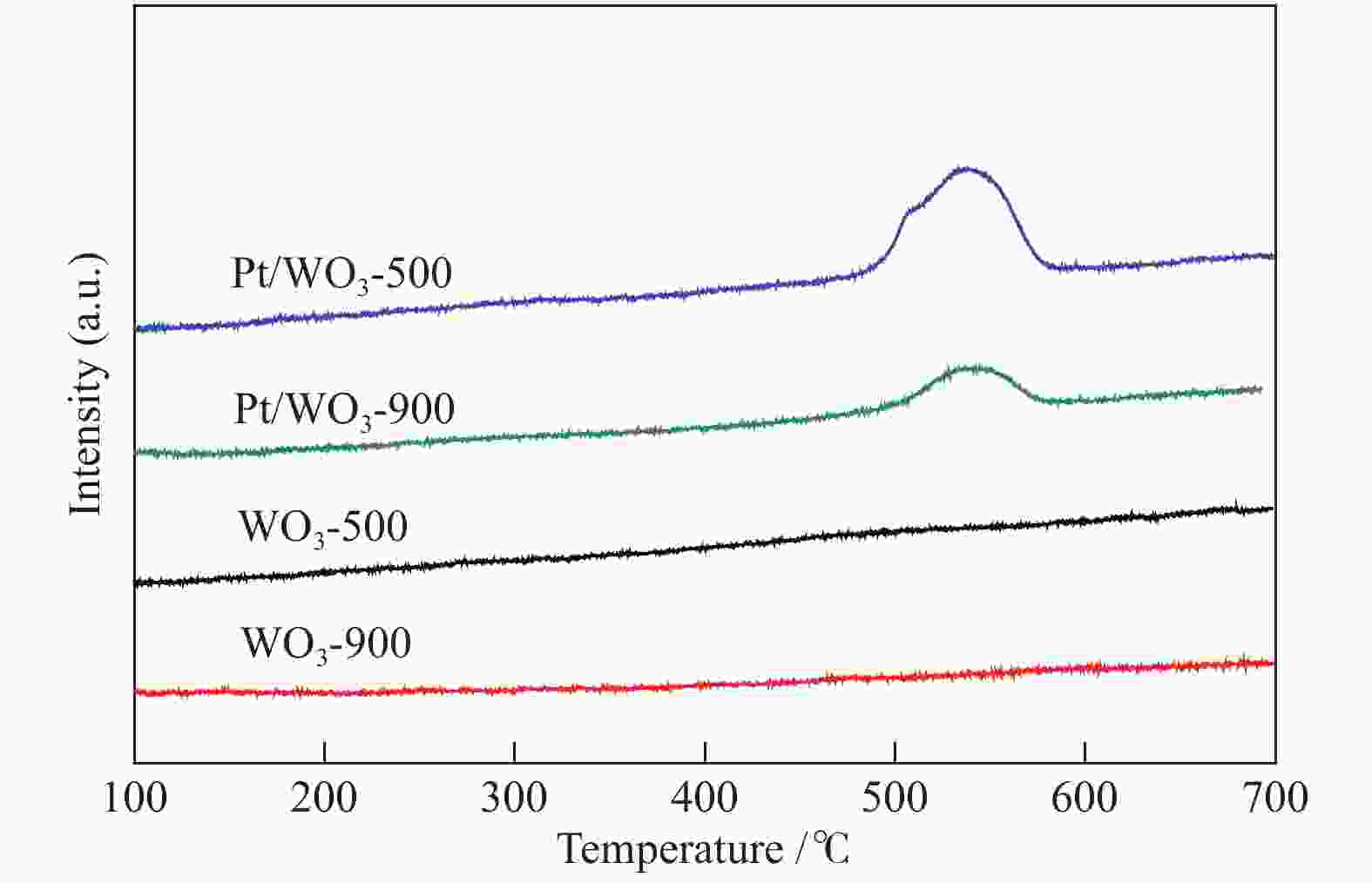
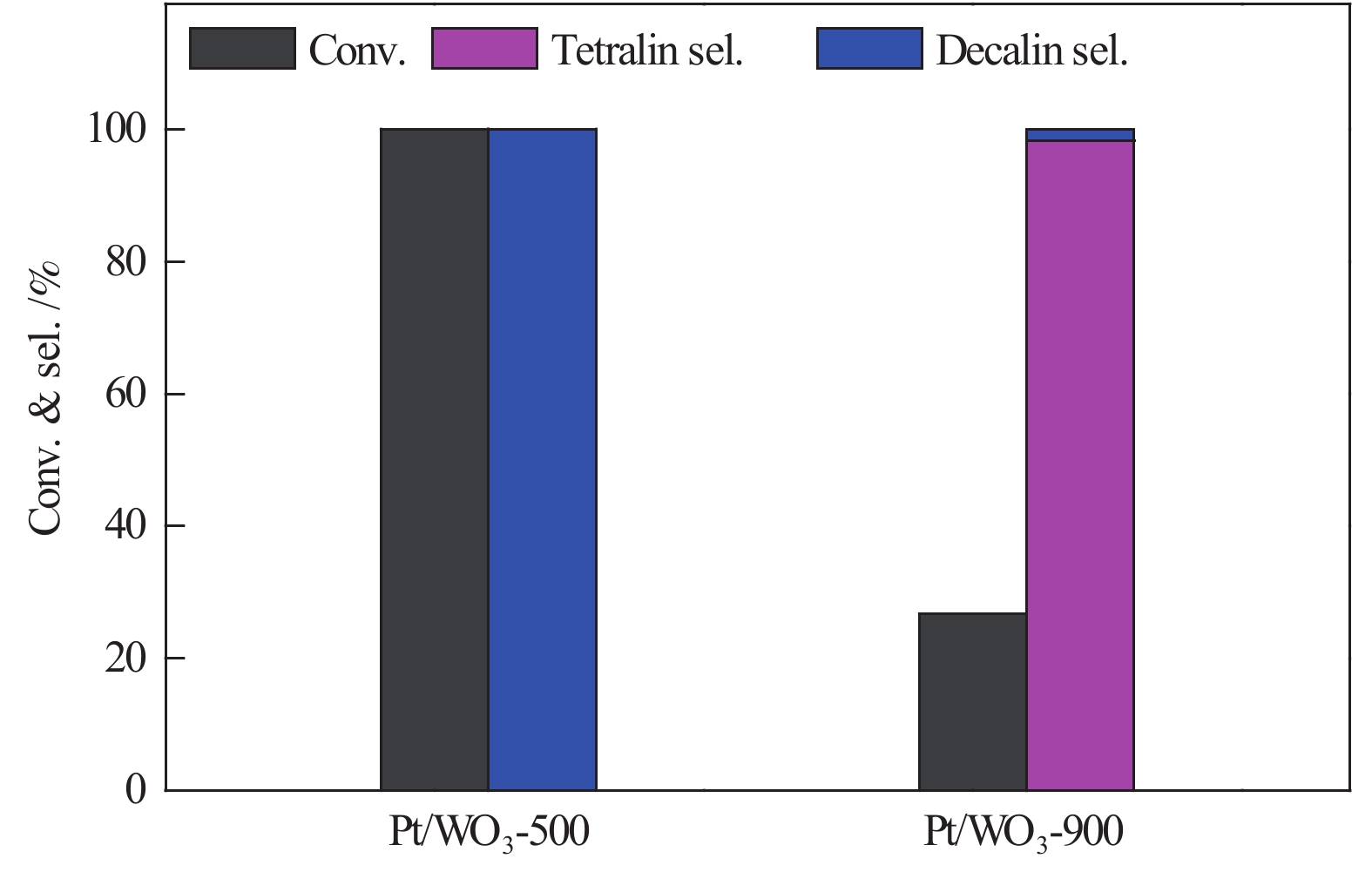
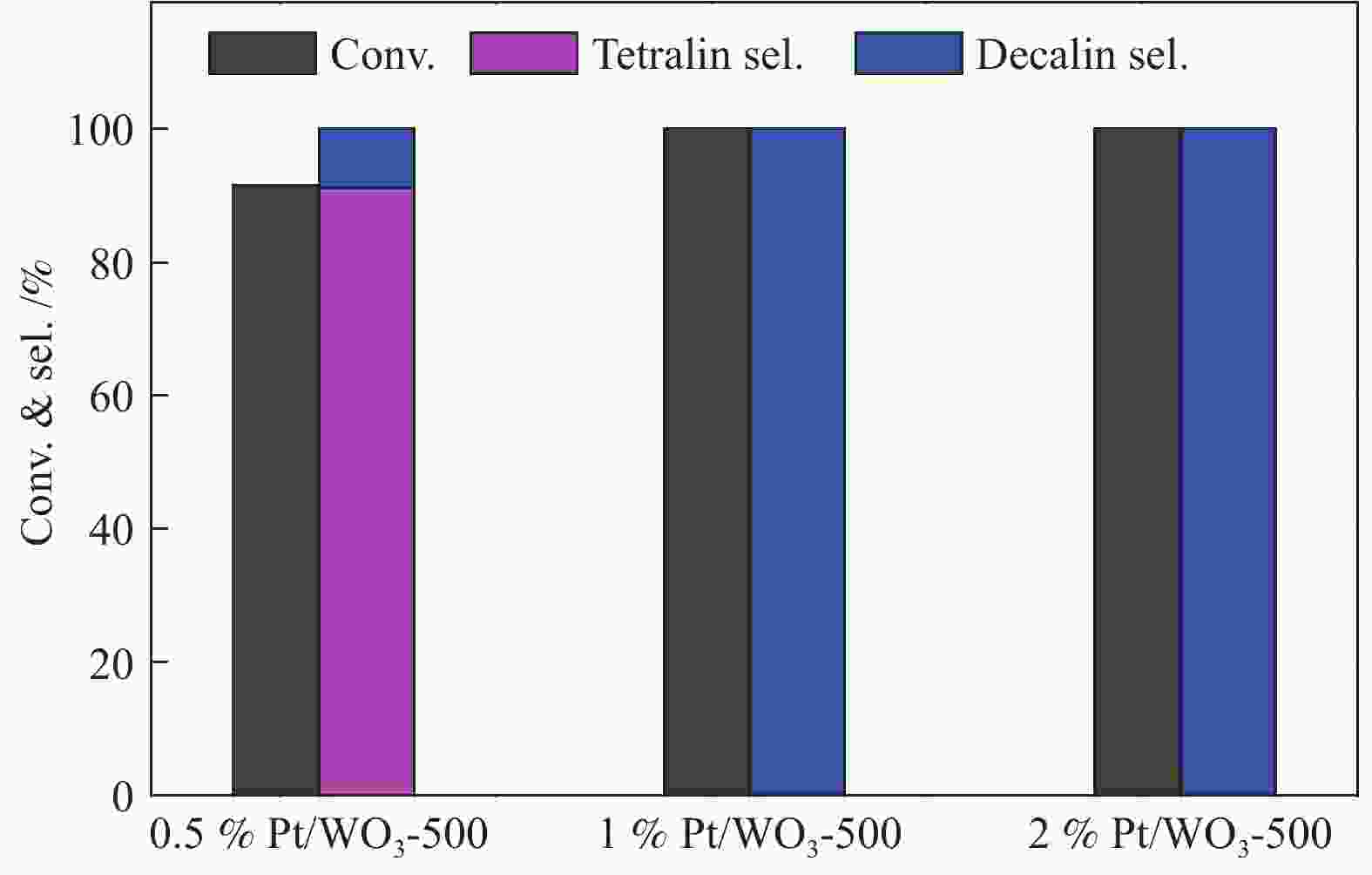
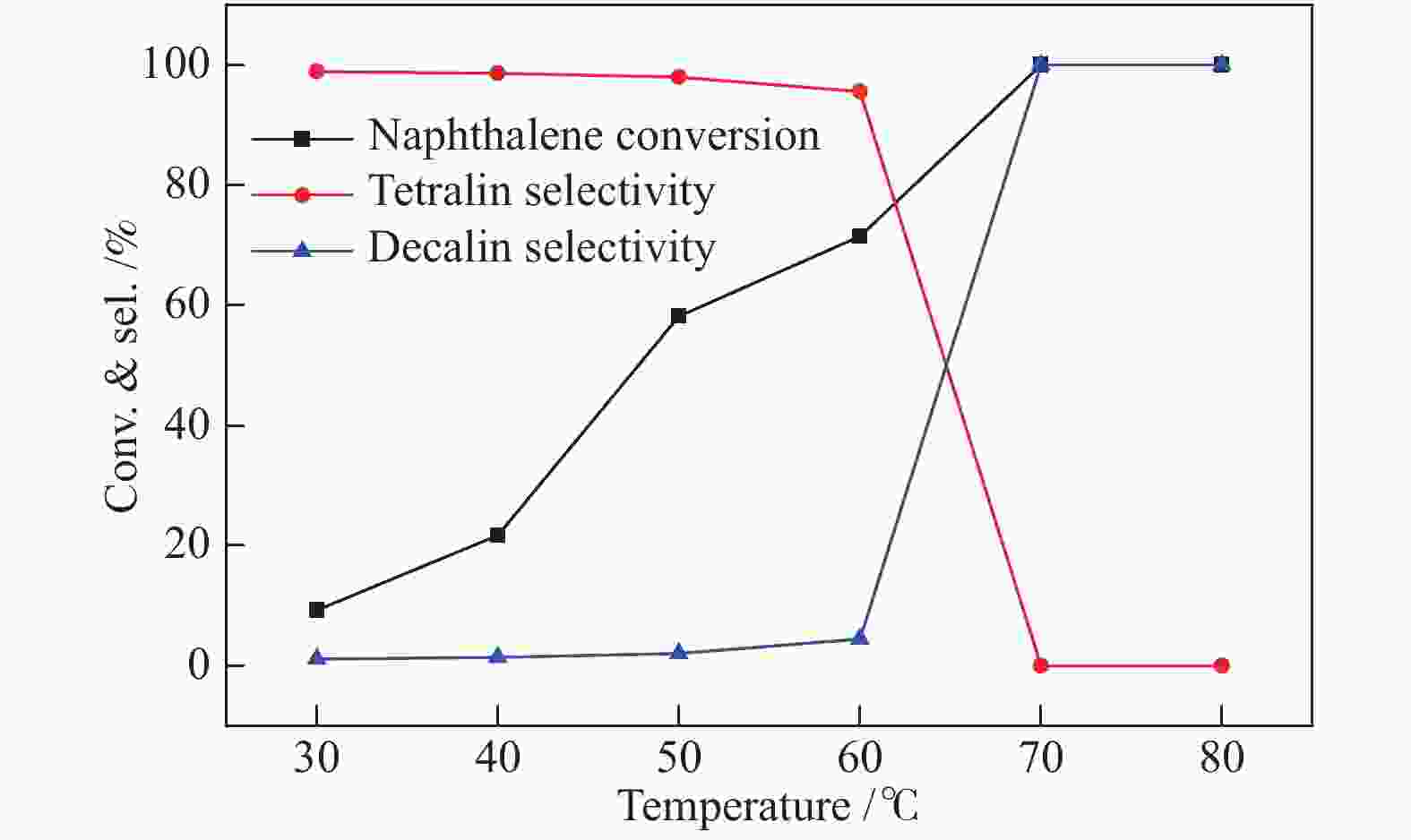
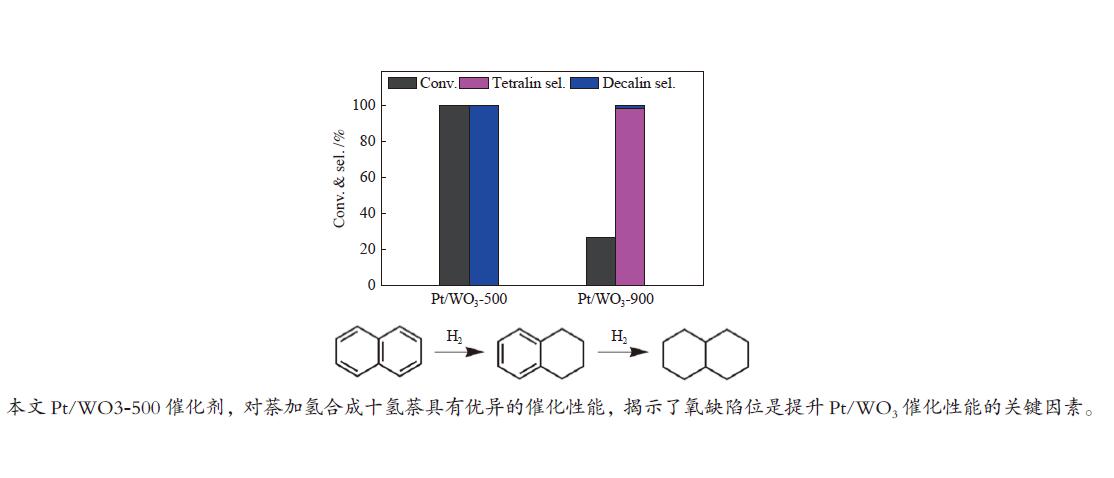
摘要: 不同温度下直接煅烧偏钨酸铵制备了晶化程度不同的两种氧化钨(WO3-500和WO3-900),通过XRD、SEM、TEM、XPS、H2-TPR和NH3-TPD手段对WO3载体负载Pt前后的物化性质进行了系统的表征,低温反应条件下研究了不同氧化钨负载Pt对萘加氢的催化性能。与WO3-900载体相比,低温煅烧得到具有较低的结晶程度,载体中大量的W5+物种和负载的Pt具有强的相互作用,并显示出较强的酸性。在低的反应温度下(70 ℃),Pt/WO3-500催化剂对萘加氢合成十氢萘具有优异的催化性能,萘的转化率和十氢萘的选择性均达到100%;在Pt/WO3-900催化下萘的转化率和十氢萘的选择性仅为26.7%和1.7%。结合催化剂的表征和催化反应结果,揭示了氧化钨中的氧缺陷位是提升Pt/WO3催化性能的关键因素,对设计高效的WO3负载 Pt催化剂催化萘合成十氢萘提供了一定的理论指导。Abstract: Two tungsten oxides (WO3-500 and WO3-900) were prepared at different calcination temperatures by using ammonium mettungstate as tungsten source. The physicochemical properties of WO3 before and after Pt loading were systematically characterized by XRD, SEM, TEM, XPS, H2-TPR, and NH3-TPD. The influence of the crystallization degree of WO3 on the hydrogenation of naphthalene was studied under low-temperature reaction conditions. Compared with the WO3-900 support, WO3-500 obtained by calcination at lower temperature exhibited a lower crystallization degree with a large amount of W5+ species, which resulted in strong interactions with Pt. The strong interaction between W species and Pt contributed to the high acidic strength. The Pt/WO3-500 catalyst demonstrated excellent catalytic performance for naphthalene hydrogenation to decalin at low reaction temperature (70 ℃) with full conversion and 100% decalin selectivity. Under identical conditions, conversion and decalin selectivity were only 26.7% and 1.7% over the Pt/WO3-900 catalyst. Combining the characteristics of the catalysts and their catalytic results, we revealed the promoting role of oxygen defects in WO3-supported Pt catalysts in the hydrogenation of naphthalene to decalin, which will provide theoretical guidance for designing efficient WO3-supported Pt catalysts for hydrogenation reactions.
-
Key words:
- naphthalene /
- decalin /
- platinum /
- tungsten oxide /
- hydrogenation /
- catalyst /
- crystallization
-
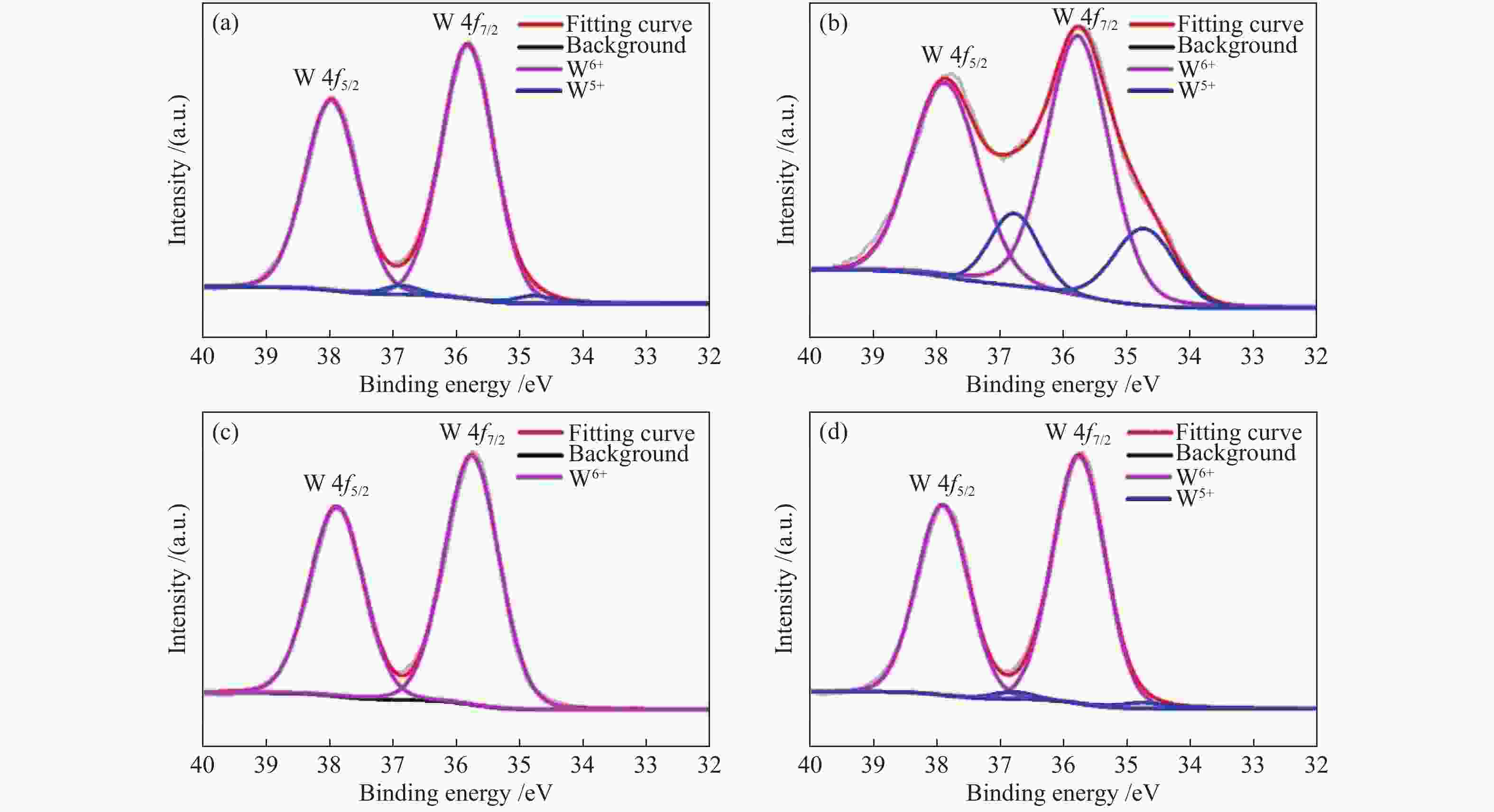
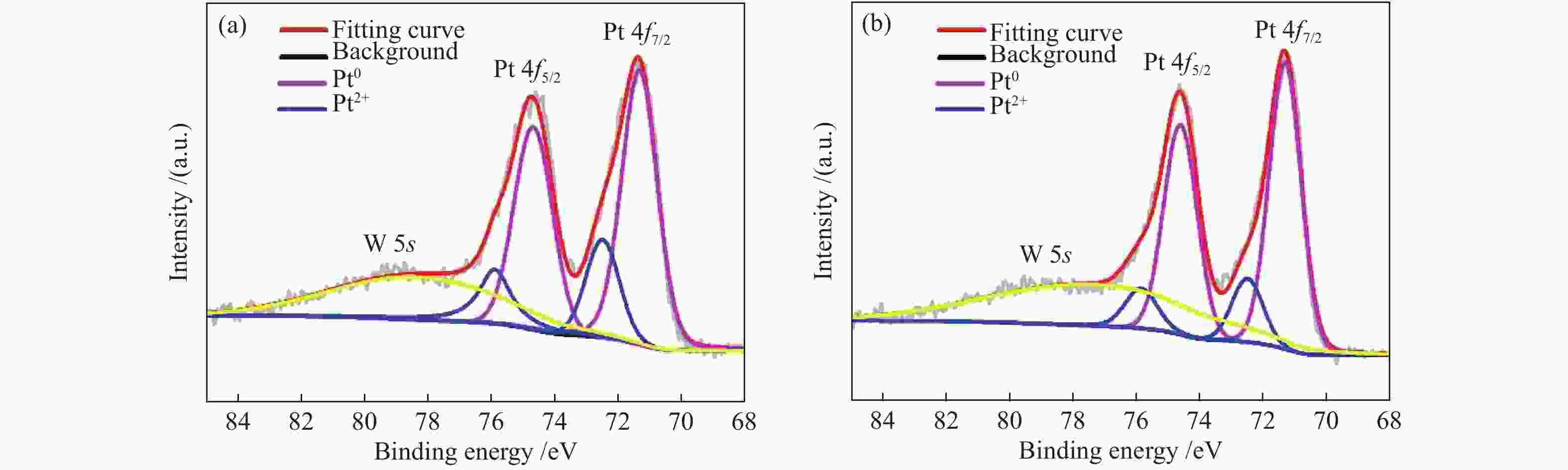
表 1 Pt/WO3-500和Pt/WO3-900两种催化剂中Pt和W物种的XPS催化剂的XPS拟合
Table 1 XPS fitting results of Pt and W species in the two catalysts
Catalyst 4f7/2 E/eV Pt0/(Pt0+Pt2+) Pt2+/(Pt0+Pt2+) W5+/(W5++W6+) Pt0 Pt2+ W5+ W6+ Pt/WO3-900 71.32 72.48 34.75 35.76 0.83 0.17 0.05 Pt/WO3-500 71.34 72.5 34.73 35.77 0.75 0.25 0.21 -
[1] AZP ROZ G, BLANCO C G, BANCIELLA C. The use of solvents for purifying industrial naphthalene from coal tar distilled oils[J]. Fuel Process. Technol,2008,89(2):111−117. doi: 10.1016/j.fuproc.2007.06.001 [2] 杨惠斌. 菲加氢反应体系的研究[D]. 上海: 华东理工大学, 2015.YANG Hui-bin. Study on hydrogenation system of phenanthrene[D]. Shanghai: East China University of Science and Technology, 2015. [3] 谭凤宜. 固定床法萘催化加氢合成十氢萘工艺研究[D]. 南京: 南京工业大学, 2006.TAN Feng-yi. Study on hydrogenation of naphthalene to decalin in a fixed bed reactor[D]. Nanjing: Nanjing University of Technology, 2006. [4] PANG M, LIU C, XIA W, MUHLER M, LIANG C. Activated carbon supported molybdenum carbides as cheap and highly efficient catalyst in the selective hydrogenation of naphthalene to tetralin[J]. Green Chem,2012,14(5):1272−1276. doi: 10.1039/c2gc35177c [5] SHIRAI M, RODE C V, MINE E, SASAKI A, SATO O, HIYOSHI N. Ring hydrogenation of naphthalene and 1-naphthol over supported metal catalysts in supercritical carbon dioxide solvent[J]. Catal Today,2006,115(1-4):248−253. doi: 10.1016/j.cattod.2006.02.048 [6] ESCOBAR J, BARRERA M C, SANTES V, TERRAZAS J E. Naphthalene hydrogenation over Mg-doped Pt/Al2O3 [J]. Catal Today,2017,296:197−204. doi: 10.1016/j.cattod.2017.04.064 [7] 杨平, 辛靖, 李明丰, 聂红. 四氢萘加氢转化研究进展[J]. 石油炼制与化工,2011,42(8):1−6.YANG Ping, XIN Jing, LI Ming-feng, NIE Hong. Research advances in the hydrogenation of tetralin[J]. Pet Procrss Petrochem,2011,42(8):1−6. [8] 郄志强, 张子毅, 荆洁颖, 杨志奋, 冯杰, 李文英. Ni2P负载量对Ni2P/Ce-Al2O3催化剂结构及萘加氢性能的影响[J]. 燃料化学学报,2019,47(6):718−724.QI Zhi-qiang, ZHANG Zi-ying, JING Jie-ying, YANG Zhi-fen, FENG Jie, LI Wen-ying. Effect of Ni2P loading on the structure and naphthalene hydrogenation performance of Ni2P/Ce-Al2O3 catalyst[J]. J Fuel Chem Technol,2019,47(6):718−724. [9] 米星, 何广湘, 郭晓燕, 杨索和, 罗国华, 徐新, 靳海波. Ni/γ-Al2O3催化剂上萘加氢生成十氢萘的催化反应研究[J]. 燃料化学学报,2018,46(7):879−885.MI Xing, HE Guang-xiang, GUO Xiao-yan, YANG Suo-he, LUO Guo-hua, XU Xin, JIN Hai-bo. Effect of reaction conditions on the hydrogenation of naphthalene to decalin over Ni/Al2O3 catalyst[J]. J Fuel Chem Technol,2018,46(7):879−885. [10] 佟瑞利, 王永刚, 张旭, 张海永, 戴谨泽, 林雄超, 许德平. P改性NiW/γ-Al2O3的低温焦油芳烃组分加氢性能研究[J]. 燃料化学学报,2015,43(12):1461−1469. doi: 10.1016/S1872-5813(16)30003-2TONG Rui-li, WANG Yong-gang, ZHANG Xu, ZHANG Hai-yong, DAI Jin-ze, LIN Xiong-chao, XU De-ping. Effect of phosphorus modification on the catalytic properties of NiW/γ-Al2O3 in the hydrogenation of aromatics from coal tar[J]. J Fuel Chem Technol,2015,43(12):1461−1469. doi: 10.1016/S1872-5813(16)30003-2 [11] CHEN H, YANG H, OMOTOSO O, DING L, BRIKER Y, YING Z, RING Z. Contribution of hydrogen spillover to the hydrogenation of naphthalene over diluted Pt/RHO catalysts[J]. Appl Catal A: Gen,2009,358(2):103−109. doi: 10.1016/j.apcata.2008.12.045 [12] ALBERTAZZI S, GANZERLA R, GOBBI C, LENARDA M, VACCARI A. Hydrogenation of naphthalene on noble-metal-containing mesoporous MCM-41 aluminosilicates[J]. J Mol Catal A: Chem,2003,200(1/2):261−270. doi: 10.1016/S1381-1169(03)00025-6 [13] LU C M, LIN Y M, WANG I. Naphthalene hydrogenation over Pt/TiO2-ZrO2 and the behavior of strong metal-Support interaction (SMSI)[J]. Appl Catal A: Gen,2000,198(s1/2):223−234. [14] LIN S D, SONG C. Noble metal catalysts for low-temperature naphthalene hydrogenation in the presence of benzothiophene[J]. Catal Today,1996,31(1):93−104. [15] 张小菲, 邵正锋, 毛国强, 何德民, 张秋民, 梁长海. 萘在贵金属Pd、Pt及Pd-Pt催化剂上的加氢活性及耐硫性能[J]. 物理化学学报,2010,26(10):2691−2698. doi: 10.3866/PKU.WHXB20101006ZHANG Xiao-fei, SHAO Zheng-feng, MAO Guo-qiang, HE De-min, ZHANG Qiu-min, LIANG Chang-hai. Naphthalene Hydrogenation Activity over Pd, Pt and Pd-Pt Catalysts and Their Sulfur Tolerance[J]. Acta Phys-Chim Sin,2010,26(10):2691−2698. doi: 10.3866/PKU.WHXB20101006 [16] 李剑, 武海顺, 杨丽娜, 杨肖嵘, 马波. 超声辅助浸渍法制备高分散Pt/CMK-3-US加氢脱萘催化剂[J]. 无机化学学报,2017,33(4):583−588.LI Jian, WU Hai-shun, YANG Li-na, YANG Xiao-rong, MA Bo. Ultrasound assisted synthesis of highly dispersed Pt/CMK-3-US as catalyst for hydrogenation of naphthalene[J]. Chin J Inorg Chem,2017,33(4):583−588. [17] HUANG T C, KANG B C. The Hydrogenation of naphthalene with platinum/alumina-aluminum phosphate catalysts[J]. Ind Eng Chem Res,1995,34(9):2955−2963. doi: 10.1021/ie00048a005 [18] REDDY K M, SONG C. Synthesis of mesoporous zeolites and their application for catalytic conversion of polycyclic aromatic hydrocarbons[J]. Catal Today,1996,31(1/2):137−144. [19] LIU J, ZHANG H, LU N, YAN X, LI R. Influence of acidity of mesoporous ZSM-5-supported Pt on naphthalene hydrogenation[J]. Ind Eng Chem Res,2020,59(3):1056−1064. doi: 10.1021/acs.iecr.9b04411 [20] 周妍, 张然, 王东生, 杨斌. 水热法制备Mo掺杂WO3纳米材料及其光致变色性质的研究[J]. 材料工程,2012,10(83):73−79+83.ZHOU Yan, ZHANG Ran, WANG Dong-sheng, YANG Bin. Hydrothermal synthesis and photochromic characterization of Mo doped WO3 nanomaterial[J]. J Mater Eng,2012,10(83):73−79+83. [21] 于卓斌, 赵强, 袁雯, 李晋平. 原位电解制备WO3/Ni-Bi析氧催化剂[J]. 太阳能学报,2014,35(10):1883−1888.YU Zhuo-bin, ZHAO Qiang, YUAN Wen, LI Jin-ping. Preparation of WO3/Ni-Bi oxygen-evolution catalyst by situ electrolysis[J]. Acta Energi Sin,2014,35(10):1883−1888. [22] 孙启梅, 花亮, 陈雨, 金江, 陈榕, 张华. 质子交换膜燃料电池阴极催化剂用WO3的制备与性能[J]. 南京工业大学学报(自然科学版),2008,30(6):10−13.SUN Qi-mei, HUA Liang, CHEN Yu, JIN Jiang, CHEN Rong, ZHANG Hua. Preparation and properties of WO3 as electrocatalyst for proton exchange membrane fuel cell cathode[J]. J Nanjing Univ Technol: (Nat Sci Ed),2008,30(6):10−13. [23] WANG J, ZHAO X C, LEI N, LI L, ZHANG L L, XU S T, MIAO S, PAN X L, WANG A Q, ZHANG T. Hydrogenolysis of glycerol to 1, 3-propanediol under low hydrogen pressure over WOx-supported single/pseudo-single atom Pt catalyst[J]. ChemSusChem,2016,9(8):784−790. doi: 10.1002/cssc.201501506 [24] ZHENG H, JIAN Z O, STRANO M S, KANER RB, MITCHELL A, KALANTAR-ZADEH K. Nanostructured tungsten oxide-properties, synthesis, and applications[J]. Adv Funct Mater,2011,21(12):2175−2196. doi: 10.1002/adfm.201002477 [25] 杨欢, 王桂赟, 田伟松, 童春杰. 单斜相WO3的水热合成及其光催化性能的研究[J]. 燃料化学学报,2018,46(11):1359−1369.YANG Huan, WANG Gui-yun, TIAN Wei-song, TONG Chun-jie. Hydrothermal synthesis of monoclinic WO3 and its photocatalytic hydrogen production performance[J]. J Fuel Chem Technol,2018,46(11):1359−1369. [26] TU J G, LEI H P, YU Z J, JIAO S Q. Ordered WO3-x nanorods: Facile synthesis and their electrochemical properties for aluminum-ion batteries[J]. Chem Commun,2018,54(11):1343−1346. doi: 10.1039/C7CC09376D [27] KARIM W, SPREAFICO C, KLEIBERT A, GOBRECHT J, VANDE V J, EKINCI Y, JEROEN A, BOKHOVEN V, CLELIA, KARIM. Catalyst support effects on hydrogen spillover[J]. Nature,2017,541(7635):68−71. doi: 10.1038/nature20782 [28] GARCIA F S, GANDARIAS I, REQUIES J, GUEMEZ M B, BENNICI S, AUROUX A, ARIAS P L. New approaches to the Pt/WOx/Al2O3 catalytic system behavior for the selective glycerol hydrogenolysis to 1, 3-propanediol[J]. J Catal,2015,323:65−75. doi: 10.1016/j.jcat.2014.12.028 [29] NIU Y F, ZHAO B B, LIANG Y, LIU L, DONG J X. Promoting role of oxygen deficiency on WO3 supported Pt catalyst for glycerol hydrogenolysis to 1, 3-propanediol[J]. Ind Eng Chem Res,2020,59(16):7389−7397. doi: 10.1021/acs.iecr.9b07067 [30] ZHU S, GAO X, ZHU Y, CUI J, ZHENG H, LI Y. SiO2 promoted Pt/WOx/ZrO2 catalysts for the selective hydrogenolysis of glycerol to 1, 3-propanediol[J]. Appl Catal B: Environ,2014,158−159:391−399. doi: 10.1016/j.apcatb.2014.04.049 [31] TIAN H, CUI X Z, ZENG L M, SU L, SONG Y L, SHI J L. Oxygen vacancy-assisted hydrogen evolution reaction of the Pt/WO3 electrocatalyst[J]. J Mater Chem A,2019,7(11):6285−6293. doi: 10.1039/C8TA12219A [32] SONG J J, HUANG Z F, PAN L, ZOU J J, ZHANG X W, WANG L. Oxygen-deficient tungsten oxide as versatile and efficient hydrogenation catalyst[J]. ACS Catal,2015,5(11):6594−6599. doi: 10.1021/acscatal.5b01522 [33] FAN Y, CHENG S J, WANG H, YE D H, XIE S H, PEI Y, HU H R, HUA W M, LI Z H, QIAO M H, ZONG B N. Nanoparticulate Pt on mesoporous SBA-15 doped with extremely low amount of W as a highly selective catalyst for glycerol hydrogenolysis to 1, 3-propanediol[J]. Green Chem,2017,19(9):2174−2183. doi: 10.1039/C7GC00317J [34] GARC A-FERN NDEZ S, GANDARIAS I, REQUIES J, SOULIMANI F. The role of tungsten oxide in the selective hydrogenolysis of glycerol to 1, 3-propanediol over Pt/WOx/Al2O3[J]. Appl Catal B: Environ,2017,204:260−272. doi: 10.1016/j.apcatb.2016.11.016 [35] ZHOU W, LUO J, WANG Y, LIU J F. WOx domain size, acid properties and mechanistic aspects of glycerol hydrogenolysis over Pt/WOx/ZrO2[J]. Appl Catal B: Environ,2019,242:410−421. doi: 10.1016/j.apcatb.2018.10.006 [36] DONG L, ZHOU Z W, QIN J, YANG L. Cu-WOx-TiO2 catalysts by modified evaporation-induced self-assembly method for glycerol hydrogenolysis to 1, 3-propanediol[J]. Chem Select,2018,3(9):2479−2486. [37] YUE C C, ZHU X C, RIGUTTTO M. Acid catalytic properties of reduced tungsten and niobium-tungsten oxides[J]. Appl Catal B: Environ,2015,163:370−381. doi: 10.1016/j.apcatb.2014.08.008 [38] FENG S H, ZHAO B B, LIU L, DONG J X. Platinum supported on WO3-doped aluminosilicate: a highly efficient catalyst for selective hydrogenolysis of glycerol to 1, 3-propanediol[J]. Ind Eng Chem Res,2017,56(39):11065−11074. doi: 10.1021/acs.iecr.7b02951 [39] PARK K C, YIM D J, IHM S K. Characteristics of Al-MCM-41 supported Pt catalysts: effect of Al distribution in Al-MCM-41 on its catalytic activity in naphthalene hydrogenation[J]. Catal Today,2002,74(3):281−290. [40] BOUCHY M, DUFRESNE P, KASZTELAN S. Hydrogenation and hydrocracking of a model light cycle oil feed. 1. Properties of a sulfided nickel-molybdenum hydrotreating catalyst[J]. Ind Eng Chem Res,1992,32(12):553−574. [41] WANG M L, QIAN X Q, XIE L Q, FENG H H, YE L M, DUAN X P, YUAN Y Z. Synthesis of a Ni phyllosilicate with controlled morphology for deep hydrogenation of polycyclic aromatic hydrocarbons[J]. ACS Sustainable Chem Eng,2019,7(2):1989−1997. doi: 10.1021/acssuschemeng.8b04256 [42] CORMA A, MARTINEZ A, MARTINEZ-SORIAY V. Hydrogenation of aromatics in diesel fuels on Pb/MCM-4l catalysts[J]. J Catal,1997,169(2):480−489. doi: 10.1006/jcat.1997.1737 -





 下载:
下载: