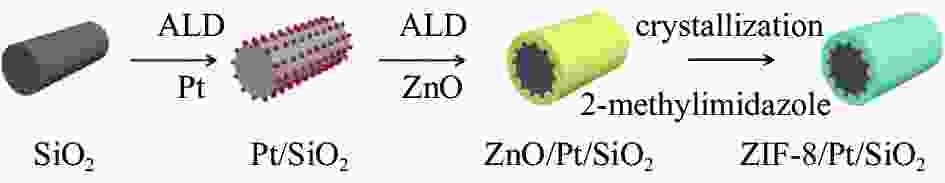
Synthesis of ZIF-8-coated Pt/SiO2 by vapor deposition for alkyne semi-hydrogenation
-
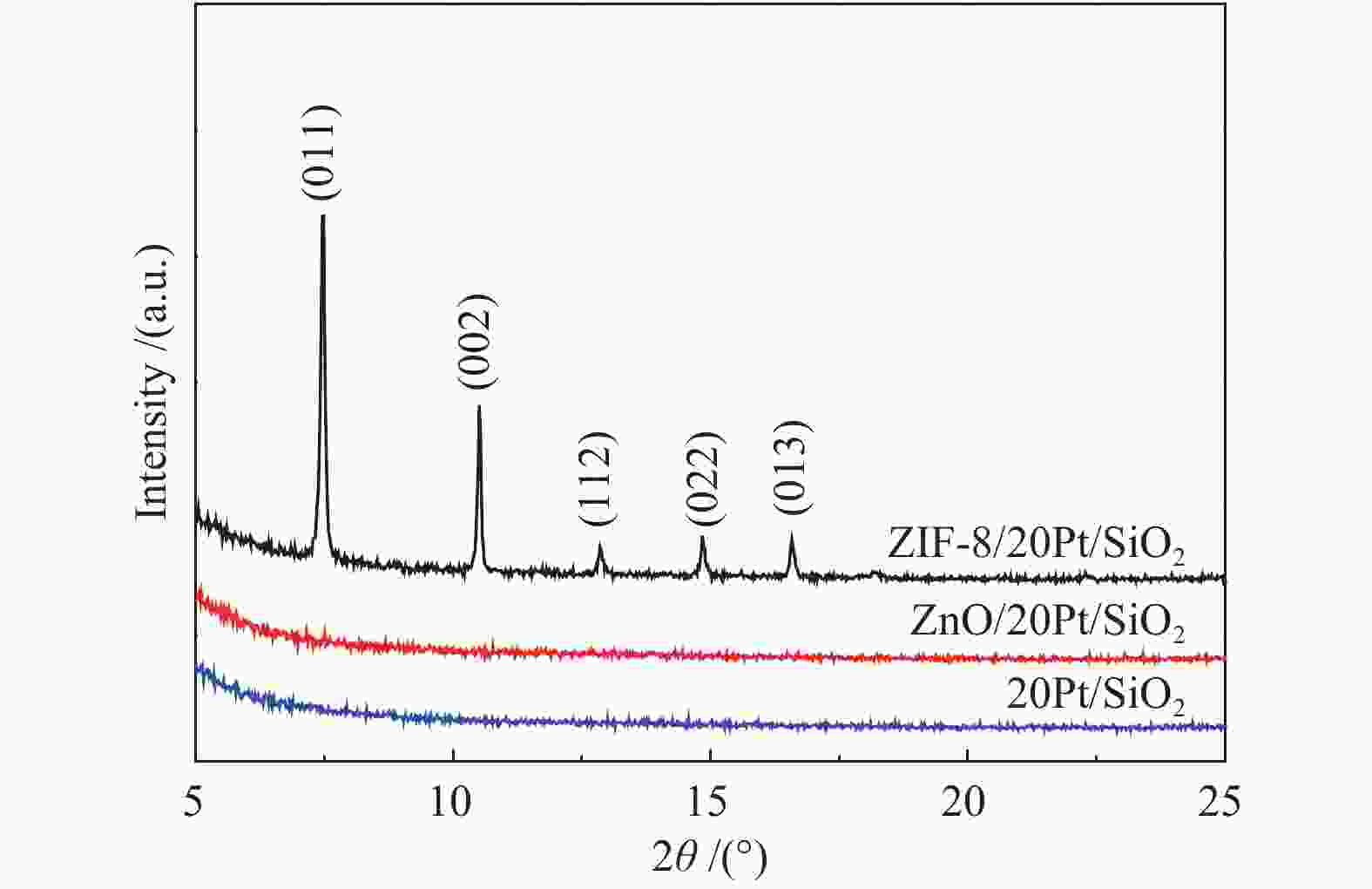
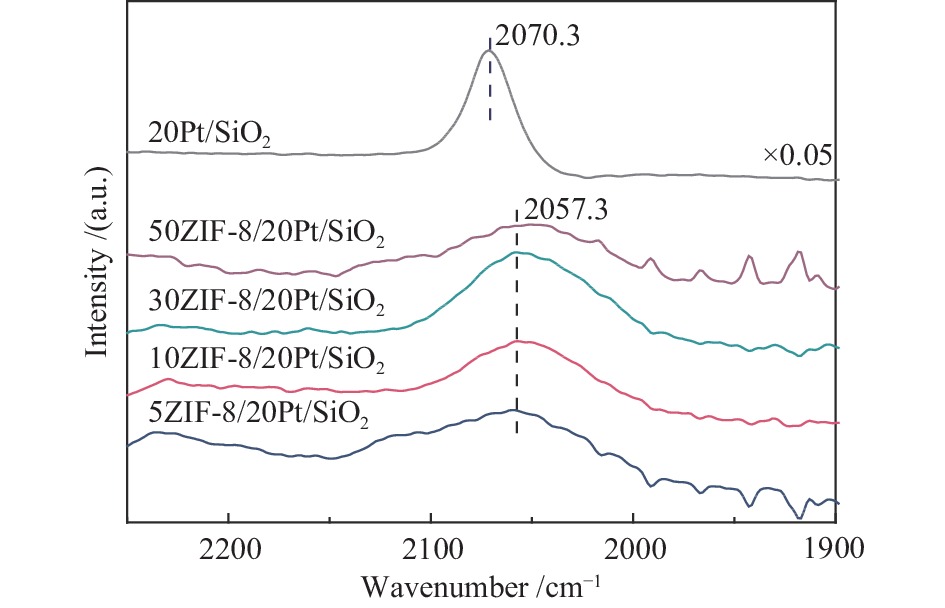
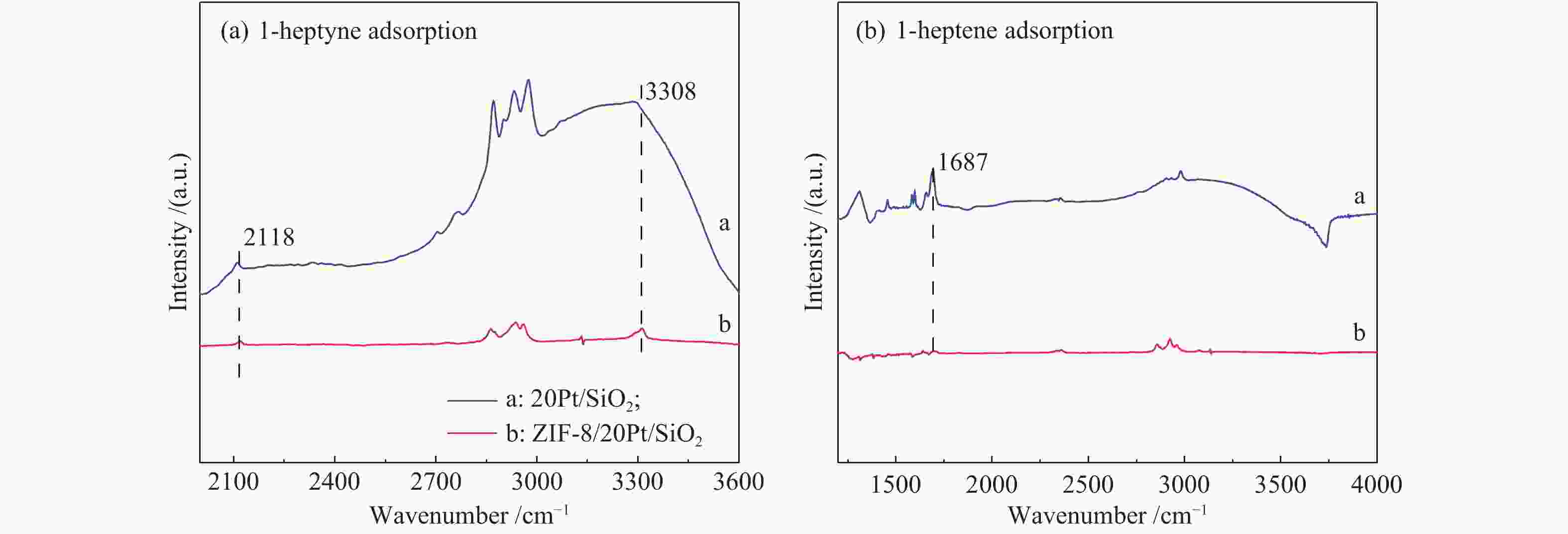
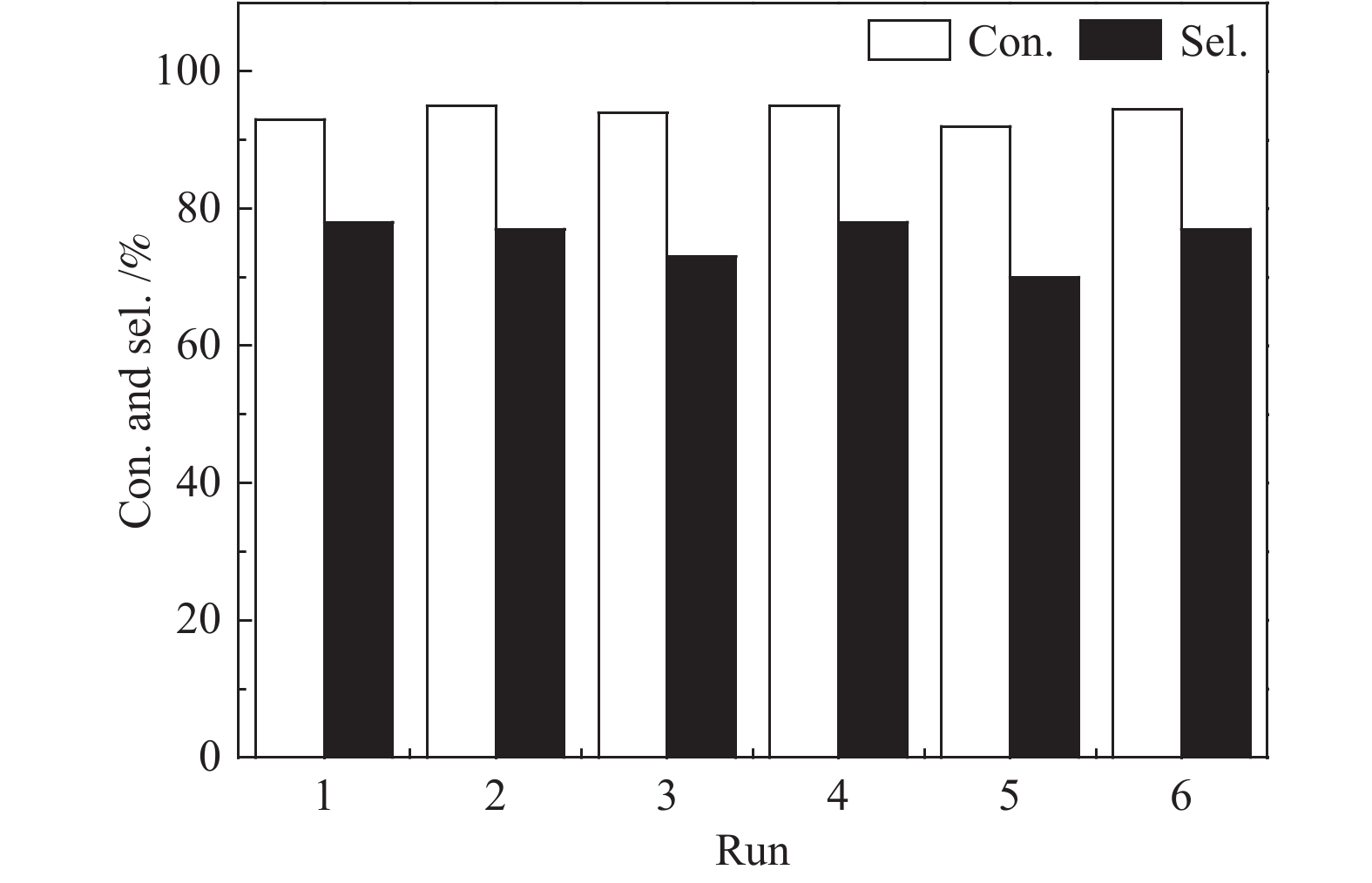
摘要: 以原子层沉积技术与气相转晶法相结合的方法,制备出ZIF-8/Pt/SiO2催化剂。该催化剂中Pt纳米颗粒沉积在SiO2纳米线表面,进一步通过气相法被厚度可控的ZIF-8纳米薄膜包覆后形成三明治结构。通过XRD、TEM、BET、ICP-MS、XPS、CO-DRIFT等表征对催化剂结构进行了系统的分析,以1-庚炔加氢为探针反应研究了ZIF-8薄膜对Pt催化性能的影响。结果表明,Pt颗粒高度分散在SiO2纳米线上,所制备的ZIF-8薄膜厚度可控,能够实现Pt/SiO2纳米催化剂表面的均一、保形性包覆。ZIF-8薄膜改变了Pt电子状态,提高了 Pt表面电子密度。在1-庚炔加氢反应中,ZIF-8包覆后, Pt催化庚炔加氢制庚烯的选择性由14%增加到70%。此外,降低ZIF-8纳米薄膜厚度能够提高反应的活性,几乎不影响烯烃的选择性。Abstract: In this work, ZIF-8/Pt/SiO2 catalysts were prepared by combining atomic layer deposition (ALD) and vapor phase conversion methods. First, Pt metal nanoparticles were deposited on SiO2 nanowires by ALD. Then, ZnO was further deposited, also by ALD. Subsequently, the ZnO film was converted into ZIF-8 film by vapor phase crystallization to form a sandwich structure (ZIF-8/Pt/SiO2). The microstructures of the catalysts were characterized by XRD, TEM, BET, IC-MS, XPS, and CO-DRIFT. It was shown that the Pt particles were highly dispersed on the SiO2 nanowires before and after coating with ZIF-8, and the ZIF-8 film was coated continuously on the entire catalyst with high conformity. The performance of the catalyst was studied by using the semi-hydrogenation of 1-heptyne as a probe reaction. The ZIF-8 film induces an electron density increase in the Pt component, leading to an increase of the olefin selectivity from 14% to 70% in the 1-heptyne hydrogenation reaction. A reduced thickness of the ZIF-8 film increases the catalytic activity but does not affect the selectivity of 1-heptylene.
-
Key words:
- atomic layer deposition /
- vapor phase crystallization /
- ZIF-8 film /
- semi-hydrogenation
-
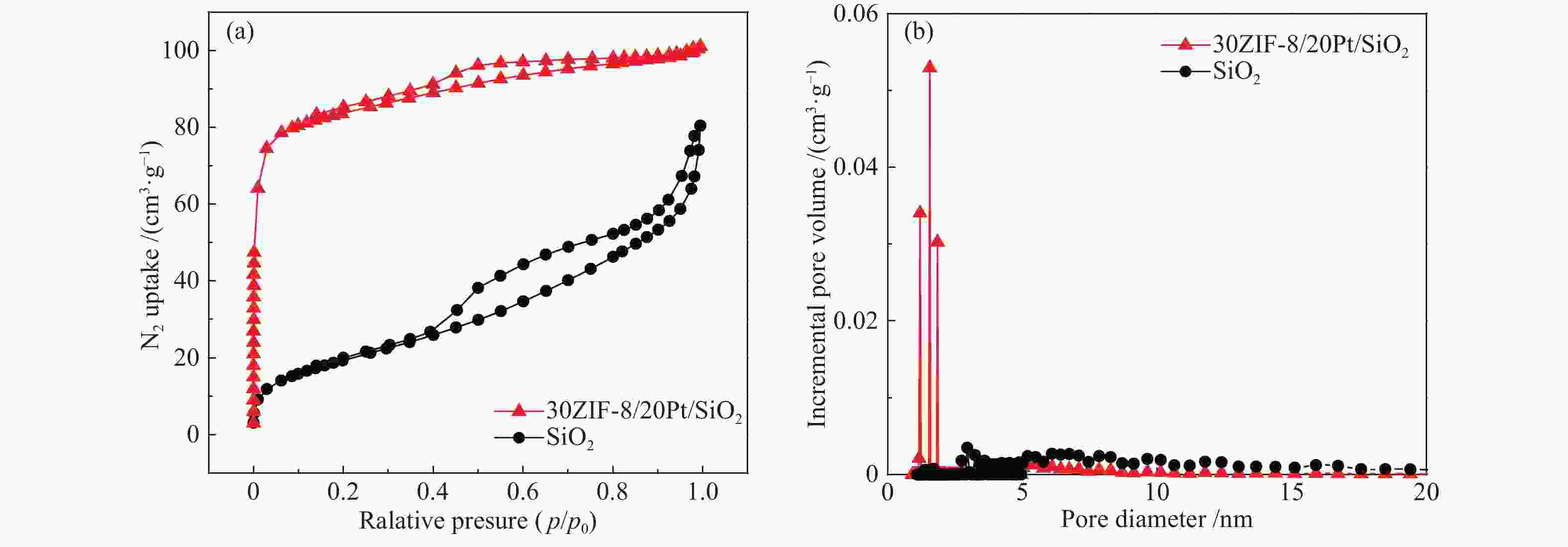
表 1 催化剂的比表面积和孔道结构
Table 1 Surface area and pore structure of the catalysts
Sample ABET/
(cm2·g−1)vmicro/
(cm3·g−1)dmicro/
nmvmeso/
(cm3·g−1)dmaso/
nmSiO2 72.0 0 0 0.1 6.6 10ZIF-8/20Pt/SiO2 290.4 0.06 1.2 0.1 3.6 30ZIF-8/20Pt/SiO2 320.2 0.10 1.2 0.1 3.6 50ZIF-8/20Pt/SiO2 420.1 0.11 1.2 0.1 3.5 ABET: BET surface area; vmicro: the HK method was used to obtain the micropore volume; dmicro: the HK method was used to obtain the average micropore diameter; Vmeso: the BJH method was used to obtain the mesopore volume; dmaso: the BJH method was used to obtain the average mesopore diameter 表 2 不同样品上Pt、Zn元素的负载量
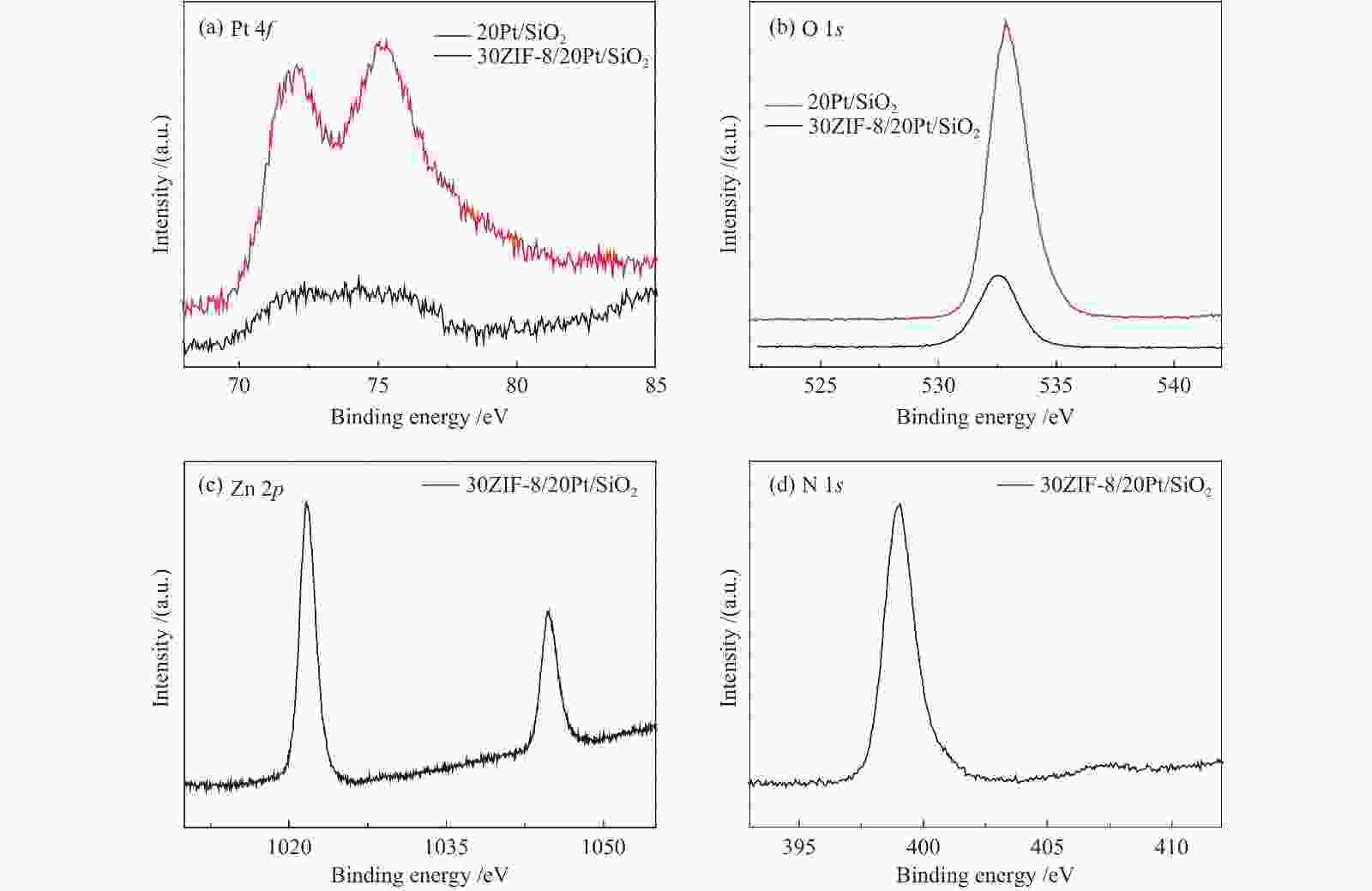
Table 2 Pt and Zn loading on different samples
Sample 20Pt/SiO2 10ZIF-8/20Pt/SiO2 30ZIF-8/20Pt/SiO2 w(Pt)/% 2.9 1.8 1.6 w(Zn)/% 0 6.9 27.3 表 3 不同催化剂的1-庚炔加氢反应性能a
Table 3 Catalytic performance of different catalysts for 1-heptyne hydrogenation
Sample Reaction time/min Conversion/% Selectivity/% CO adsorption/(mmol·g−1)b TOFc/ (102·h−1) 20Pt/SiO2 30 100 14 0.10 51.6 10ZIF-8/20Pt/SiO2 235 100 65 0.04 1.62 30ZIF-8/20Pt/SiO2 445 100 70 0.02 2.36 50ZIF-8/20Pt/SiO2 1155 100 67 0.006 0.66 a: reaction conditions: 100 μL 1-heptyne, 15 mg catalyst, 10 mL isopropanol, reaction temperature 60 ℃, 1 MPa hydrogen; b: CO adsorption data; c: TOF=Nconversed heptyne /(NCO × t), where N represents the number of moles and t represents the reaction time (h) -
[1] WANG X, LI M, CAO C, LIU C, LIU J, ZHU Y, ZHANG S, SONG W. Surfactant-free palladium nanoparticles encapsulated in ZIF-8 Hollow nanospheres for size-selective catalysis in liquid–phase solution[J]. ChemCatChem,2016,8(20):3224−3228. doi: 10.1002/cctc.201600846 [2] CHOE K, ZHENG F, WANG H, YUAN Y, ZHAO W, XUE G, QIU X, RI M, SHI X, WANG Y, LI G, TANG Z. Fast and selective semi-hydrogenation of alkynes by palladium nanoparticles sandwiched in metal-organic frameworks[J]. Angew Chem Int Ed,2020,59(9):3650−3657. doi: 10.1002/anie.201913453 [3] KHOUYA A A, BA H, BAAZIZ W, NHUT J M, ROSSIN A, ZAFEIRATOS S, ERSEN O, GIAMBASTIANI G, RITLENG V, PHAM-HHH C. Palladium nanosheet-carbon black powder composites for selective hydrogenation of alkynes to alkenes[J]. ACS Appl Nano Mater,2021,4(2):2265−2277. doi: 10.1021/acsanm.1c00002 [4] 荆洁颖, 杨志奋, 王九占, 刘道诚, 冯杰, 李文英. 制备方法对Ni2P/SiO2催化剂结构及萘加氢性能的影响[J]. 燃料化学学报,2020,48(7):842−851. doi: 10.3969/j.issn.0253-2409.2020.07.009JING Jie-ying, YANG Zhi-fen, WANG Jiu-zhan, LIU Dao-cheng, FENG Jie, LI Wen-ying. Effect of preparation methods on the structure and naphthalene hydrogenation performance of Ni2P/SiO2 catalyst[J]. J Fuel Chem Technol,2020,48(7):842−851. doi: 10.3969/j.issn.0253-2409.2020.07.009 [5] 林敏, 纳薇, 叶海船, 霍海辉, 高文桂. 不同助剂对CuO-ZnO/SBA-15催化CO2加氢制甲醇性能影响的研究[J]. 燃料化学学报,2019,47(10):1214−1225. doi: 10.3969/j.issn.0253-2409.2019.10.008LIN Min, NA Wei, YE Hai-chuan, HUO Hai-hui, GAO Wen-gui. Effect of additive on CuO-ZnO/SBA-15 catalytic performance of CO2 hydrogenation to methanol[J]. J Fuel Chem Technol,2019,47(10):1214−1225. doi: 10.3969/j.issn.0253-2409.2019.10.008 [6] LIANG H, ZHANG B, GE H, GU X, ZHANG S, QIN Y. Porous TiO2/Pt/TiO2 sandwich catalyst for highly selective semihydrogenation of alkyne to olefin[J]. ACS Catal,2017,7(10):6567−6572. doi: 10.1021/acscatal.7b02032 [7] ZHAO J, CHEN C, ZHANG B, JIAO Z, ZHANG J, YANG J, QIN Y. Tuning the selectivity of Pt-catalyzed tandem hydrogenation of nitro compounds via controllable NiO decoration by atomic layer deposition[J]. Catal Commun,2019,121:48−52. doi: 10.1016/j.catcom.2018.12.015 [8] HUANG X C, LIN Y Y, ZHANG J P, CHEN X M. Ligand-directed strategy for zeolite-type metal-organic frameworks: Zinc(II) imidazolates with unusual zeolitic topologies[J]. Angew Chem Int Ed,2006,45(10):1557−1559. doi: 10.1002/anie.200503778 [9] KITTISAKMONTREE P, PONGTHAWORNSAKUN B, YOSHIDA H, FUJITA S I, ARAI M, PANPRANOT J. The liquid-phase hydrogenation of 1-heptyne over Pd-Au/TiO2 catalysts prepared by the combination of incipient wetness impregnation and deposition-precipitation[J]. J Catal,2013,297:155−164. doi: 10.1016/j.jcat.2012.10.007 [10] YAGHI O M, O’KEEFFE M, OCKWIG N W, CHAE H K, EDDAOUDI M, KIM J. Reticular synthesis and the design of new materials[J]. Nature,2003,423:705−714. [11] DHAKSHINAMOORTHY A, GARCIA H. Catalysis by metal nanoparticles embedded on metal-organic frameworks[J]. Chem Soc Rev,2012,41(15):5262−5284. doi: 10.1039/c2cs35047e [12] CHEN J, SHEN K, LI Y. Greening the processes of metal-organic framework synthesis and their use in sustainable catalysis[J]. ChemSusChem,2017,10(16):3165−3187. doi: 10.1002/cssc.201700748 [13] LIAO P Q, SHEN J Q, ZHANG J P. Metal-organic frameworks for electrocatalysis[J]. Coord Chem Rev,2018,373:22−48. doi: 10.1016/j.ccr.2017.09.001 [14] CONG S, LIU X, JIANG Y, ZHANG W, ZHAO Z. Surface enhanced raman scattering revealed by interfacial charge-transfer transitions[J]. The Innovation,2020,1(3):100051. doi: 10.1016/j.xinn.2020.100051 [15] LIU X, PANG H, LIU X, LI Q, ZHANG N, MAO L, QIU M, HU B, YANG H, WANG X. Orderly porous covalent organic frameworks-based materials: Superior adsorbents for pollutants removal from aqueous solutions[J]. The Innovation,2021,2(1):100076. doi: 10.1016/j.xinn.2021.100076 [16] SHI X, WANG L, YAN N, WANG Z, GUO L, STEINHART M, WANG Y. Fast evaporation enabled ultrathin polymer coatings on nanoporous substrates for highly permeable membranes[J]. The Innovation,2021,2(1):100088. doi: 10.1016/j.xinn.2021.100088 [17] BAKURU V R, SAMANTA D, MAJI T K, KALIDINDI S B. Transfer hydrogenation of alkynes into alkenes by ammonia borane over Pd-MOF catalysts[J]. Dalton Trans,2020,49(16):5024−5028. doi: 10.1039/D0DT00472C [18] GOLE B, SANYAL U, BANERJEE R, MUKHERJEE P S. High loading of Pd nanoparticles by interior functionalization of MOFs for heterogeneous catalysis[J]. Inorg Chem,2016,55(5):2345−2354. doi: 10.1021/acs.inorgchem.5b02739 [19] LI Y, YU L, CHEN L, HAN C, JIANG H, LIU Z, ZHENG N, WANG J, SUN M, YANG R, BAO X. Subtle side chain triggers unexpected two-channel charge transport property enabling 80% fill factors and efficient thick-film organic photovoltaics[J]. The Innovation,2021,2(1):100090. doi: 10.1016/j.xinn.2021.100090 [20] ZHAO M, YUAN K, WANG Y, LI G, GUO J, GU L, HU W, ZHAO H, TANG Z. Metal-organic frameworks as selectivity regulators for hydrogenation reactions[J]. Nature,2016,539(7627):76−80. doi: 10.1038/nature19763 [21] KIM R, RYU U, JEE S, CHOI K M. Surface coating of MOF layers on the nanocrystals of other MOFs using nanoparticle mediated nucleation for the efficient removal of formaldehyde[J]. Appl Surf Sci,2020,505:144612. doi: 10.1016/j.apsusc.2019.144612 [22] STASSEN I, STYLES M, GRENCI G, GORP H V, VANDERLINDEN W, FEYTER S D, FALCARO P, VOS D D, VEREECKEN P, AMELOOT R. Chemical vapour deposition of zeolitic imidazolate framework thin films[J]. Nat Mater,2016,15(3):304−310. doi: 10.1038/nmat4509 [23] XU T, SUN K, GAO D, LI C, HU X, CHEN G. Atomic-layer-deposition-formed sacrificial template for the construction of an MIL-53 shell to increase selectivity of hydrogenation reactions[J]. Chem Comm,2019,55(53):7651−7654. doi: 10.1039/C9CC02727K [24] GAO Z, QIN Y. Design and properties of confined nanocatalysts by atomic layer deposition[J]. Acc Chem Res,2017,50(9):2309−2316. doi: 10.1021/acs.accounts.7b00266 [25] YAN L, LIU J, ZHAO S, ZHANG B, GAO Z, GE H, CHEN Y, CAO M, QIN Y. Coaxial multi-interface hollow Ni-Al2O3-ZnO nanowires tailored by atomic layer deposition for selective-frequency absorptions[J]. Nano Res,2016,10(5):1595−1607. [26] ZHANG S, ZHANG B, LIANG H, LIU Y, QIAO Y. QIN Y. Encapsulation of homogeneous catalysts in mesoporous materials using diffusion-limited atomic layer deposition[J]. Angew Chem Int Ed,2018,57(4):1091−1095. doi: 10.1002/anie.201712010 [27] GU X M, ZHANG B, LIANG H J, GE H B, YANG H M, QIN Y. Pt/HZSM-5 catalyst synthesized by atomic layer deposition for aqueous–phase hydrogenation of levulinic acid to valeric acid[J]. J Fuel Chem Technol,2017,45(6):714−722. doi: 10.1016/S1872-5813(17)30035-X [28] CAO L, LIU W, LUO Q, YIN R, WANG B, WEISSENRIEDER J, SOLDEMO M, YAN H, LIN Y, SUN Z, MA C, ZHANG W, CHEN S, WANG H, GUAN Q, YAO T, WEI S, YANG J, LU J. Atomically dispersed iron hydroxide anchored on Pt for preferential oxidation of CO in H2[J]. Nature,2019,565(7741):631−635. doi: 10.1038/s41586-018-0869-5 [29] WANG M, GAO Z, ZHANG B, YANG H, QIAO Y, CHEN S, GE H, ZHANG J, QIN Y. Ultrathin coating of confined Pt nanocatalysts by atomic layer deposition for enhanced catalytic performance in hydrogenation reactions[J]. Chem,2016,22(25):8438−8443. doi: 10.1002/chem.201601039 [30] WU H, ZHANG B, LIANG H, ZHAI L, WANG G, QIN Y. Distance effect of Ni-Pt dual sites for active hydrogen transfer in tandem reaction[J]. The Innovation,2020,1(2):100029. doi: 10.1016/j.xinn.2020.100029 [31] YANG H, CHEN Y, QIN Y. Application of atomic layer deposition in fabricating high–efficiency electrocatalysts[J]. Chin J Catal,2020,41(2):227−241. doi: 10.1016/S1872-2067(19)63440-6 [32] ZHANG B, CHEN Y, LI J, PIPPEL E, YANG H, GAO Z, QIN Y. High efficiency Cu-ZnO hydrogenation catalyst: The tailoring of Cu-ZnO interface sites by molecular layer deposition[J]. ACS Catal,2015,5(9):5567−5573. doi: 10.1021/acscatal.5b01266 [33] ZHANG B, GUO X W, LIANG H, GE H, GU X, CHEN S, YANG H, QIN Y. Tailoring Pt-Fe2O3 Interfaces for selective reductive coupling reaction to synthesize imine[J]. ACS Catal,2016,6(10):6560−6566. doi: 10.1021/acscatal.6b01756 [34] 魏云彪, 苏策, 李思良, 王庆利. ZIF-8复合催化剂的制备及催化性能研究[J]. 化工新型材料,2020,48(8):219−226.WEI Yun-biao, SU Ce, LI Si-liang, WANG Qing-li. Preparation and performance of Au@ZIF-8 composite catalyst[J]. New Chem Mater,2020,48(8):219−226. [35] KUO C H, TANG Y, CHOU L Y, SNEED B T, BRODSKY C N, ZHAO Z, TSUNG C K. Yolk-shell nanocrystal@ZIF-8 nanostructures for gas-phase heterogeneous catalysis with selectivity control[J]. J Am Chem Soc,2012,134(35):14345−14348. doi: 10.1021/ja306869j [36] LU G, FARHA O K, ZHANG W, HUO F, HUPP J T. Engineering ZIF-8 thin films for hybrid MOF-based devices[J]. Adv Mater,2012,24(29):3970−3974. doi: 10.1002/adma.201202116 [37] NGUYEN L T L, LE K K A, PHAN N T S. A zeolite imidazolate framework ZIF-8 catalyst for Friedel-Crafts acylation[J]. Chin J Catal,2012,33(4/6):688−696. doi: 10.1016/S1872-2067(11)60368-9 [38] LIU Z, WU A, YAN H, SU D, JIN C, GUO H, WANG L, TIAN C. An effective "precursor-transformation" route toward the high-yield synthesis of ZIF-8 tubes[J]. Chem Commun,2020,56(19):2913−2916. doi: 10.1039/C9CC08724A -





 下载:
下载: