Molecular dynamics simulation of diffusion and separation of CO2/CH4/N2 on MER zeolites
-
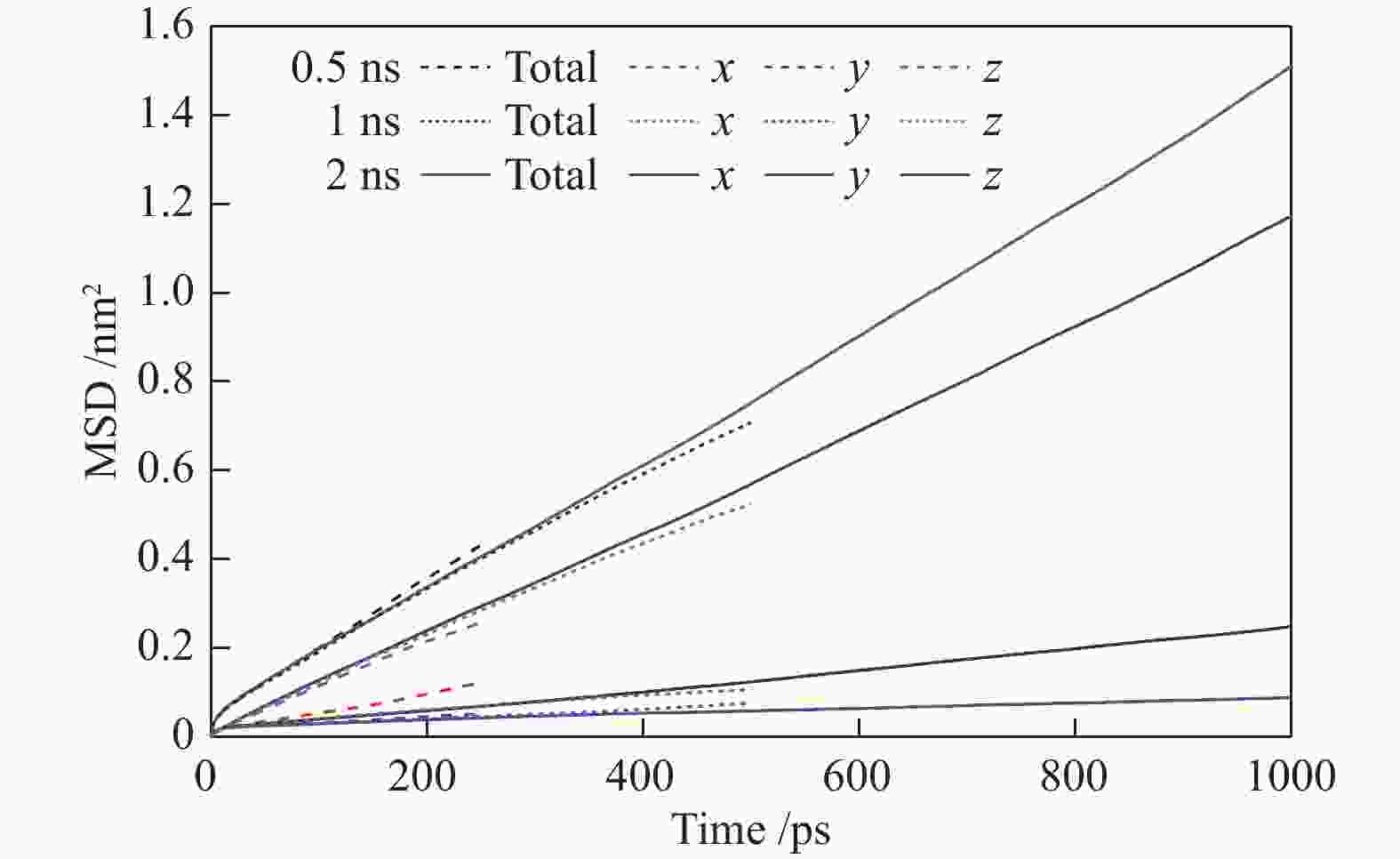
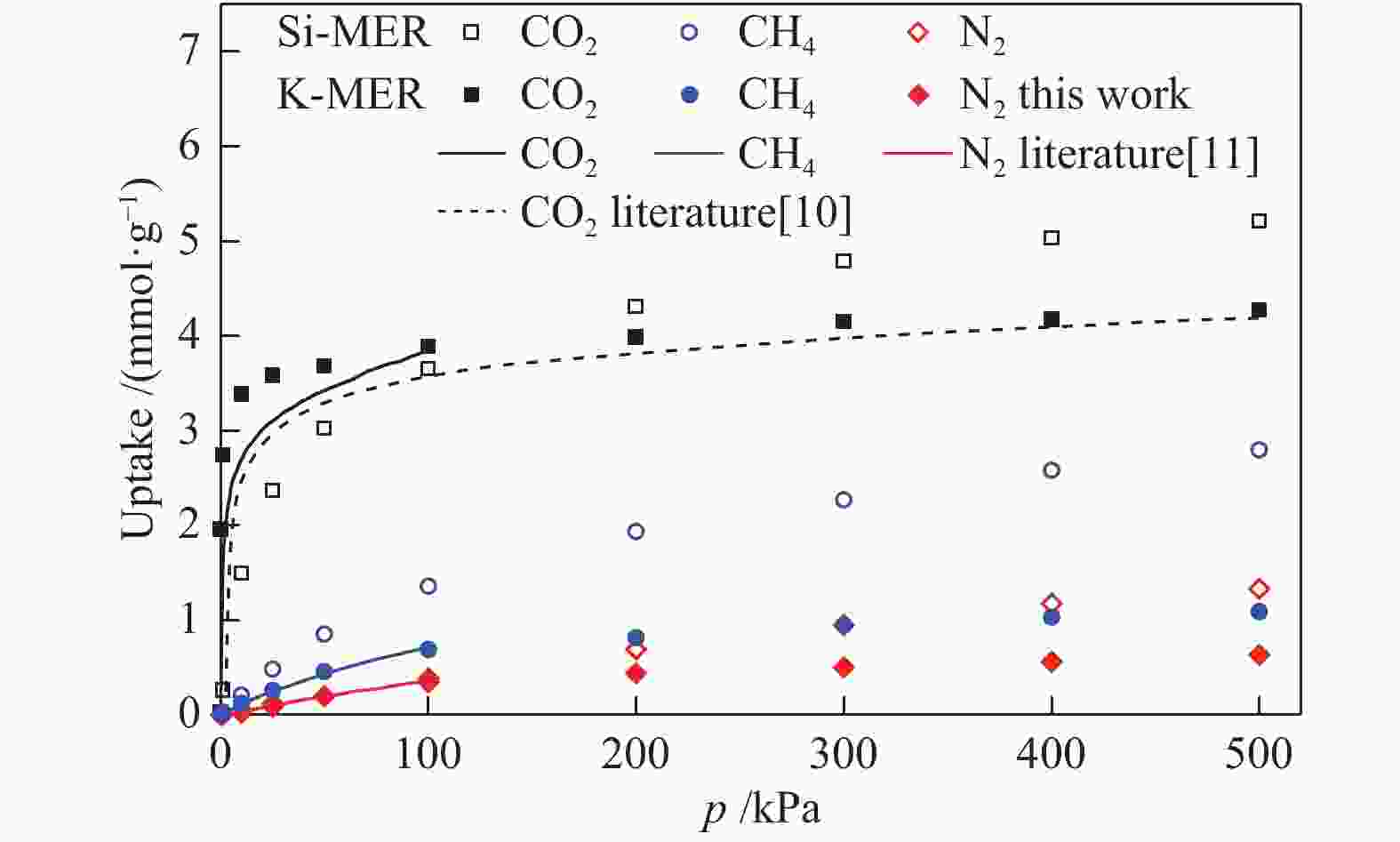
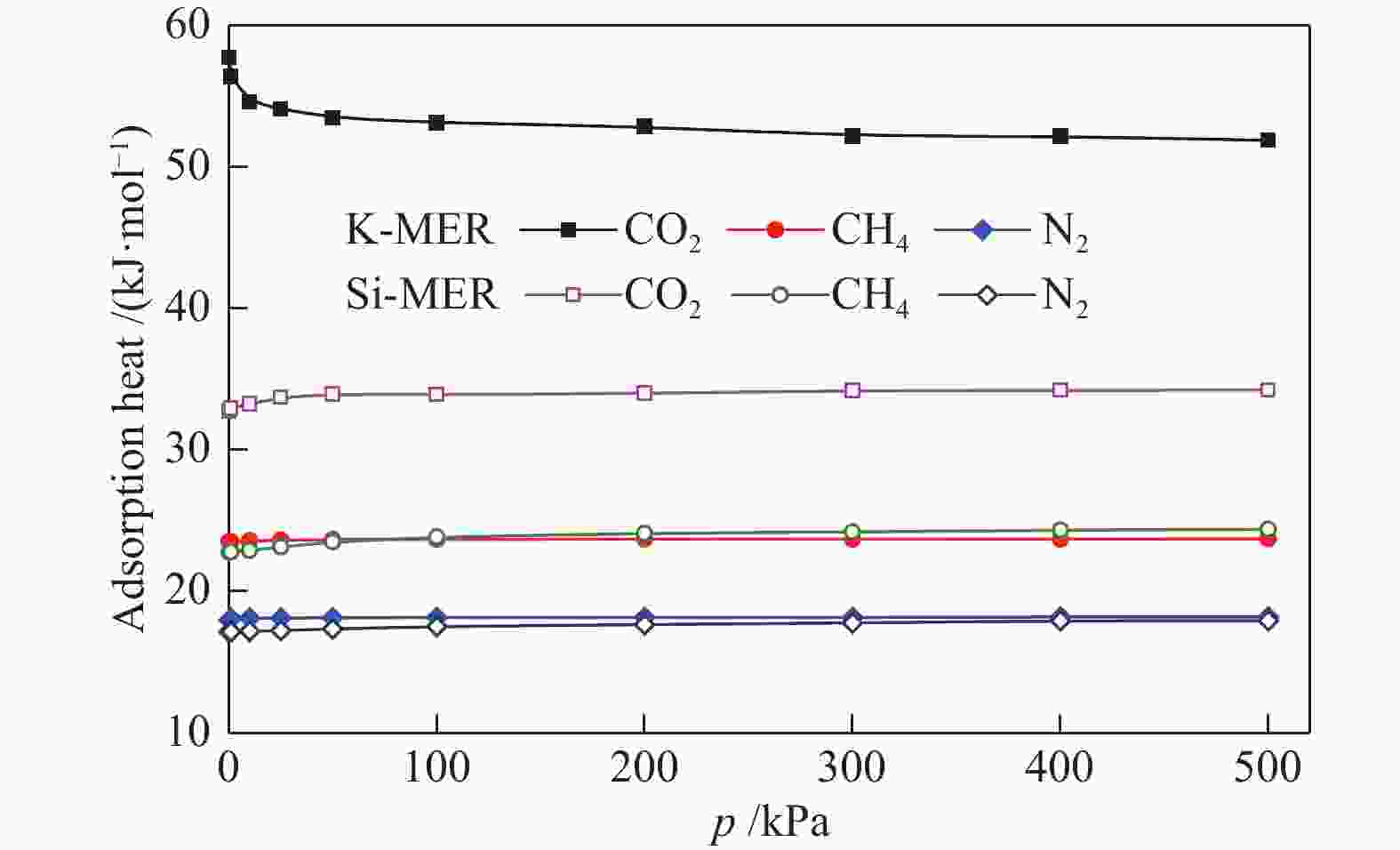
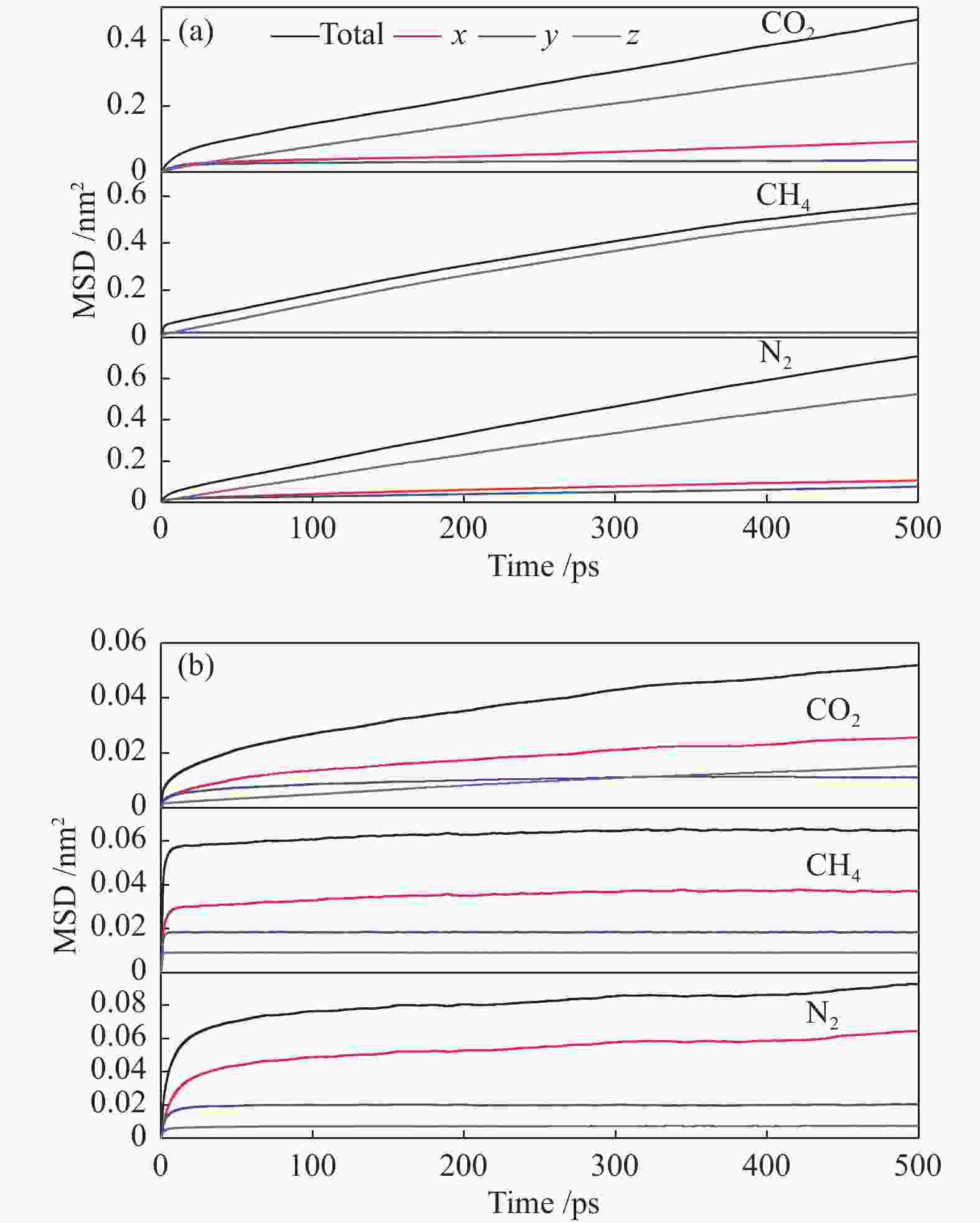
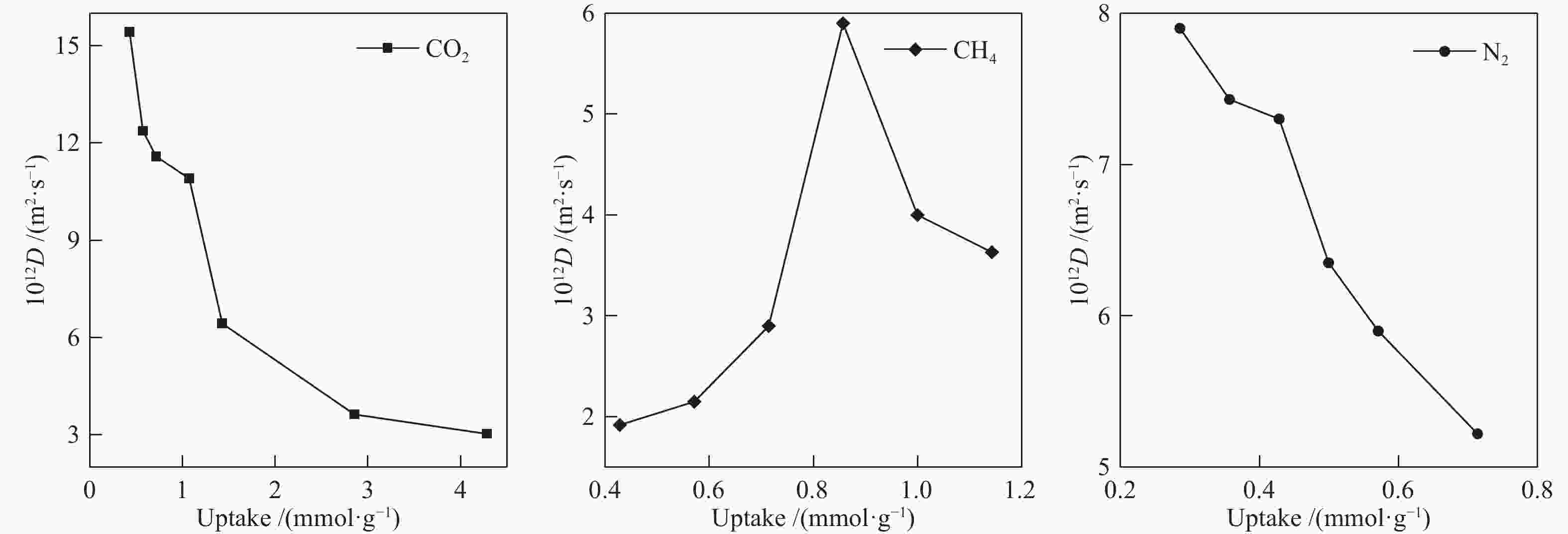
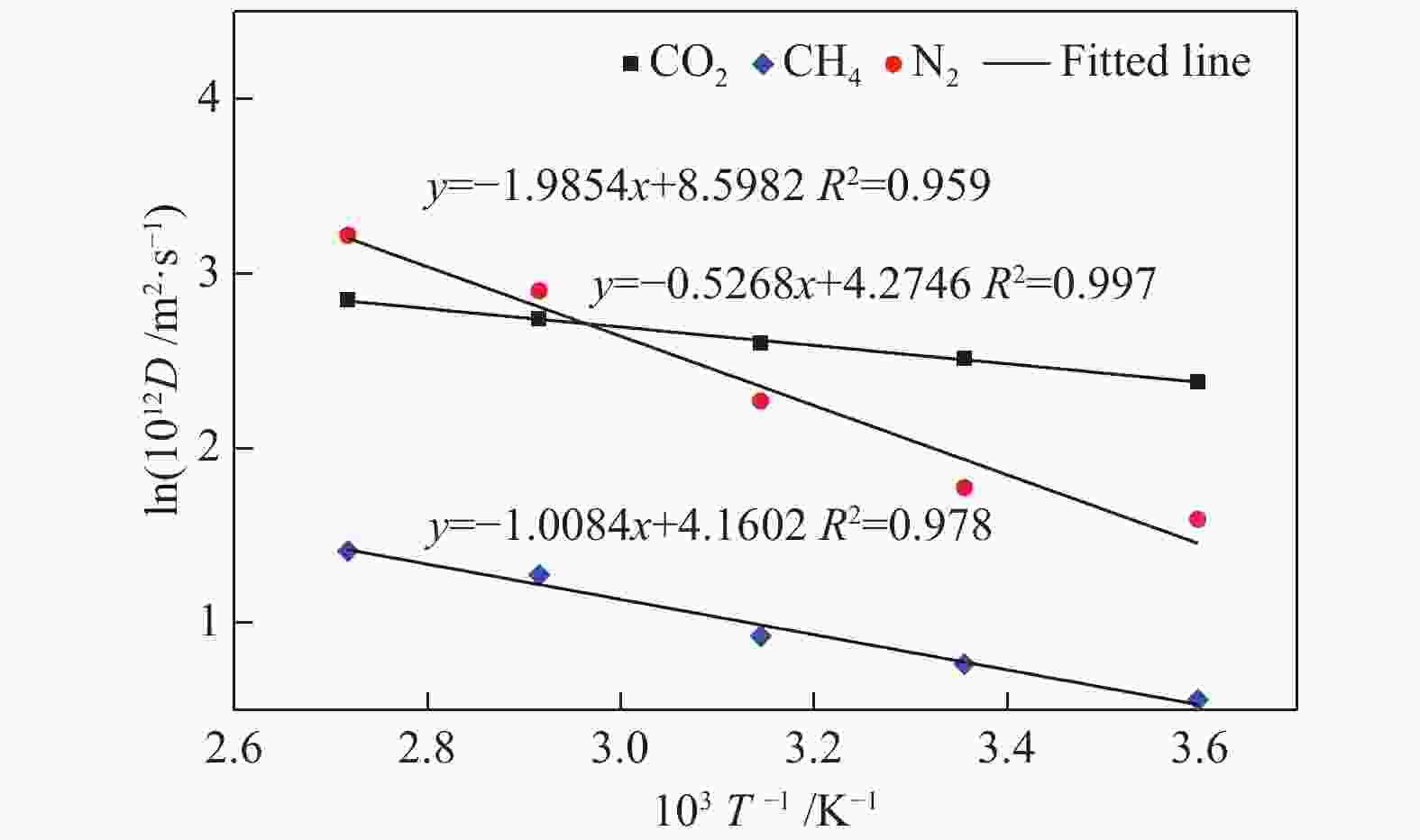
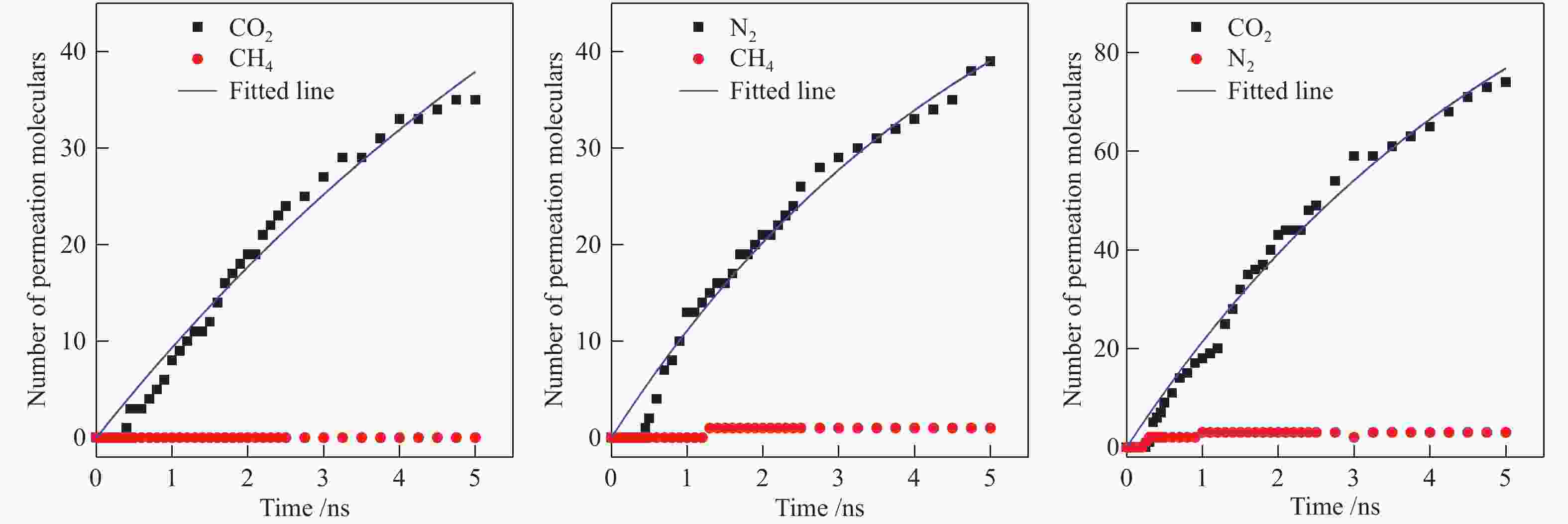
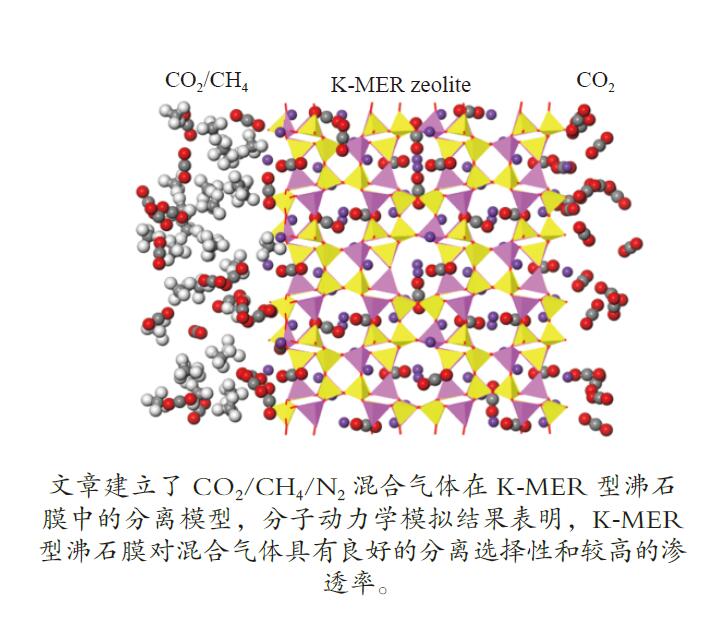
摘要: 采用巨正则蒙特卡洛(GCMC)模拟方法研究了CO2、CH4和N2在MER型沸石中的吸附性能,模拟结果与实验结果吻合证明模型和力场是可靠的。在此基础上,以纯硅MER型沸石作为对照,采用分子动力学(MD)模拟方法研究了CO2、CH4和N2在K-MER型沸石中的扩散和分离性能。结果表明,CO2、CH4和N2在MER型沸石中存在亚扩散现象,扩散方式为构型扩散,在沸石三维通道中的扩散存在各向异性。沸石与气体之间的作用力和沸石骨架外阳离子均影响气体分子的扩散能力,而沸石骨架外阳离子是影响气体分子扩散能力的主要因素。CO2和N2的自扩散系数随吸附浓度的增加而减小;CH4的自扩散系数随吸附浓度的增大先增加后减小。CO2、CH4和N2的自扩散系数随温度的升高均增加,扩散活化能大小顺序为N2 (16.51 kJ/mol) > CH4 (8.39 kJ/mol) > CO2 (4.38 kJ/mol)。K-MER型沸石膜对CO2/CH4、CO2/N2和N2/CH4分离体系均有良好的分离选择性。气体分子的渗透率~104 GPU(1 GPU = 3.35×10−10 mol/(s·m2·Pa))。Abstract: The adsorption behaviors of CO2, CH4 and N2 on MER zeolites were investigated by Grand Canonical Monte Carlo (GCMC) simulation method. The calculated pure gas uptake agreed well with the experimental data, which proved that the current simulation model and COMPASS force field are reliable. On this basis, molecular dynamics (MD) simulations were carried out for diffusion and separation of CO2, CH4 and N2 on K-MER zeolites, with the silicon MER zeolite as the reference. The results show that mean squared displacement (MSD) versus simulation time is sublinear. In MER zeolites, the configuration diffusion regime is dominant resulted from the tight fit of the gas molecules and the zeolite pore size. The diffusion of CO2, CH4 and N2 in MER zeolites with three-dimensional cage structures is anisotropic. The gas molecules diffuse preferentially along with the direction of x axis in K-MER zeolites. Extra-framework cations in zeolite plays important influence on the gas diffusion. In K-MER zeolites, the self-diffusion coefficients of CO2 and N2 are negative correlated with loading, whereas for the self-diffusion coefficient of CH4, it firstly increases and then decreases with the increase of loading. All of the self-diffusion coefficients of CO2, CH4 and N2 increase with the elevation of temperature. The order of diffusion activation energy is N2 (16.51 kJ/mol)﹥CH4 (8.39 kJ/mol)﹥CO2 (4.38 kJ/mol). K-MER zeolite membrane has good separation selectivity for gas mixture system of CO2/CH4, CO2/N2 and N2/CH4. The permeance of CO2 and N2 through K-MER zeolite membrane is as high as 104 GPU (1 GPU= 3.35×10−10 mol/(s·m2·Pa)).
-
Key words:
- carbon dioxide /
- MER zeolite /
- diffusion /
- separation /
- molecular dynamics simulation
-
表 1 CO2、CH4和N2在MER型沸石中的自扩散系数
Table 1 Self-diffusion coefficient of CO2, CH4 and N2 on MER zeolites at 298 K
Adsorbate DSi-MER/(10−12 m2·s−1) DK-MER/(10−12 m2·s−1) total x y z total x y z CO2 132.18 22.45 3.40 106.18 12.37 6.10 1.78 4.48 CH4 170.92 0 0 170.92 2.15 2.15 0 0 N2 218.18 28.28 18.70 171.18 5.90 5.75 0 0.10 -
[1] 鲁雪婷, 蒲彦锋, 李磊, 赵宁, 王峰, 肖福魁. 氨基修饰的金属有机框架Cu3(BTC)2的制备及其CO2吸附性能研究[J]. 燃料化学学报,2019,47(3):338−343. doi: 10.1016/S1872-5813(19)30016-7LU Xue-ting, PU Yan-feng, LI Lei, ZHAO Ning, WANG Feng, XIAO Fu-kui. Preparation of metal-organic frameworks Cu3(BTC)2 with amino-functionalization for CO2 adsorption[J]. J Fuel Chem Technol,2019,47(3):338−343. doi: 10.1016/S1872-5813(19)30016-7 [2] 贾晓霞, 杨江峰, 王勇, 李晋平. 纳米多孔石墨烯膜分离N2/CH4的密度泛函计算与分子动力学模拟[J]. 石油学报(石油加工),2018,34(6):1247−1254.JIA Xiao-xia, YANG Jiang-feng, WANG Yong, LI Jin-ping. Density functional theory and molecular dynamic simulation of N2/CH4 separation over nanoporous graphene membranes[J]. Acta Pet Sin (Pet Process Sect),2018,34(6):1247−1254. [3] WANG B, WU T Y, YU M, LI S G, ZHOU R F, XING W H. Highly ordered nanochannels in a nanosheet‐directed thin zeolite nanofilm for precise and fast CO2 separation[J]. Small,2020,16:2002836. doi: 10.1002/smll.202002836 [4] YUAN Y, WEI J W, GENG L L, MEI D J, LIAO L. An amine-bifunctionalization strategy with Beta/KIT-6 composite as a support for CO2 adsorbent preparation[J]. RSC Adv,2020,10:34187−34196. doi: 10.1039/D0RA05044J [5] BOYD P G, CHIDAMBARAM A, GARCÍA-DÍEZ E. Data-driven design of metal-organic frameworks for wet flue gas CO2 capture[J]. Nature,2019,576(7786):253−256. doi: 10.1038/s41586-019-1798-7 [6] 周智慧, 金灿, 张豪益, 梁晓蕾, 张富民, 肖强. CO2在三聚氰胺酚醛纤维上的吸附分离[J]. 燃料化学学报,2019,47(2):242−248. doi: 10.3969/j.issn.0253-2409.2019.02.014ZHOU Zhi-hui, JIN Can, ZHANG Hao-yi, LIANG Xiao-lei, ZHANG Fu-min, XIAO Qiang. CO2 adsorption and separation on phloroglucinol-melamine-formaldehyde polymeric nanofibers[J]. J Fuel Chem Technol,2019,47(2):242−248. doi: 10.3969/j.issn.0253-2409.2019.02.014 [7] 孔童童, 王霞, 郭庆杰. 新型多级微/介孔固态胺吸附剂的制备及其CO2吸附性能研究[J]. 燃料化学学报,2015,43(12):1489−1497. doi: 10.3969/j.issn.0253-2409.2015.12.013KONG Tong-tong, WANG Xia, GUO Qing-jie. Preparation and CO2 adsorption performance of a novel hierarchical micro/mesoporous solid amine sorbent[J]. J Fuel Chem Technol,2015,43(12):1489−1497. doi: 10.3969/j.issn.0253-2409.2015.12.013 [8] YEO Z Y, CHAI S P, ZHU P W, MAH S K, MOHAMED A R. Preparation of self-supported crystalline merlinoite type zeolite W membranes through vacuum filtration and crystallizations for CO2/CH4 separations[J]. New J Chem,2015,39(5):4135−4140. doi: 10.1039/C5NJ00008D [9] MIRFENDERESK S M. Synthesis and application of high-permeable zeolite MER membrane for separation of carbon dioxide from methane[J]. J Aust Ceram Soc,2019,55:103−114. doi: 10.1007/s41779-018-0216-1 [10] GEORGIEVA V M, BRUCE E L, VERBRAEKEN M C, SCOTT A R, CASTEEL W J, BRANDANI S, WRIGHT P A. Triggered gate opening and breathing effects during selective CO2 adsorption by merlinoite zeolite[J]. J Am Chem Soc,2019,141(32):12744−12759. doi: 10.1021/jacs.9b05539 [11] CHOI H J, JO D, MIN J G, HONG S B. The origin of selective adsorption of CO2 on merlinoite zeolites[J]. Angew Chem,2021,60:4307−4314. [12] 石勤, 席静, 张富民. MER型沸石吸附分离CO2/CH4的分子模拟[J]. 化工进展,2020,39(11):4408−4417.SHI Qin, XI Jing, ZHANG Fu-min. Molecular simulation of adsorption separation of CO2/CH4 by MER-type zeolites[J]. Chem Ind Eng Prog,2020,39(11):4408−4417. [13] 石勤, 王建强, 席静, 张富民. MER型沸石的结构、合成及应用进展[J]. 石油化工,2020,49(10):1004−1011. doi: 10.3969/j.issn.1000-8144.2020.10.013SHI Qin, WANG Jian-qiang, XI Jing, ZHANG Fu-min. Advances in structure, synthesis and application of MER zeolite[J]. Petrochem Technol,2020,49(10):1004−1011. doi: 10.3969/j.issn.1000-8144.2020.10.013 [14] 石勤, 黄雪莉. MER型沸石骨架和骨架外阳离子的分子模拟[J]. 离子交换与吸附,2016,32(1):38−46.SHI Qin, HUANG Xue-li. Molecular simulation of MER type zeolite framework and extra-framework cations[J]. Ion Exchange Adsorpt,2016,32(1):38−46. [15] YANG J, REN Y, TIAN A M. COMPASS force field for 14 inorganic molecules, He, Ne, Ar, Kr, Xe, H2, O2, N2, NO, CO, CO2, NO2, CS2, and SO2 in liquid phases[J]. J Phys Chem B,2000,104(20):4951−4957. doi: 10.1021/jp992913p [16] BEI L, BEREND S. Molecular simulation studies of separation of CO2/N2, CO2/CH4 and CH4/N2 by ZIFs[J]. J Phys Chem C,2010,114(18):8515−8522. doi: 10.1021/jp101531m [17] 肖永厚, 周梦雪, 赵颖, 王传明, 贺高红. 金属离子改性分子筛吸附水的 Monte Carlo 模拟[J]. 化工进展,2018,37(9):3430−3436.XIAO Yong-hou, ZHOU Meng-xue, ZHAO Ying, WANG Chuan-ming, HE Gao-hong. Monte Carlo simulation of water adsorption on metal ions modified molecular sieves[J]. Chem Ind Eng Prog,2018,37(9):3430−3436. [18] 沈文龙, 李嘉旭, 杨颖, 李平, 于建国. 基于沸石ZSM-5的CH4/N2/CO2二元体系吸附平衡[J]. 化工学报,2014,65(9):3490−3498. doi: 10.3969/j.issn.0438-1157.2014.09.025SHEN Wen-long, LI Jia-xu, YANG Ying, LI Ping, YU Jian-guo. Binary adsorption equilibrium of CH4, N2 and CO2 on zeolite ZSM-5[J]. CIESC J,2014,65(9):3490−3498. doi: 10.3969/j.issn.0438-1157.2014.09.025 [19] 韦小丽, 孙天军, 柯权力, 刘小伟, 王树东. 晶种法合成UZM-9分子筛及其CO2/CH4/N2吸附分离性能[J]. 燃料化学学报,2017,45(7):863−870. doi: 10.3969/j.issn.0253-2409.2017.07.011WEI Xiao-li, SUN Tian-jun, KE Quan-li, LIU Xiao-wei, WANG Shu-dong. Adsorptive separation properties of CO2/CH4/N2 on UZM-9 synthesized by seed-assisted method[J]. J Fuel Chem Technol,2017,45(7):863−870. doi: 10.3969/j.issn.0253-2409.2017.07.011 [20] RYO E, SAYAKA U, NORITAKA M. Highly selective sorption and separation of CO2 from a gas mixture of CO2 and CH4 at room temperature by a zeolitic organic–inorganic ionic crystal and investigation of the interaction with CO2[J]. J Phys Chem C,2012,116(30):16105−16110. doi: 10.1021/jp305890s [21] 郑安民. 分子筛催化理论计算—从基础到应用[M]. 北京: 科学出版社, 2020: 296−342.ZHENG An-ming. Theoretical Calculation of Zeolite Catalysis–from Foundation to Application[M]. Beijing: Science Press, 2020: 296−342. [22] 鲁相, 陈循, 汪亚顺, 谭源源, 高木子源. 气体在无定型聚异戊二烯中扩散的分子动力学模拟[J]. 物理化学学报,2016,32(10):2523−2530. doi: 10.3866/PKU.WHXB201606292LU Xiang, CHEN Xun, WANG Ya-shun, TAN Yuan-yuan, GAOMU Zi-yuan. Molecular dynamics simulation of gas transport in amorphous polyisoprene[J]. Acta Phys-Chim Sin,2016,32(10):2523−2530. doi: 10.3866/PKU.WHXB201606292 [23] YANG Z, LIU L, GUI T, ZHOU R F, CHEN X S. Mean residence time of CO2 molecules in flexible ZIF-8 cages explored by molecular dynamics simulations[J]. Chin J Chem Phys,2013,26(5):553−557. doi: 10.1063/1674-0068/26/05/553-557 [24] HARTMANN M, MACHOKEB A G, SCHWIEGER W. Catalytic test reactions for the evaluation of hierarchical zeolites[J]. Chem Soc Rev,2016,45:3313−3330. doi: 10.1039/C5CS00935A [25] BEERDSEN E, DUBBELDAM D, SMIT B. Loading dependence of the diffusion coefficient of methane in nanoporous materials[J]. J Phys Chem B,2006,110(45):22754−22772. doi: 10.1021/jp0641278 [26] LIU Z, YUAN J, BATEN J M, ZHOU J, TANG X, ZHAO C, CHEN W, YI X, KRISHNA R, SASTRE G, ZHENG A. Synergistically enhance confined diffusion by continuum intersecting channels in zeolites[J]. Sci Adv,2021,7(11):eabf0775. [27] XIAO J R, WEI J. Diffusion mechanism of hydrocarbons in zeolites–I. Theory[J]. Chem Eng Sci,1992,47:1123−1141. doi: 10.1016/0009-2509(92)80236-6 [28] 温伯尧, 孙成珍, 白博峰. 多孔石墨烯分离CH4/CO2的分子动力学模拟[J]. 物理化学学报,2015,31(2):261−267. doi: 10.3866/PKU.WHXB201411271WEN Bo-yao, SUN Cheng-zhen, BAI Bo-feng. Molecular dynamics simulation of the separation of CH4/CO2 by nanoporous graphene[J]. Acta Phys-Chim Sin,2015,31(2):261−267. doi: 10.3866/PKU.WHXB201411271 -




 下载:
下载: