Effect of sulfidation temperature on component transformation and catalytic performance of direct coal liquefaction catalyst
-
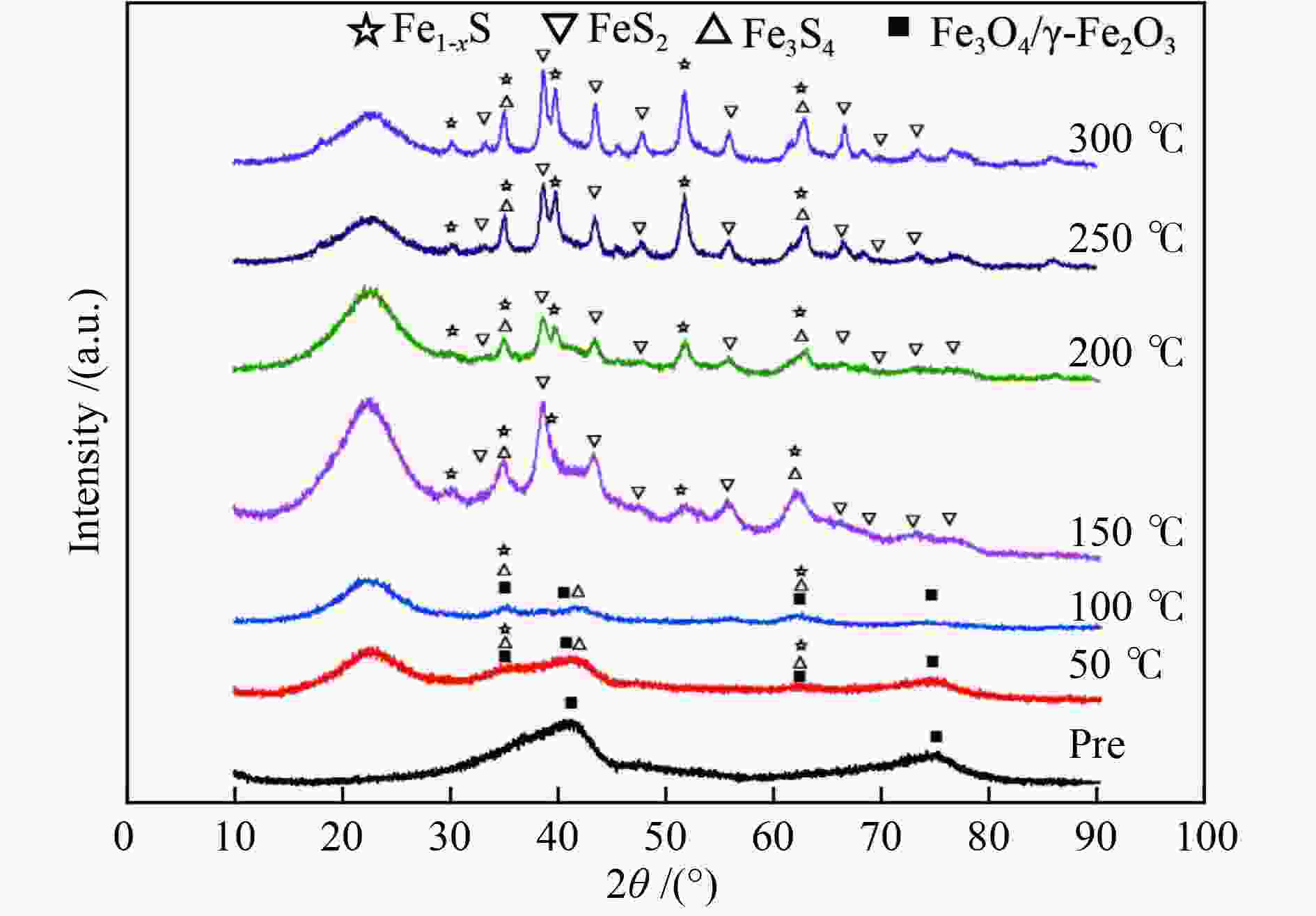
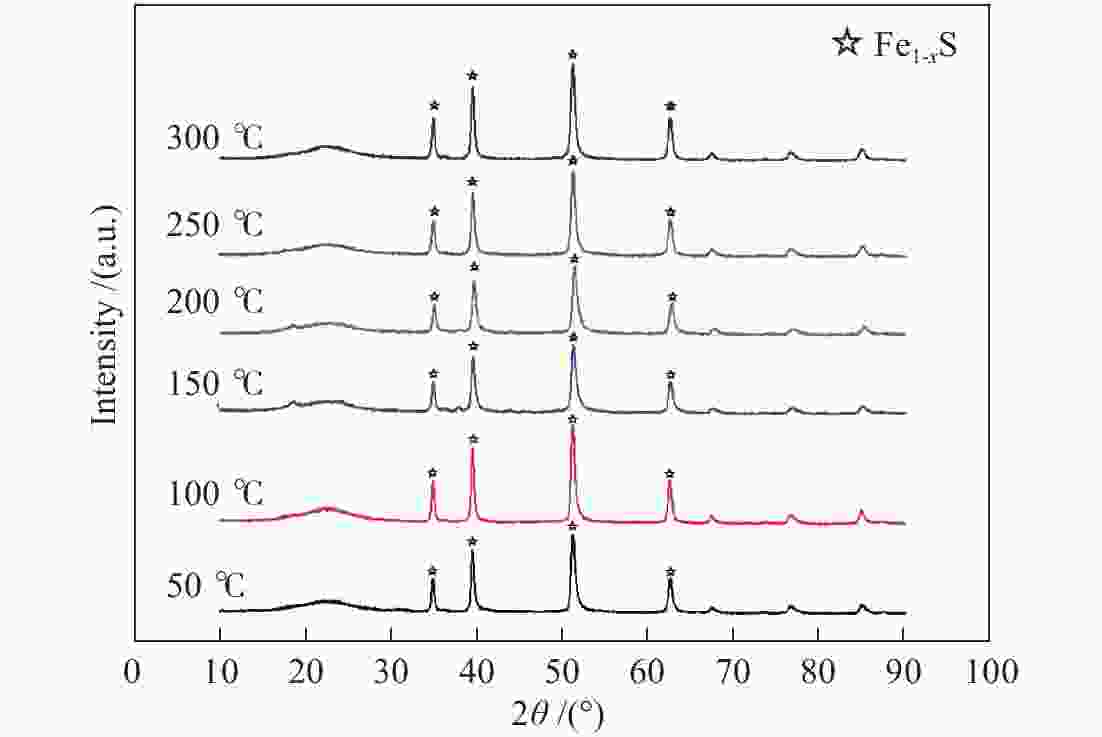
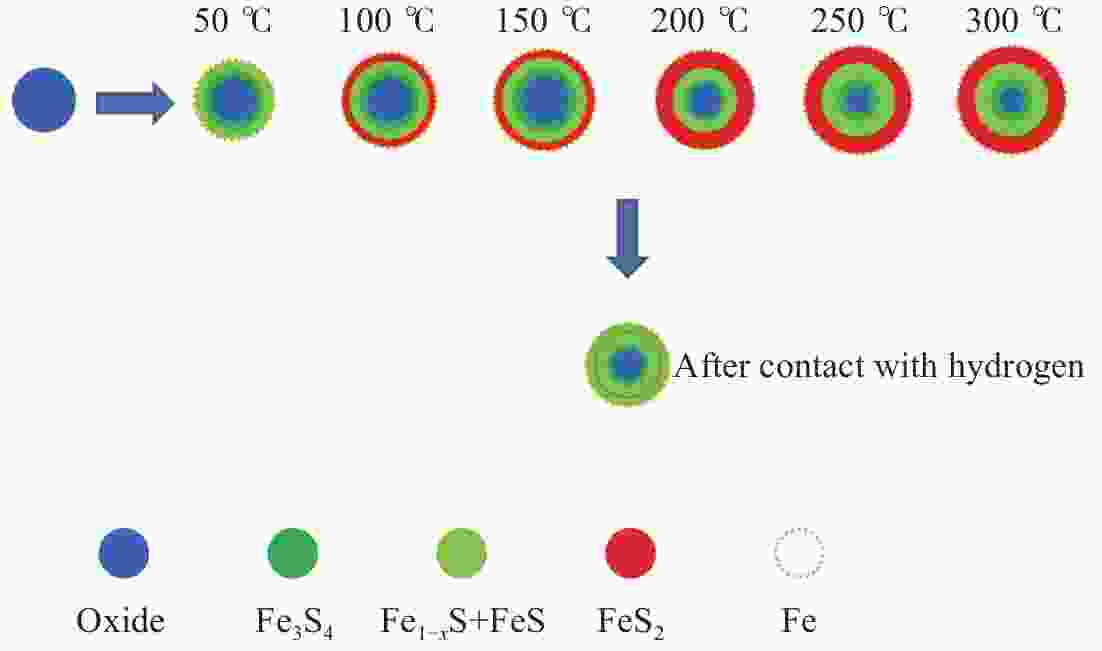
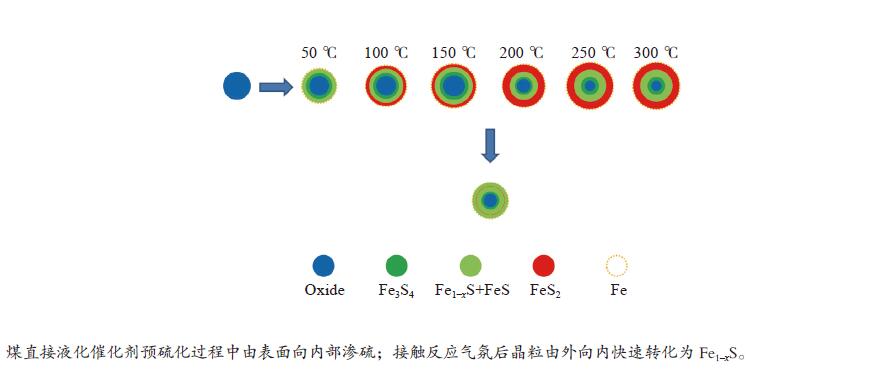
摘要: 在5% H2S/N2气氛,不同预硫化温度下制备了系列硫铁催化剂,并在5 MPa 的1% H2S/H2气氛、360 ℃下研究了其催化萘的加氢活性。借助MES、XRD和XPS等表征手段,探究了催化剂在不同预硫化温度及反应条件下组分转变规律。结果表明,预硫化过程是硫由表面向内部,依照FeS2→FeS、Fe1−xS→Fe3S4→Fe2O3顺序渗硫的过程,而升高温度有利于硫向体相内的传递;接触1% H2S/H2气氛后晶粒由外向内各组分均快速转化为Fe1–xS的过程;调控预硫化条件可以实现活性组分Fe1–xS的含量高、晶粒小,于是可获得最高活性。Abstract: A series of iron sulfide catalysts were prepared in 5% H2S/N2 atmosphere at different pre-sulfidation temperatures, and their catalytic activity for naphthalene hydrogenation was studied at 360 ℃ in 1% H2S/H2 atmosphere at 5 MPa. The component transformation of the catalyst under varied pre-sulfidation temperature and reaction conditions was researched using MES, XRD and XPS. The results revealed that the sulfidation process was in the form of sulfurizing from surface to interior in the order of FeS2→FeS, Fe1–
xS→Fe3S4→Fe2O3 during which transferring sulfur to the body phase was benefited from the rise of temperature. After contacting 1% H2S/H2 gas, the particles rapidly transformed to Fe1–xS from outside to the inside. By adjusting sulfidation conditions, the Fe1–xS content was high while the particle size was small, which made it possible to obtain the greatest activity of the catalyst. -
表 1 Mössbauer谱解谱参数及其来源
Table 1 Parameters of Mössbauer spectrum and data sources
Name Site IS/
(mm·s−1)QS/
(mm·s−1)H(T) Hexagonal
Pyrrhotite[21]1 0.74 0.01 33.01 2 0.72 0.02 30.44 3 0.71 0.02 28.18 4 0.74 0.01 25.63 Monoclinic
Pyrrhotite[22]1 0.85(1) −0.035(4) 33.4(1) 2 0.86(2) 0.084(10) 31.4(2) 3 0.81(1) −0.086(10) 27.1(1) 4a 0.83(1) 0.084(15) 24.4(2) 4b 0.82(1) 0.166(15) 20.7(2) Greigite[23] octahedral 0.66 − 32.7 tetrahedral 0.38 − 31.9 Troilite[24] − 0.89 −0.14 32.8 Pyrite[25] − 0.43(1) 0.66(1) − Maghemite[26] x 0.47 −0.01 51.0 y 0.34 −0.03 48.1 Magnetite[26] A 0.42 0.06 51.6 B 0.99 0.89 51.0 Highly dispersed
Fe[27]− 0.02 −0.03 25.0 α-Fe[28] − 0.09 0.04 33.8 表 2 不同预硫化温度(5% H2S/N2)所得催化剂的Mössbauer谱解析
Table 2 Mössbauer spectrum results of catalysts obtained at different sulfidation temperature (5% H2S/N2)
Sample Fe3O4 Fe2O3 Fe3S4 Fe1–xS FeS FeS2 Fe Fe2+(spm) Fe3+(spm) Pre 20.3 79.7 − − − − − − − Cat-50 17.9 45.5 9.2 15.0 3.8 0 5.3 1.4 1.8 Cat-100 10.8 44.7 7.9 22.1 0 9.3 5.3 − − Cat-150 12.8 49.8 8.3 14.1 0.9 12.2 2.5 − − Cat-200 6.2 29.9 10.5 28.2 3.0 21.4 0.9 − − Cat-250 6.7 20.5 8.3 37.0 4.9 20.7 1.9 − − Cat-300 6.2 20.1 12.8 33.2 4.5 23.2 0 − − 表 3 不同预硫化温度(5% H2S/N2)硫化1 h后XPS所得Fe 2p和S 2p参数
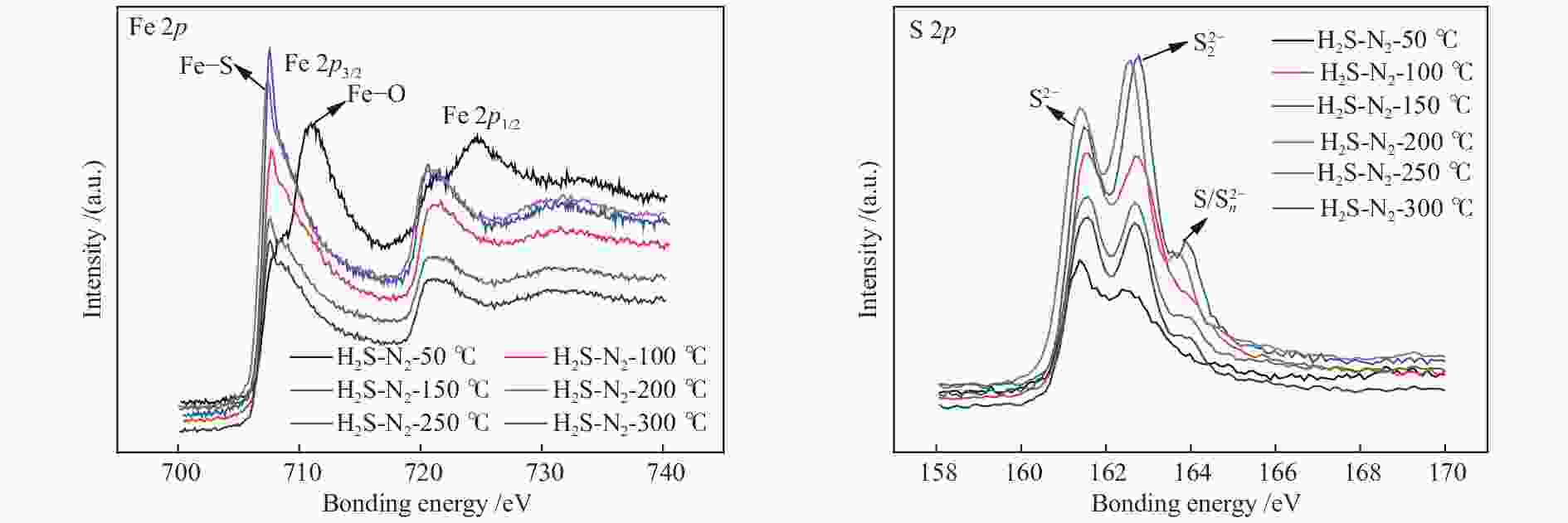
Table 3 Contents of Fe 2p and S 2p obtained by XPS after sulfidation at different temperatures at 5% H2S/N2 for 1 h
Sample Fe Fe2+–S Fe3+–S Fe2+–O Fe3+–O S2− $ {\rm{S}}^{2-}_2 $ $ {\rm{S}}/{\rm{S}}^{2-}_{{{n}}} $ Cat-50 4.4 29.2 9.7 12.3 44.4 75.6 24.4 − Cat-100 5.0 48.4 20.6 7.0 19.0 61.3 30.8 7.9 Cat-150 3.3 52.6 18.4 15.6 10.1 53.9 41.1 5.0 Cat-200 6.5 48.4 23.1 7.3 14.7 58.2 36.8 5.0 Cat-250 8.7 39.9 19.5 16.0 15.8 61.1 30.8 8.1 Cat-300 7.6 43.0 21.7 10.6 17.1 62.2 28.3 9.5 表 4 不同预处理温度(5% H2S/N2)所得催化剂经过反应条件(1% H2S/H2)后Fe1−xS的晶粒尺寸及Fe1−xS中1−x值
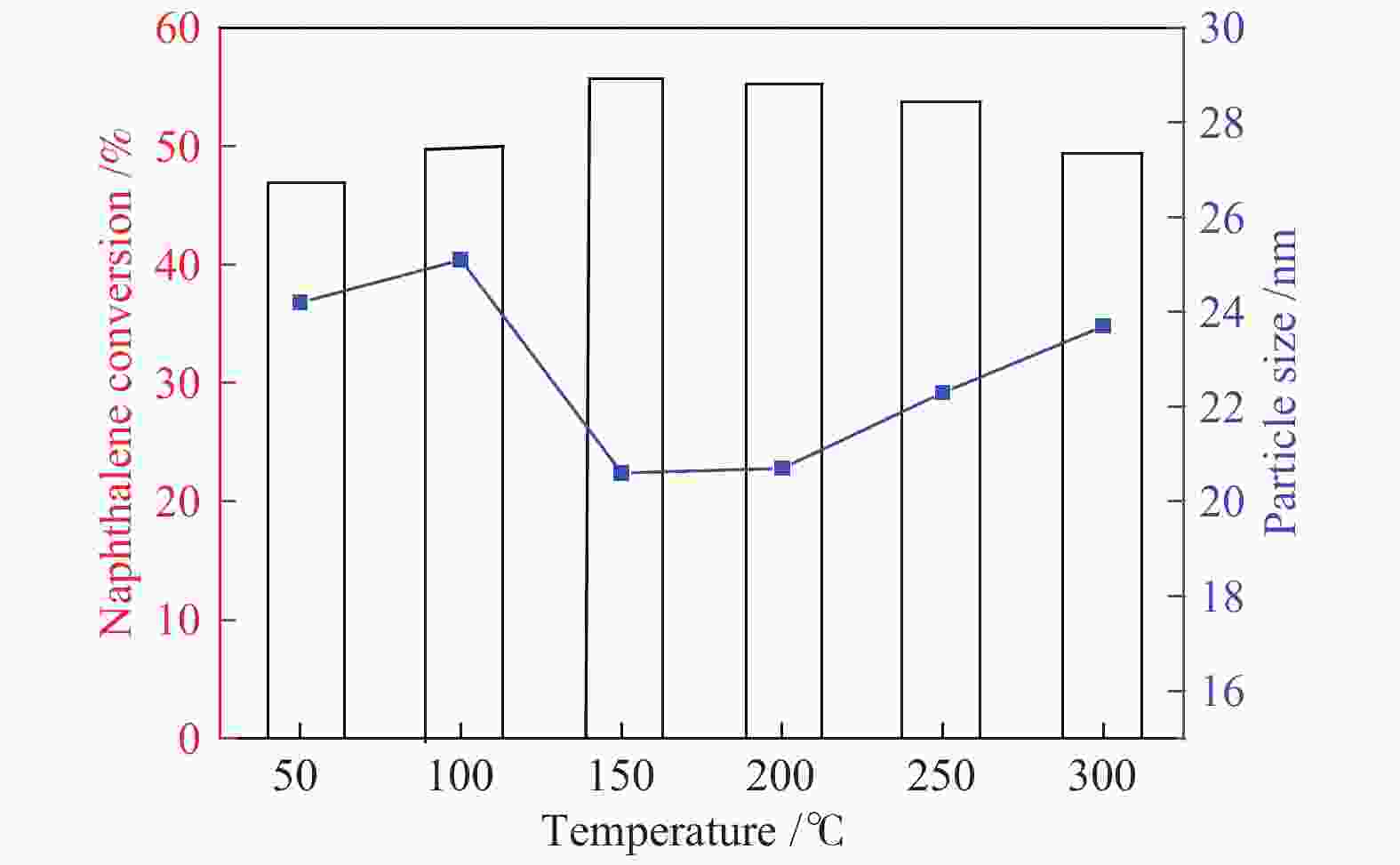
Table 4 Particle size of Fe1–xS and the 1–x value of Fe1–xS for the catalysts prepared at different pretreatment temperature (5% H2S/N2) after reaction conditions (1% H2S/H2)
Sample Particle size of Fe1–xS/nm 1–x value of Fe1–xS Cat-50-360 24.2 0.91 Cat-100-360 25.1 0.91 Cat-150-360 20.6 0.90 Cat-200-360 20.7 0.88 Cat-250-360 22.3 0.90 Cat-300-360 23.7 0.91 表 5 不同预处理温度(5% H2S/N2)所得催化剂经过反应条件(1% H2S/H2)后Mössbauer谱所得不同物质占比
Table 5 Mössbauer spectra for the samples prepared at different pretreatment temperature (5% H2S/N2) and reaction conditions (1% H2S/H2)
Sample Fe3O4 Fe2O3 Fe3S4 Fe1–xS FeS FeS2 Fe Cat-50-360 8.2 39.8 6.7 25.3 0.7 11.2 8.2 Cat-100-360 0 3.3 0 64.1 20.6 6.2 5.8 Cat-150-360 1.6 0.8 3.2 61.8 19.3 5.6 7.8 Cat-200-360 0 0.4 1.9 70.5 15.5 4.9 6.9 Cat-250-360 0 0.5 0 69.8 18.1 5.2 6.4 Cat-300-360 0.2 1.3 2.7 70.6 14.8 5.0 5.5 -
[1] GUO M, XU Y. Coal-to-liquids projects in China under water and carbon constraints[J]. Energy Policy,2018,117:58−65. doi: 10.1016/j.enpol.2018.02.038 [2] LIU Z, GUAN D B, WEI W, DAVIS S J, CIAIS P, BAI J, PENG S S, ZHANG Q, HUBACEK K, MARLAND G, ANDRES R J, CRAWFORD-BROWN D, LIN J T, ZHAO H Y, HONG C P, BODEN T A, FENG K S, PETERS G P, XI F M, LIU J G, LI Y, ZHAO Y, ZENG N, HE K B. Reduced carbon emission estimates from fossil fuel combustion and cement production in China[J]. Nature,2015,524(7565):335−338. doi: 10.1038/nature14677 [3] SHUI H, CAI Z, XU C. Recent advances in direct coal liquefaction[J]. Energies,2010,3(2):155−170. doi: 10.3390/en3020155 [4] XIE J, LU H, SHU G, LI K, ZHANG X, WANG H, YUE W, GAO S, CHEN Y. The relationship between the microstructures and catalytic behaviors of iron-oxygen precursors during direct coal liquefaction[J]. Chin J Catal,2018,39(4):857−866. doi: 10.1016/S1872-2067(17)62919-X [5] HIRANO K, KOUZU M, OKADA T, KOBAYASHI M, IKENAGA N, SUZUKI T. Catalytic activity of iron compounds for coal liquefaction[J]. Fuel,1999,78(15):1867−1873. doi: 10.1016/S0016-2361(99)00095-2 [6] IKENAGA N-O, UEDA C, MATSUI T, OHTSUKI M, SUZUKI T. Co-liquefaction of micro algae with coal using coal liquefaction catalysts[J]. Energy Fuels,2001,15(2):350−355. doi: 10.1021/ef000129u [7] MONTANO P A, VAISHNAVA P P, KING J A, EISENTROUT E N. Mössbäuer study of decomposition of pyrite in hydrogen[J]. Fuel,1981,60(8):712−716. doi: 10.1016/0016-2361(81)90224-6 [8] IKENAGA N-O, TANIGUCHI H, WATANABE A, SUZUKI T. Sulfiding behavior of iron based coal liquefaction catalyst[J]. Fuel,2000,79(3):273−283. [9] HUFFMAN G P, GANGULY B, ZHAO J, RAO K R P M, SHAH N, FENG Z, HUGGINS F E, TAGHIEI M M, LU F. Structure and dispersion of iron-based catalysts for direct coal liquefaction[J]. Energy Fuels,1993,7(2):285−296. doi: 10.1021/ef00038a020 [10] KANEKO T, TAZAWA K, KOYAMA T, SATOU K, SHIMASAKI K, KAGEYAMA Y. Transformation of iron catalyst to the active phase in coal liquefaction[J]. Energy Fuels,1998,12(5):897−904. doi: 10.1021/ef9702310 [11] KANEKO T, SUGITA S, TAMURA M, SHIMASAKI K, MAKINO E, SILALAHI L H. Highly active limonite catalysts for direct coal liquefaction[J]. Fuel,2002,81(11/12):1541−1549. doi: 10.1016/S0016-2361(02)00079-0 [12] 郭贵贵. 煤直接液化油加氢改质组合催化剂硫化过程分析[J]. 神华科技,2013,11(6):68−72. doi: 10.3969/j.issn.1674-8492.2013.06.022GUO Gui-gui. Analysis of catalyst vulcanization of coal direct liquefaction to oil hydrogenation modification combination[J]. Energy Sci Technol,2013,11(6):68−72. doi: 10.3969/j.issn.1674-8492.2013.06.022 [13] 王仲义, 闫作杰, 单敏, 陈平平, 童健. 器外预硫化加氢裂化催化剂开工技术应用总结[J]. 炼油技术与工程,2021,51(1):10−12+32. doi: 10.3969/j.issn.1002-106X.2021.01.003WANG Zhong-yi, YAN Zuo-jie, SHAN Min, CHEN Ping-ping, TONG Jian. Application summary of start-up technology of ex-situ presulfiding hydrocracking catalyst[J]. Pet Refin Eng,2021,51(1):10−12+32. doi: 10.3969/j.issn.1002-106X.2021.01.003 [14] 孙欣欣, 袁铭遥, 吴雪晴. 硫化铵器外预硫化选择性加氢催化剂研究[J]. 炼油技术与工程,2020,50(12):42−45. doi: 10.3969/j.issn.1002-106X.2020.12.012SUN Xin-xin, YUAN Ming-yao, WU Xue-qing. Study on ex-situ presulfurization of selective hydrogenation catalyst[J]. Pet Refin Eng,2020,50(12):42−45. doi: 10.3969/j.issn.1002-106X.2020.12.012 [15] 陶帅江. 加氢催化剂预硫化技术进展[J]. 鞍钢技术,2020,(4):9−12. doi: 10.3969/j.issn.1006-4613.2020.04.002TAO Shuai-jiang. Progress in presulfurization technology for hydrotreating catalyst[J]. Angang Technol,2020,(4):9−12. doi: 10.3969/j.issn.1006-4613.2020.04.002 [16] 张黎, 范文青, 肖文灿, 徐琳, 刘长坤. 加氢催化剂预硫化技术探讨[J]. 广东化工,2020,47(12):126−127. doi: 10.3969/j.issn.1007-1865.2020.12.053ZHANG Li, FAN Wen-qing, XIAO Wen-can, XU Lin, LIU Chang-kun. Study on the technology of pre-sulfurization for hydrogenated catalyst[J]. Guangdong Chem Ind,2020,47(12):126−127. doi: 10.3969/j.issn.1007-1865.2020.12.053 [17] 金吉海, 刘丽芝, 宋君辉, 焦祖凯, 严金龙, 甄涛, 张铎. 器外预硫化催化剂加氢脱酸性能研究[J]. 无机盐工业,2020,52(9):100−104.JIN Ji-hai, LIU Li-zhi, SONG Jun-hui, JIAO Zu-kai, YAN Jin-long, ZHEN Tao, ZHANG Duo. Study on hydrodeacidification performance of off-site pre-sulfidation catalysts[J]. Inorg Chem Ind,2020,52(9):100−104. [18] DE WIND M, HEINERMAN J J L, LEE S L, PLANTENGA F L. Air quality and economics spur use of presulfided catalysts[J]. Oil Gas J,1992,90(8):49−53. [19] DJEGA-MARIADASSOU G, BESSON M, BRODZKI D, CHARCOSSET H, TRAN VINH H, VARLOUD J. Evolution of highly dispersed catalysts during hydroliquefaction of coal[J]. Fuel Process Technol,1986,12:143−153. doi: 10.1016/0378-3820(86)90072-X [20] LAMBERT J M, SIMKOVICH G, WALKER P L. Production of pyrrhotites by pyrite reduction[J]. Fuel,1980,59(10):687−690. doi: 10.1016/0016-2361(80)90019-8 [21] KONDORO J W A. Mossbauer study of vacancies in natural pyrrhotite[J]. J Alloys Compd,1999,289(1/2):36−41. doi: 10.1016/S0925-8388(99)00170-X [22] JEANDEY C, ODDOU J L, MATTEI J L, FILLION G. Mössbauer investigation of the pyrrhotite at low temperature[J]. Solid State Commun,1991,78(3):195−198. doi: 10.1016/0038-1098(91)90282-Z [23] CHANG L, ROBERTS A P, TANG Y, RAINFORD B D, MUXWORTHY A R, CHEN Q W. Fundamental magnetic parameters from pure synthetic greigite (Fe3S4)[J]. J Geophys Res-Sol Ea,2008,113(B6):1−16. [24] CUDA J, KOHOUT T, TUCEK J, FILIP J, MALINA O, KRIZEK M, ZBORIL R. Mossbauer Spectroscopy in Materials Science[M]. New York: AIP Publishing, 2014: 8−11. [25] MONTANO P A, SEEHRA M S. Magnetism of iron pyrite (FeS2) - a Mossbauer study in an external magnetic-field[J]. Solid State Commun,1976,20(9):897−898. doi: 10.1016/0038-1098(76)91300-4 [26] OH S J, COOK D C, TOWNSEND H E. Characterization of iron oxides commonly formed as corrosion products on steel[J]. Hyperfine Interact,1998,112(1/4):59−65. [27] KOBZI B, WATANABE Y, AKIYAMA K, KUZMANN E, HOMONNAY Z, KREHULA S, RISTIĆ M, NISHIDA T, KUBUKI S. 57Fe-Mössbauer study and methylene blue decomposing effect of nanoparticle mixtures composed of metallic iron and maghemite[J]. J Alloys Compd,2017,722:94−100. doi: 10.1016/j.jallcom.2017.06.083 [28] KUBONO I, NISHIDA N, KOBAYASHI Y, YAMADA Y. Mossbauer spectra of iron (III) sulfide particles[J]. Hyperfine Interact,2017,238:1−10. doi: 10.1007/s10751-016-1375-5 [29] ZHAO R, YANG L, SONG X, ZHANG W, WANG B, HUANG S, WU S, WU Y. Effects of sulfur additive on the transformation behaviors of γ-Fe2O3 and coal liquefaction performances under mild conditions[J]. Asia-Pac J Chem Eng,2018,13(4):1−9. [30] ZUO W B, PELENOVICH V, LI Q D, ZENG X M, FU D J. Study on velocity mode of Fe-57 Mossbauer spectroscopy and determination of lattice dynamics in Fe3S4[J]. Results Phys,2019,12:1214−1217. doi: 10.1016/j.rinp.2019.01.010 [31] SKINNER B J, GRIMALDI F S, ERD R C. Greigite thio-spinel of iron-new mineral[J]. Am Mineral,1964,49(5/6):543−555. [32] SADEGH-VAZIRI R, BABLER M U. Removal of hydrogen sulfide with metal oxides in packed bed reactors-a review from a modeling perspective with practical implications[J]. Appl Sci-Basel,2019,9(24):1−24. [33] MORRISH R, SILVERSTEIN R, WOLDEN C A. Synthesis of stoichiometric FeS2 through plasma-assisted sulfurization of Fe2O3 nanorods[J]. J Am Chem Soc,2012,134(43):17854−17857. doi: 10.1021/ja307412e [34] YU L, LANY S, KYKYNESHI R, JIERATUM V, RAVICHANDRAN R, PELATT B, ALTSCHUL E, PLATT H A S, WAGER J F, KESZLER D A, ZUNGER A. Iron chalcogenide photovoltaic absorbers[J]. Adv Energy Mater,2011,1(5):748−753. doi: 10.1002/aenm.201100351 [35] JAGADEESH M S, SEEHRA M S. Thermomagnetic studies of conversion of pyrite and marcasite in different atmospheres (vacuum, H2, He And CO)[J]. J Phys D Appl Phys,1981,14(11):2153−2167. doi: 10.1088/0022-3727/14/11/023 [36] HONG Y, FEGLEY B. The kinetics and mechanism of pyrite thermal decomposition[J]. Ber Bunsen-Ges Phys,1997,101(12):1870−1881. doi: 10.1002/bbpc.19971011212 -




 下载:
下载: