-
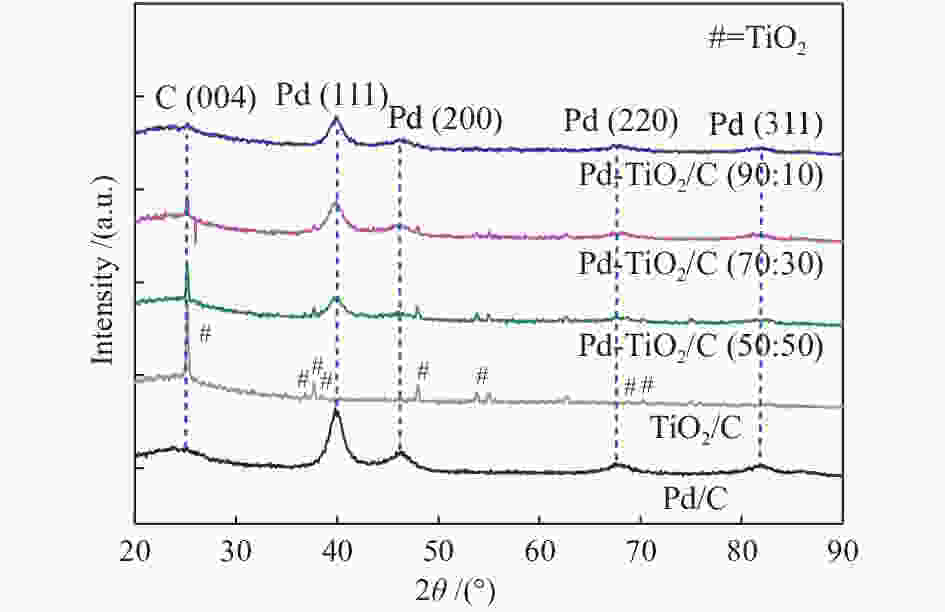
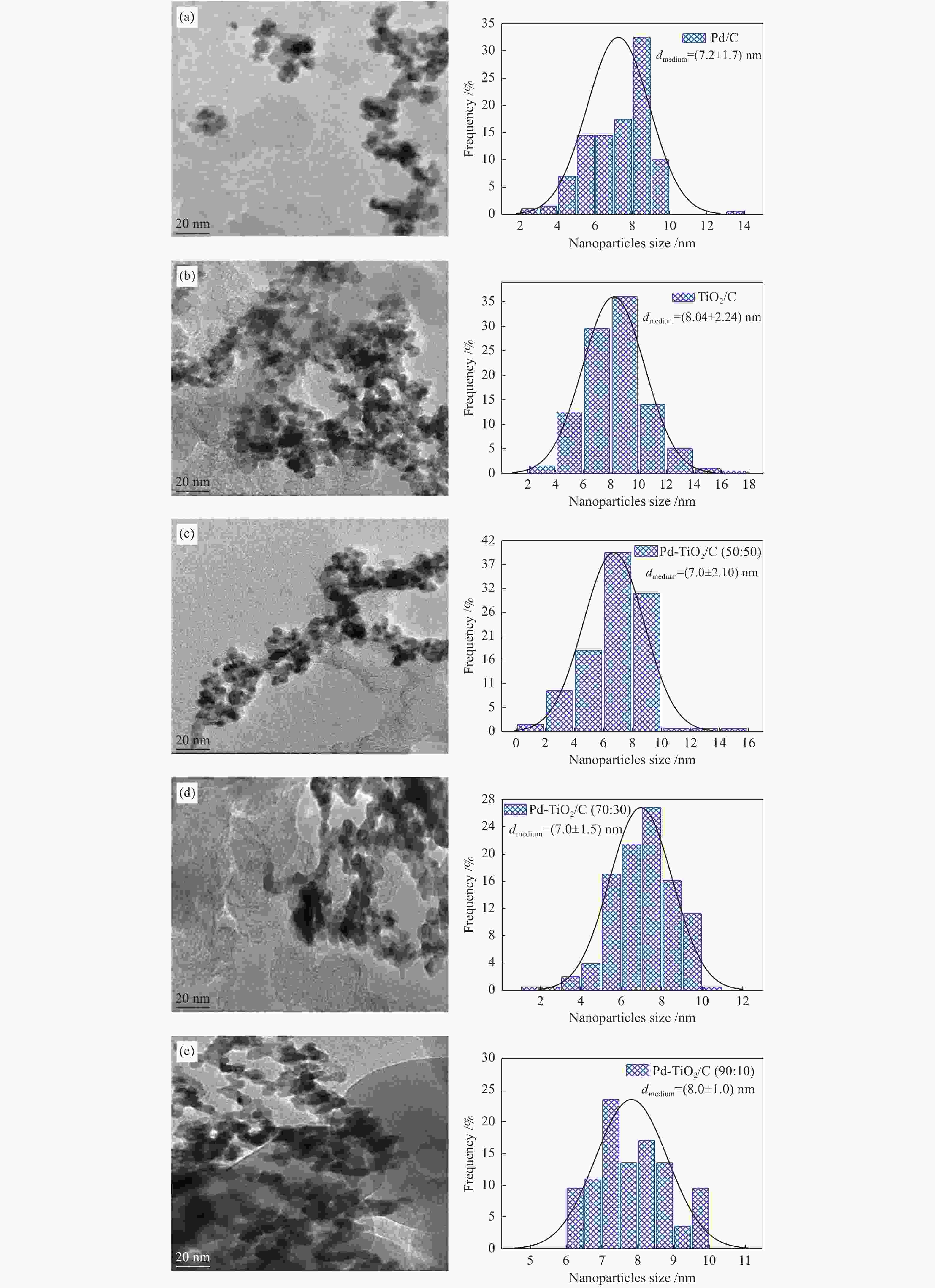
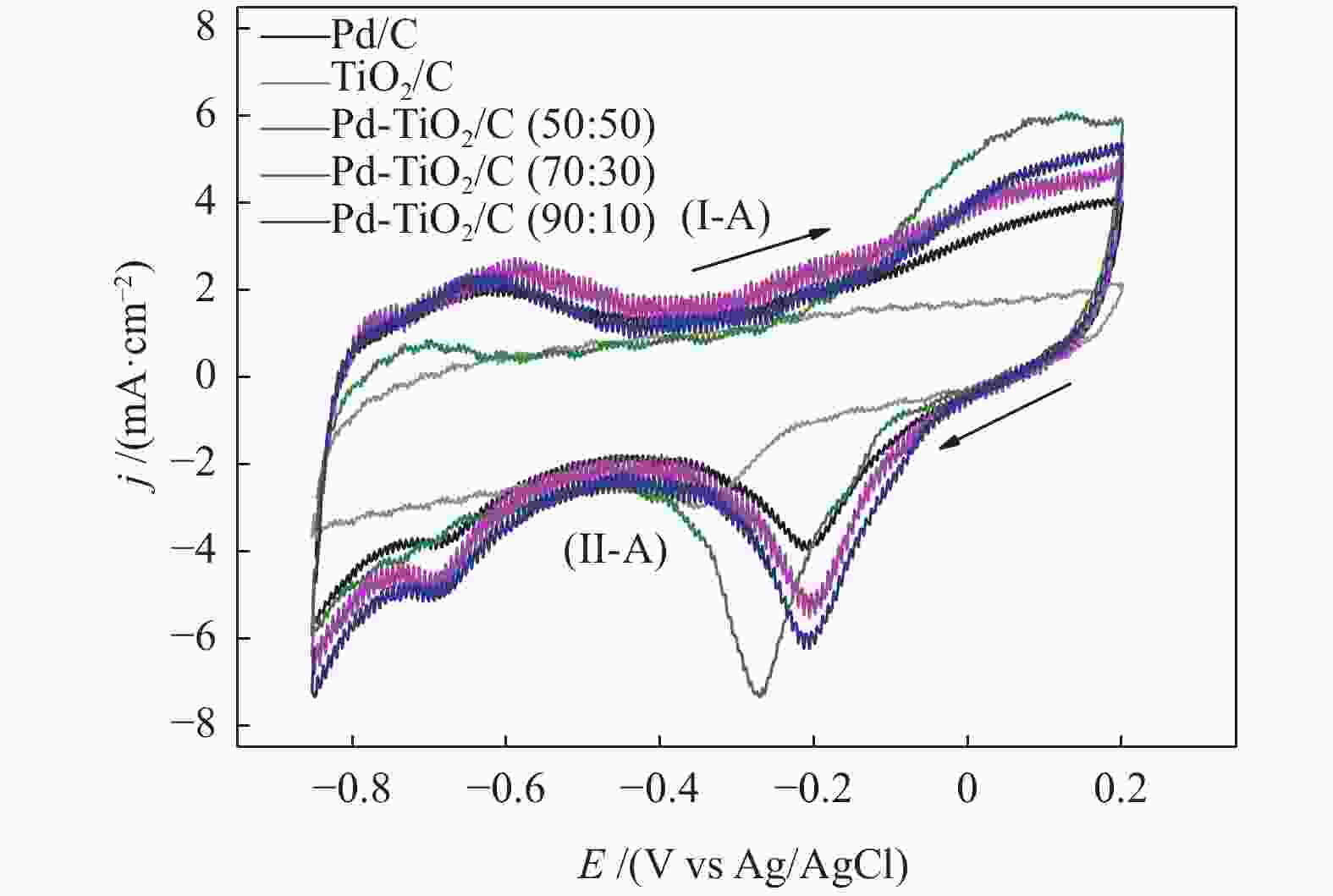
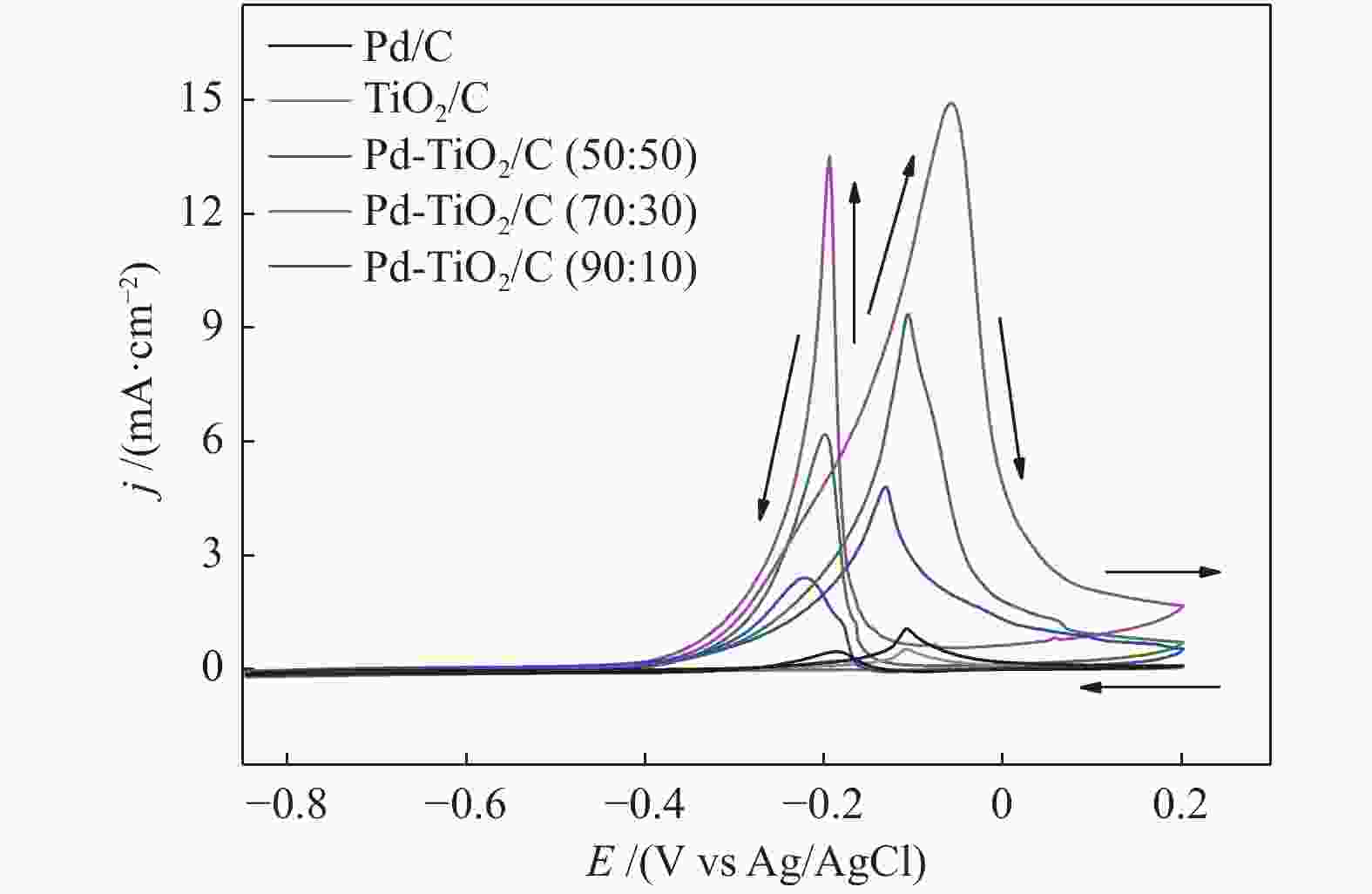
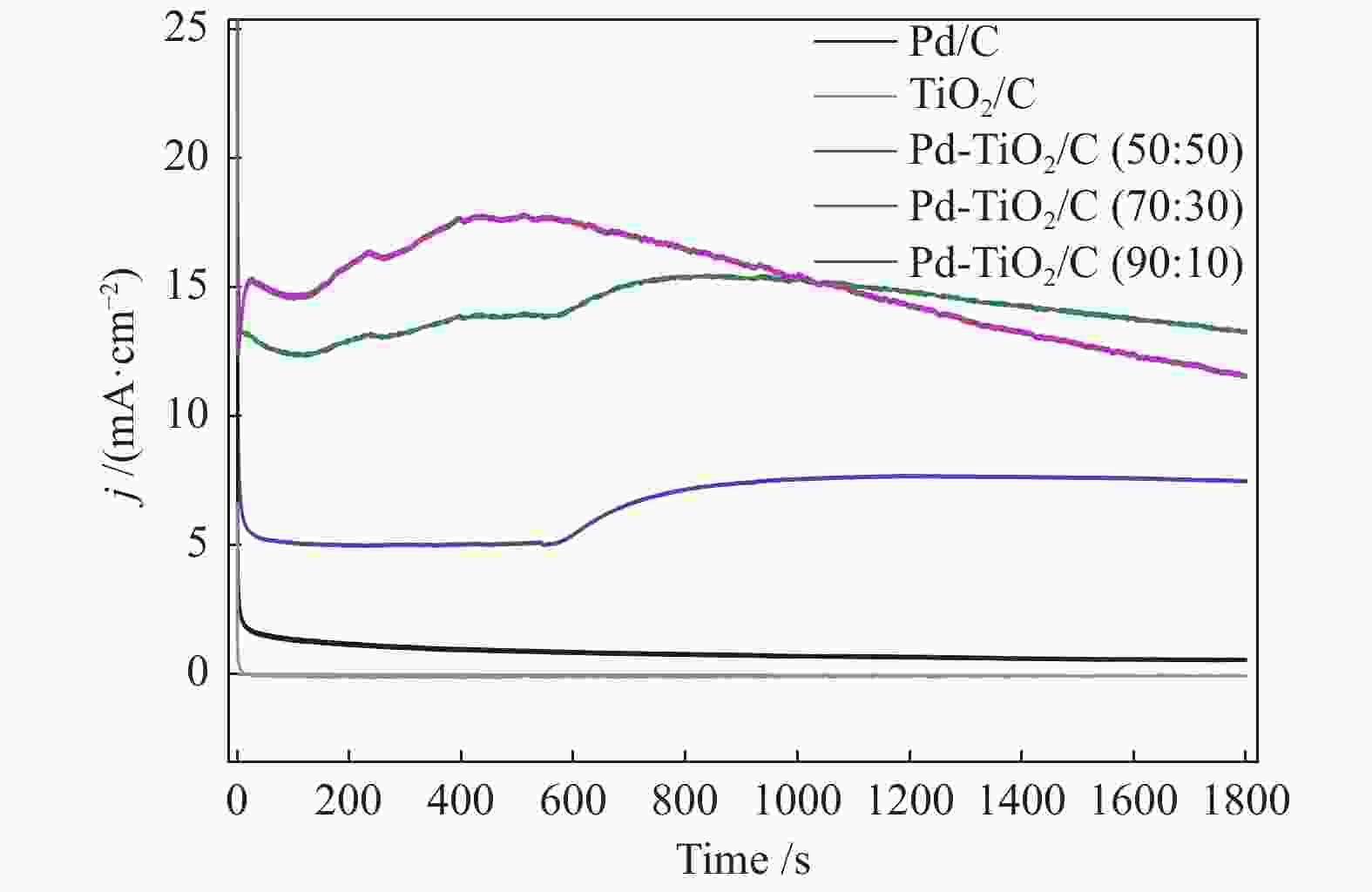
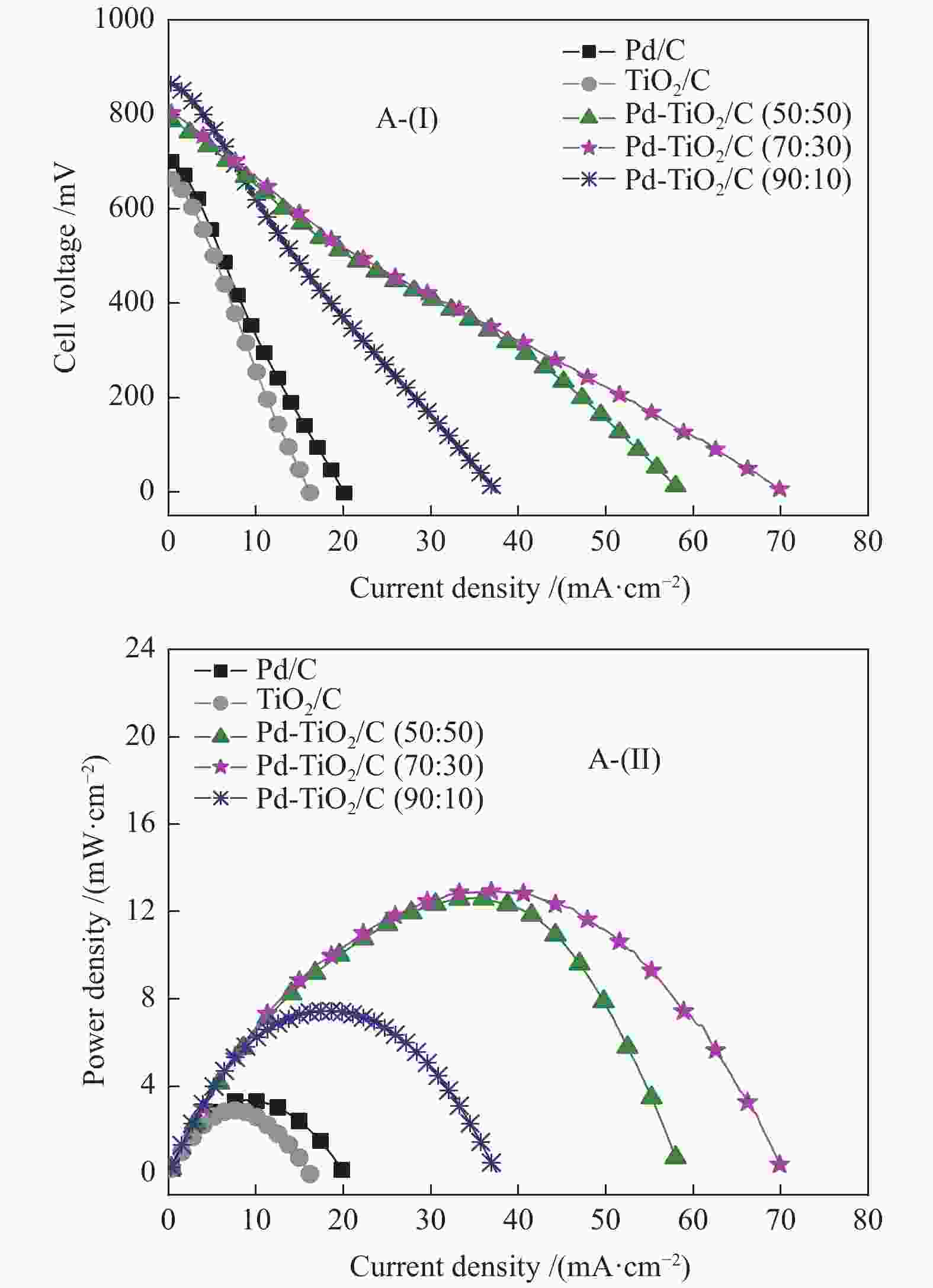
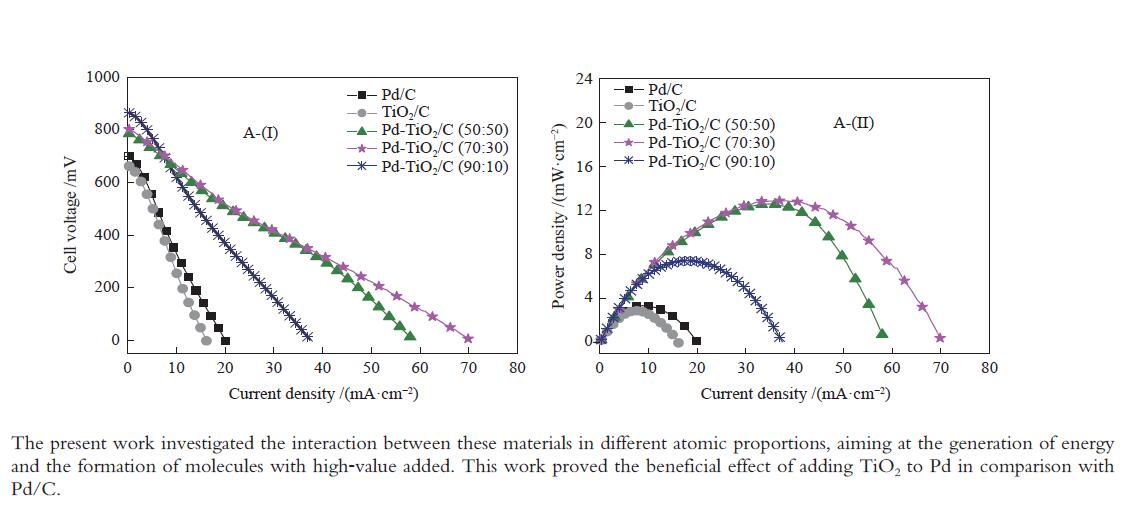
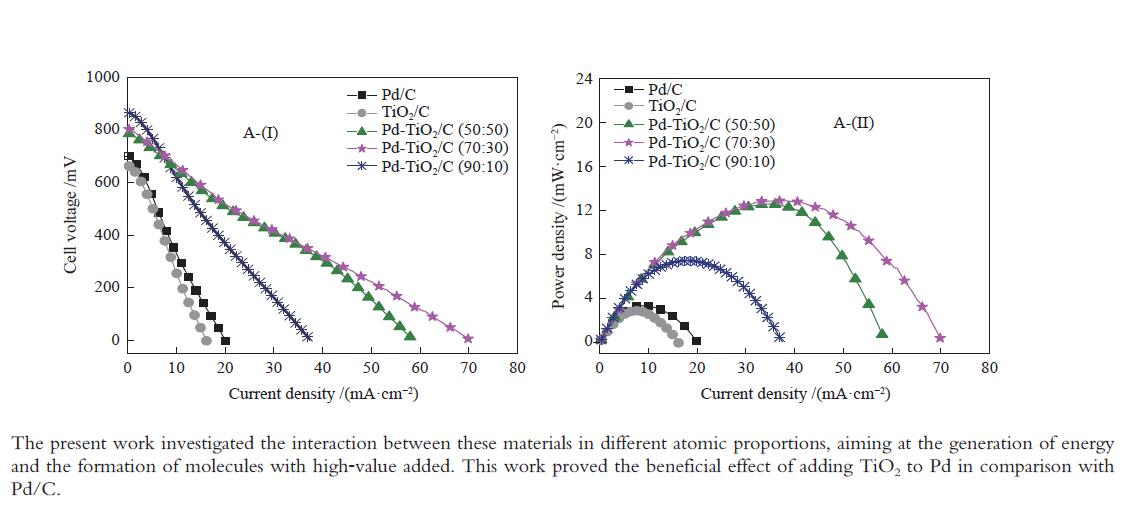
摘要: 以Vulcan XC-72炭为载体,采用硼氢化钠还原法制备了Pd-TiO2/C电催化剂用于直接丙三醇燃料电池阳极材料,并用能量色散谱(EDX)、X射线衍射(XRD)、透射电子显微镜(TEM)、循环伏安法(CV)、计时电流法(I-t)和全反射-傅里叶变换红外光谱(ATR-FTIR)对催化剂进行了表征。EDX结果表明,Pd-TiO2/C中Pd和Ti含量接近名义原子比。Pd-TiO2的X射线衍射结果表明,Pd具有面心立方fcc结构,同时TiO2展现出四方结构的锐钛矿相的峰特征。TEM图像显示Pd和TiO2纳米颗粒在碳载体中分布均匀并存在一些簇状区域,纳米颗粒大小为7.0 − 8.0 nm。循环伏安显示在1 mol/L KOH电解液中催化剂Pd-TiO2/C在 约 −0.7 V vs Ag/AgCl有明显的析氢行为和电容增加现象,表明催化剂在TiO2修饰后活性明显增加。通过循环伏安对碱性体系的丙三醇电催化氧化表明,Pd-TiO2/C(70∶30)显示了非常好的催化活性:起始氧化电位也变得更负达到−0.4V vs Ag/AgCl,氧化电流达到14.91 mA/cm,优于Pd-TiO2/C(90∶10)的9.37 mA/cm和Pd-TiO2/C(50∶50) 的4.88 mA/cm,明显高于Pd/C和TiO2/C的1.88 和0.55 mA/cm。I-t实验表明1000 s之前Pd-TiO2/C展现出最高的电流密度,但在1000 s后电流密度有一定程度的下降,表明催化剂的催化活性很高但长期稳定性方面需要进一步提升。在碱性直接丙三醇燃料电池中进行的实验表明,所制备的Pd-TiO2/C(50∶50)、Pd-TiO2/C(70∶30)和Pd-TiO2/C(90∶10)对甘油的电化学氧化性能均优于Pd/C电催化剂,其中,Pd-TiO2/C(70∶30)的催化活性最好:开路电压达到817 mV,最大功率密度达到12.5 mW/cm,高于Pd-TiO2/C(50∶50)的12.4 mW/cm、Pd-TiO2/C(90∶10)的7.9 mW/cm、Pd/C的3.7 mW/cm以及 TiO2/C的2.25 mW/cm,这表明,TiO2对Pd/C催化剂有很好的促进作用,这种协同效应可以归因为TiO2对Pd的电子效应和双功能机理,同时Pd和TiO2比例为70∶30时,其催化性能达到最优。ATR-FTIR结果表明,反应后所有Pd-TiO2/C电催化剂表面均有甘油醛、羟丙酮酸、甲酸等高附加值产物生成,为直接丙三醇燃料电池电-化学品联产提供一定的依据。
-
关键词:
- 丙三醇氧化 /
- Pd-TiO2 电催化剂 /
- 原位ATR-FTIR /
- 循环伏安 /
- 碱性燃料电池
Abstract: The Pd-TiO2 electrocatalysts were synthesized via sodium borohydride reduction and characterized by X-ray diffraction (XRD), transmission electron microscopy (TEM), cyclic voltammetry, chronoamperometry and attenuated total reflectance-Fourier transform infrared (ATR-FTIR). The X-ray diffraction experiments of the Pd-TiO2 showed peaks associated with Pd face-centered cubic (fcc) structure and peaks characteristics of TiO2 (anatase phase) with a tetragonal structure. The TEM images showed that the Pd and TiO2 nanoparticles were well distributed in the carbon support showing some clustered regions with nanoparticle sizes between 7 and 8 nm. Cyclic voltammograms showed an increase in current density values after the glycerol adsorption process. Experiments in alkaline direct glycerol fuel cells at 60 °C showed a higher power density for Pd-TiO2/C (70∶30) in comparison to the commercial Pd/C electrocatalyst indicating that the use of the TiO2 co-catalyst with Pd nanoparticles had a beneficial behavior. This effect can be attributed to the electronic effect or to the bifunctional mechanism. Molecules with high-value added glyceraldehyde, hydroxypyruvate and formate were identified as electrochemical reaction products of glycerol on all prepared electrocatalysts. -
Figure 6 Polarization (A-(I)) curves and power density (A-(II)) in a 5 cm2 alkaline direct glycerol fuel cell (ADGFC) at 60 °C, using Pd/C, TiO2/C and Pd-TiO2/C electrocatalysts with different atomic proportions fed with 2.0 mol/L glycerol in a 2.0 mol/L KOH solution and oxygen flux was set to 150 mL min at 85 °C
Table 1 Nominal atomic ratios and atomic ratios obtained by EDX of the Pd-TiO2/C (50∶50, 70∶30 and 90∶10) electrocatalysts synthesized by the sodium borohydride reduction method
Electrocatalyst Nominal atomic ratios (%)
(Pd:TiO2)Atomic ratios EDX (%)
(Pd:TiO2)Pd-TiO2/C 50:50 45:55 Pd-TiO2/C 70:30 68:33 Pd-TiO2/C 90:10 89:11 Table 2 Molecules formation in the partial electro-oxidation reaction of glycerol at different potentials using combined Pd/C and TiO2/C electrocatalysts
Pd TiO2 50% 70% 90% Formate 0.0→0.69 (0.59↑) 0.21 → 0.51 0.34 → 0.74 (0.52↓) 0.0 to 0.70 Hydroxypyruvate 0.0→0.69 (0.59↑) 0.21 → 0.51 0.34 → 0.74
(0.34↓)(0.52↓) 0.0 to 0.70 Glycerate 0.0→0.69 (0.59↑ and 0.69↑) 0.21 → 0.51 0.34 → 0.74
(0.34↓ and 0.44↓)0.0 → 0.62
(0.52↓)0.0 to 0.70 Carbonate 0.0→0.69 (0.59↑ and 0.69↑) 0.21 → 0.51 0.34 → 0.74 (0.34↓ and 0.44↓) 0.0 → 0.62 (0.52↓) 0.0 to 0.70 Carboxylate 0.0→0.69 (0.59↑ and 0.69↑) N.O. 0.54 → 0.74 (0.74↑) (0.62↑) N.O. Symbol used: → : increase to; ↓: less intense; ↑: more intense; N.O.: not observed -
[1] ONG C B, KAMARUDIN K S, BASRI S. Direct liquid fuel cells: A review[J]. Int J Hydrogen Energy,2017,42(15):10142−10457. [2] ANTOLINI E, GONZALEZ R E. Alkaline direct alcohol fuel cells[J]. J Power Sources,2010,195(11):3431−3450. doi: 10.1016/j.jpowsour.2009.11.145 [3] BANJONG J, THERDTHIANWONG A, THERDTHIANWONG S, YONGPRAGAT S, WONGYAO N. High performance alkaline-acid direct glycerol fuel cells for portable power supplies via electrode structure design[J]. Int J Hydrogen Energy,2020,45(3):2244−2256. doi: 10.1016/j.ijhydene.2019.11.041 [4] YAHYA N, KAMARUDIN S K, KARIM A N, MASDAR S M, LOH S K, LIM L K. Durability and performance of direct glycerol fuel cell with palladium-aurum/vapor grown carbon nanofiber support[J]. Energy Convers Manag,2019,188:120−130. doi: 10.1016/j.enconman.2019.02.087 [5] GERALDES N A, DA SILVA F D, SILVA A G L, SPINACÉ V E, NETO O A, DOS SANTOS C M. Binary and ternary palladium based electrocatalysts for alkaline direct glycerol fuel cell[J]. J Power Sources,2015,293:823−830. doi: 10.1016/j.jpowsour.2015.06.010 [6] KIM M, LEE C, KO M S, NAM M-J. Metal alloy hybrid nanoparticles with enhanced catalytic activities in fuel cell applications[J]. J Solid State Chem,2019,270:295−303. doi: 10.1016/j.jssc.2018.11.014 [7] SANTOS C B J, VIEIRA C, CRISAFULLI R, LINARES J J. Promotional effect of auxiliary metals Bi on Pt, Pd, and Ag on Au, for glycerol electrolysis[J]. Int J Hydrogen. Energy,2020,45(47):25658−25671. doi: 10.1016/j.ijhydene.2019.11.225 [8] NANDENHA J, FONTES H E, PIASENTIN M R, FONSECA C F, NETO O A. Direct oxidation of methane at low temperature using Pt/C, Pd/C, Pt/C-ATO and Pd/C-ATO electrocatalysts prepared by sodium borohydride reduction process[J]. J Fuel Chem Technol,2018,46(9):1137−1145. doi: 10.1016/S1872-5813(18)30046-X [9] DASH S, MUNICHANDRAIAH N. Nanoflowers of PdRu on PEDOT for electrooxidation of glycerol and its analysis[J]. Electrochim Acta,2015,180:339−352. doi: 10.1016/j.electacta.2015.07.020 [10] HOUACHE E S M, SHUBAIR A, SANDOVAL G M, SAFARI R, BOTTON A G, JASEN V P, GONZÁLEZ A E, BARANOVA A E. Influence of Pd and Au on electrochemical valorization of glycerol over Ni-rich surfaces[J]. J Catal,2021,396:1−13. doi: 10.1016/j.jcat.2021.02.008 [11] VILLA A, DIMITRATOS N THAW-C E C, HAMMOND C, PRATI L, HUTCHING J G. Glycerol oxidation using old-containing catalysts[J]. Acc Chem Res.,2015,48:1403−1412. doi: 10.1021/ar500426g [12] BENIPAL N, QI J, LIU Q, LI W. Carbon nanotube supported PdAg nanoparticles for electrocatalytic oxidation of glycerol in anion Exchange membrane fuel cells[J]. Appl Catal,2017,210:121−130. doi: 10.1016/j.apcatb.2017.02.082 [13] NANDENHA J, RAMOS C E D, DA SILVA G S, DE SOUZA R F B, FONTES H E, OTTONI C A, NETO A O. Borohydride reduction method for PdIn/C electrocatalysts synthesis towards glycerol electrooxidation under alkaline condition[J]. Eletroanalysis,2021,33(4):1115−1120. doi: 10.1002/elan.202060322 [14] ROSTAMI H, OMRANI A, ROSTAMI A A. On the role of electrodeposited nanostrutured Pd-Co alloy on Au for the electrocatalytic oxidation of glycerol in alcaline media[J]. Int J Hydrogen Energy,2015,40(30):9444−9451. doi: 10.1016/j.ijhydene.2015.05.154 [15] HAN J, KIM Y, KIM W H, JACKSON K H D, LEE D, CHANG H, CHAE J-H, LEE Y-K, KIM J H. Effect of atomic-layer-deposited TiO2 on carbono-supported Ni catalysts for electrocatalytic glycerol oxidation in alkaline media[J]. Electrochem Commun,2017,83:46−50. doi: 10.1016/j.elecom.2017.08.023 [16] SILVA M C J, BUZZO S G, DE SOUZA B F R, SPINACÉ V E, NETO O A, ASSUMPÇÃO T M H M. Enhanced eletrooxidation of ethanol using Pd/C + TiO2 electrocatalysts in alkaline media[J]. Electrocatalysis,2015,6:86−91. doi: 10.1007/s12678-014-0224-z [17] HAN J, KIM Y, JACKSON K H D, JEONG E-K, CHAE J-H, LEE Y-K, KIM J-H. Role of Au-TiO2 interfacial sites in enchancing the electrocatalytic glycerol oxidation performance[J]. Electrochem Commun,2018,96:16−21. doi: 10.1016/j.elecom.2018.09.004 [18] DE SOUZA M F, DE SOUZA B F R, BATISTA L B, DOS SANTOS C M, FONSECA C F, NETO, O A, NANDENHA J. Methane activation at low temperature in an acidic electrolyte using PdAu/C, PdCu/C, and PdTiO2/C electrocatalysts for PEMFC[J]. Res Chem Intermed,2020,46:2481−2496. doi: 10.1007/s11164-020-04102-1 [19] DELGADO A J, CLAVER C, CASTILLÓN S, CURULLA-FERRÉ D, ORDOMSKY V V, GODARD C. Fisher-Tropsch synthesis catalyzed by small TiO2 supported cobalt nanoparticles prepared by sodium borohydride reduction[J]. Appl Catal A: Gen,2016,513:39−46. doi: 10.1016/j.apcata.2015.12.019 [20] KIRKLAND A I, HUTCHISON J L. Nanocharacterization[M]. RSC Publishing: Cambridge, 2007, 304-307. [21] RADMILOVIC V, GASTEIGER H A, ROSS P N. Structure and chemical composition of a supported Pt-Ru electrocatalysts for methanol oxidation[J]. J Catal,1995,154(1):98−106. doi: 10.1006/jcat.1995.1151 [22] ANTONIASSI M R, SILVA M C J, NETO O A, SPINACÉ V E. Synthesis of Pt+SnO2/C electrocatalysts containing Pt nanoparticles with preferential (100) orientation for direct ethanol fuel cell[J]. Appl Catal,2017,218:91−100. doi: 10.1016/j.apcatb.2017.06.031 [23] OTTONI A C, DE SOUZA R R, DA SILVA S G, SPINACÉ V E, DE SOUZA B F R, NETO O A. Performance of Pd electrocatalyst supported on a physical mixture Indium tin oxide-carbon for glycerol electro-oxidation in alkaline media[J]. Electroanalysis,2017,29:960−964. doi: 10.1002/elan.201600569 [24] NETO O A, NANDENHA J, DE SOUZA B F R, BUZZO S G, SILVA M C J, SPINACÉ V E, ASSUMPÇÃO T M H M. Anodic oxidation of formic acid on PdAuIr/C-Sb2O5. SnO2 electrocatalysts prepared by borohydride reduction[J]. J Fuel Chem Technol,2014,42(7):851−857. doi: 10.1016/S1872-5813(14)60037-2 [25] GERALDES N A, SILVA F D, SILVA M C J, SOUZA B F R, SPINACÉ V E, NETO O A, LINARDI M, SANTOS C M. Glycerol electrooxidation in alkaline medium using Pd/C, Au/C and PdAu/C electrocatalysts prepared by electron beam irradiation[J]. J Braz Chem Soc,2014,25(5):831−840. [26] SIMÕES M, BARANTON S, COUTANCEAU C. Electro-oxidation of glycerol at Pd based nano-catalysts for an application in alkaline fuel cells for chemicals and energy cogeneration[J]. Appl Catal,2010,93(3/4):354−362. doi: 10.1016/j.apcatb.2009.10.008 [27] GRDÉN M, CZERWINSKI A. EQCM studies on Pd-Ni alloy oxidation in basic solution[J]. J Solid State Electrochem,2008,12:375−385. doi: 10.1007/s10008-007-0452-8 [28] ZHIANI M, ROSTAMI H, MAJIDI S, KARAMI K. Bis (dibenzylidene acetone) palladium (O) catalysts for glycerol oxidation in half cell and in alkaline direct glycerol fuel cell[J]. Int J Hydrogen Energy,2013,38(13):5435−5441. doi: 10.1016/j.ijhydene.2012.09.001 [29] WINIWARTER A, SILVIOLI L, SCOTT B S, RASMUSSEN-E K, SARIÇ M, TRIMARCO B D, VESBORG K C P, MOSES G P, STEPHENS L E I, SEGER B, ROSSMEISL J, CHORKENDORFF I. Towards an atomistic understanding of electrocatalytic partial hydrocarbon oxidation: propene on palladium[J]. Energy Environ Sci,2019,12(3):1055−1067. doi: 10.1039/C8EE03426E [30] GOMES F J, GARCIA C A, GASPAROTTO S H L, DE SOUZA E N, FERREIRA B E, PIRES C, FILHO-TREMILIOSI G. Influence of silver on the glycerol electro-oxidation over AuAg/C catalysts in alkaline medium: a cyclic voltammetry and in situ FTIR spectroscopy study[J]. Electrochim Acta,2014,144:361−368. doi: 10.1016/j.electacta.2014.08.035 [31] ZALINEEVA A, BARANTON S, COUTANCEAU C. How do Bi-modified palladium nanoparticles work toward glycerol electrooxidation? An in situ FTIT study[J]. Electrochim Acta,2015,176:705−717. doi: 10.1016/j.electacta.2015.07.073 [32] NANDENHA J, NAGAHAMA F H L, YAMASHITA Y J, FONTE H E, AYOUB S M J, DE SOUZA B F R, FONSECA C F, NETO O A. Activation of methane on PdZn/C electrocatalysts in an acidic electrolyte at low temperatures[J]. Int J Electrochem Sci,2019,14:10819−10834. [33] LAI L, HUANG G, WANG X WENG J. Preparation of Pt nanoparticle-loaded three-dimensional Fe3O4/carbon with high electro-oxidation activity[J]. Carbon,2011,49(5):1581−1587. doi: 10.1016/j.carbon.2010.12.040 -




 下载:
下载: