Cerium-modified copper/hexagonal mesoporous silica catalyst for efficient dimethyl oxalate hydrogenation to ethylene glycol under moderate reaction conditions
-
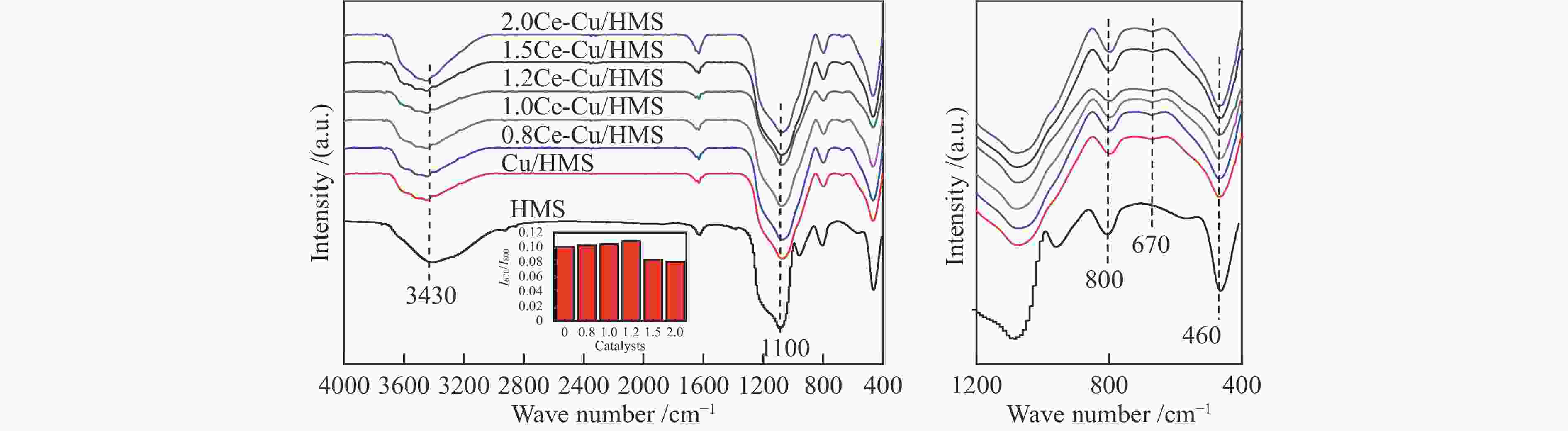
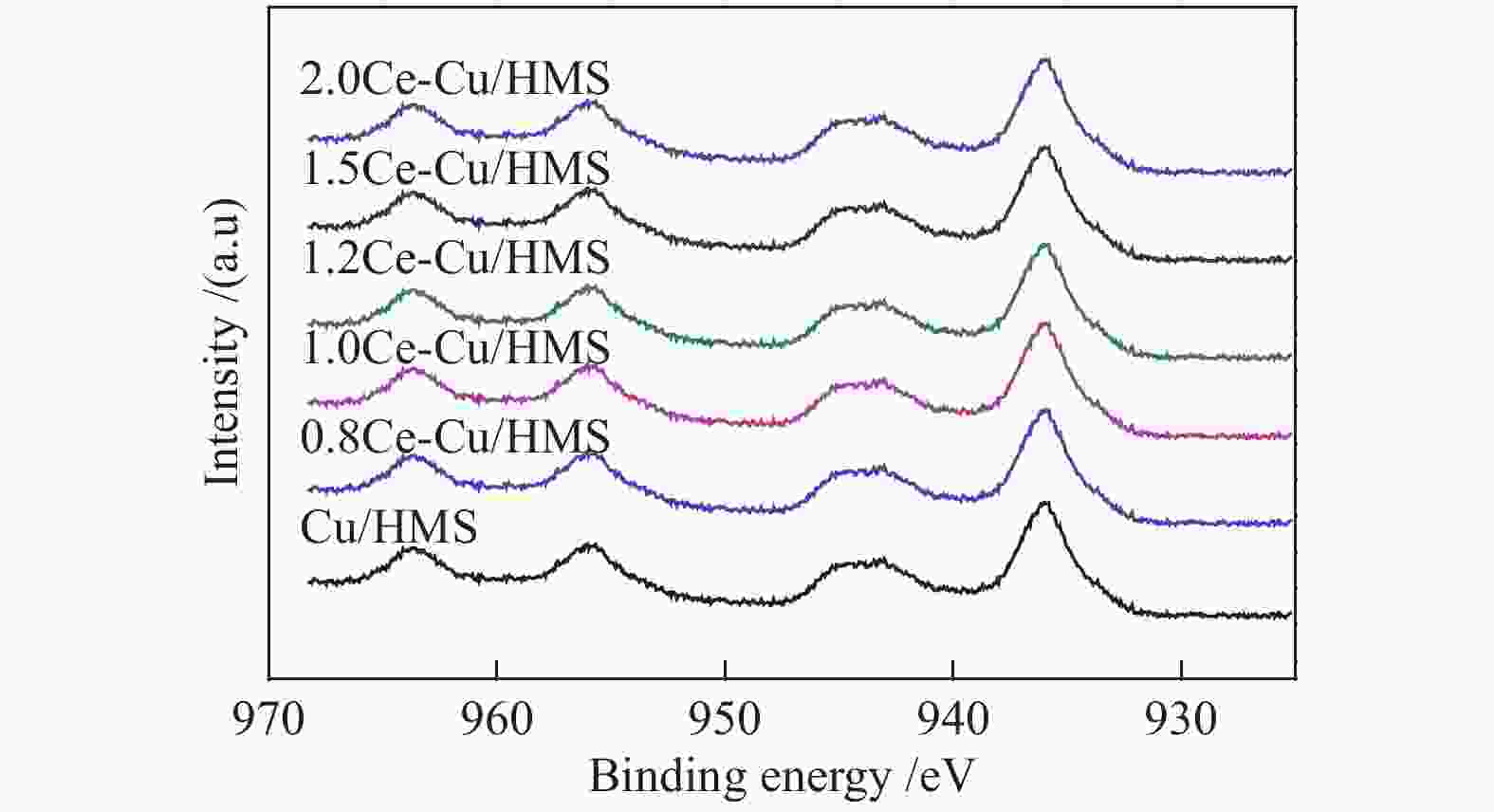
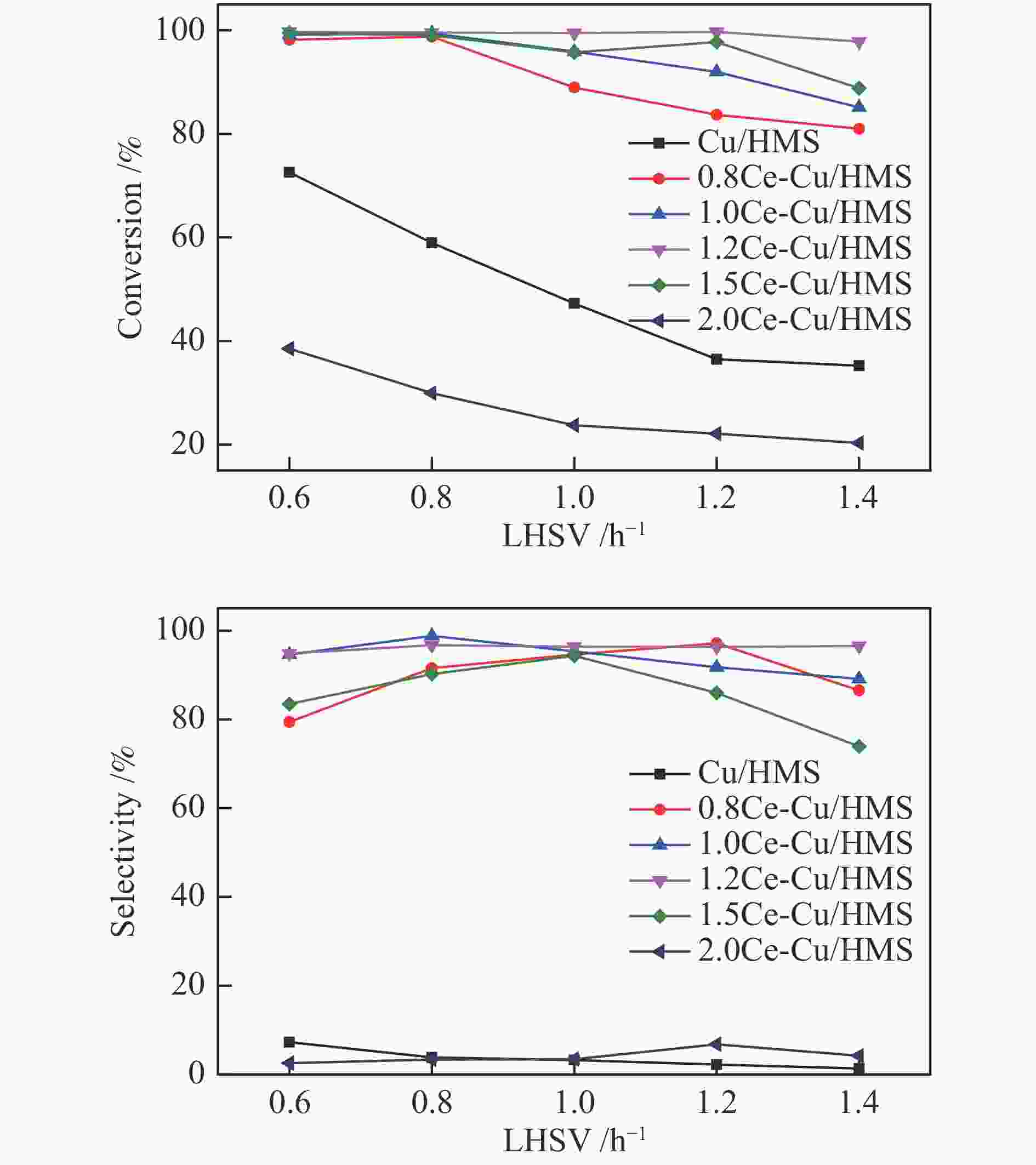
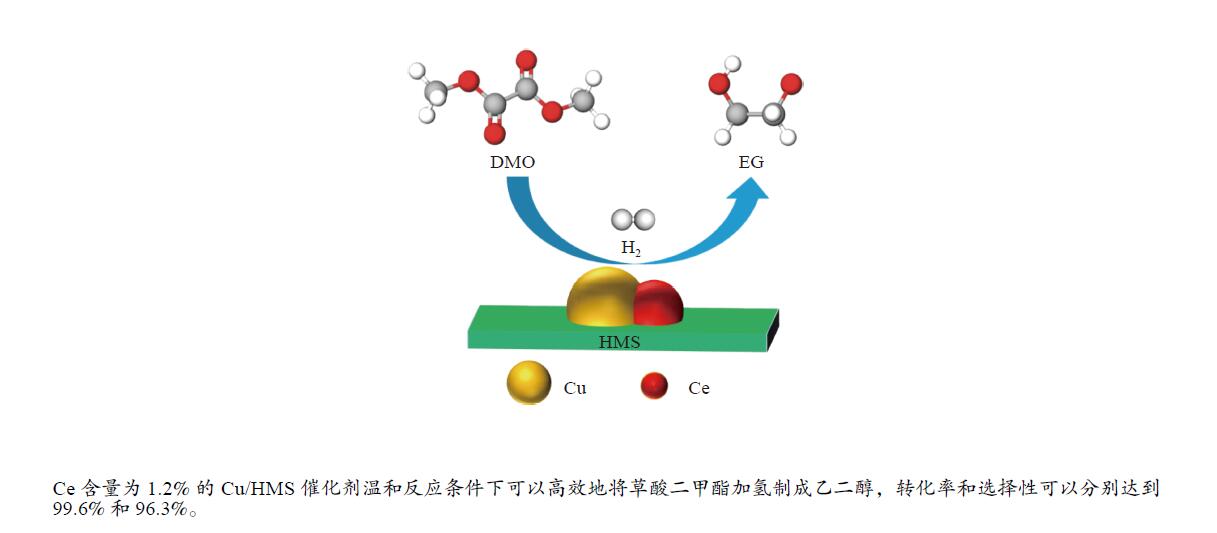
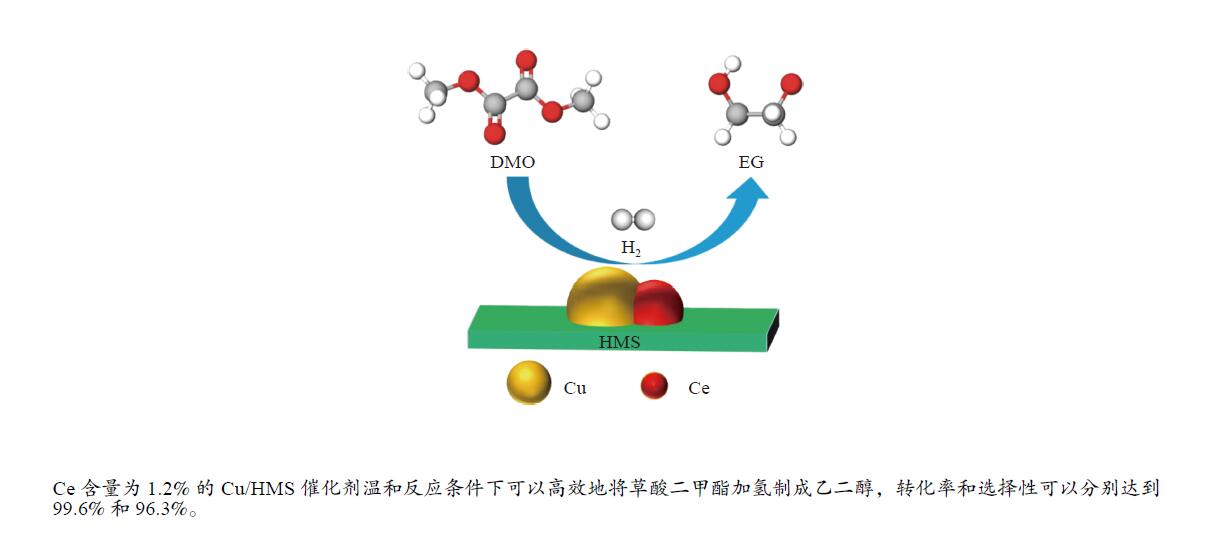
摘要: 采用蒸氨法制备了一种高效的铈改性铜/六方介孔二氧化硅(xCe-Cu/HMS)催化剂,用于草酸二甲酯(DMO)气相化学选择性氢化成乙二醇(EG)。铈助剂可以显著提高催化剂的性能,在引入1.2%的铈后,催化剂的性能最好。在温和的条件下(200 ℃,2.0 MPa,H2/DMO = 100,LHSVDMO = 1.2 h−1),DMO转化率和EG选择性分别达到了99.6%和96.3%。表征结果显示,Ce修饰的Cu/HMS可以增强Cu与载体之间的相互作用,改善Cu的分散性,并保持适当的Cu+/(Cu++Cu0)的比例。本研究采用简单、低成本的路线,合成了具有优良催化性能的Ce改性的Cu-HMS催化剂,实现了在温和的条件下DMO向EG的高选择性转化。Abstract: A highly efficient cerium-modified Cu/hexagonal mesoporous silica (xCe-Cu/HMS) catalyst for the vapor-phase hydrogenation of dimethyl oxalate (DMO) into ethylene glycol (EG) was prepared using an ammonia evaporation method. The Ce promoter can significantly improve the performance of the catalyst, and the best catalytic performance was obtained after the introduction of 1.2% Ce promoter on Cu/HMS. The DMO conversion and EG selectivity got to 99.6% and 96.3%, respectively, under moderate conditions (200 °C, 2.0 MPa, H2/DMO = 100 and LHSVDMO = 1.2 h−1). Characterization results revealed that Ce modification can enhance the interaction between Cu and the support, improve the dispersion of Cu on HMS, and maintain the appropriate ratio of Cu+/(Cu++Cu0). In this study, a simple and low-cost method was used to synthesize Ce-modified Cu-HMS catalysts, which showed excellent catalytic performance in conversion of DMO to EG under moderate conditions.
-
Key words:
- Cu/HMS /
- cerium promoter /
- dimethyl oxalate /
- hydrogenation /
- ethylene glycol
-
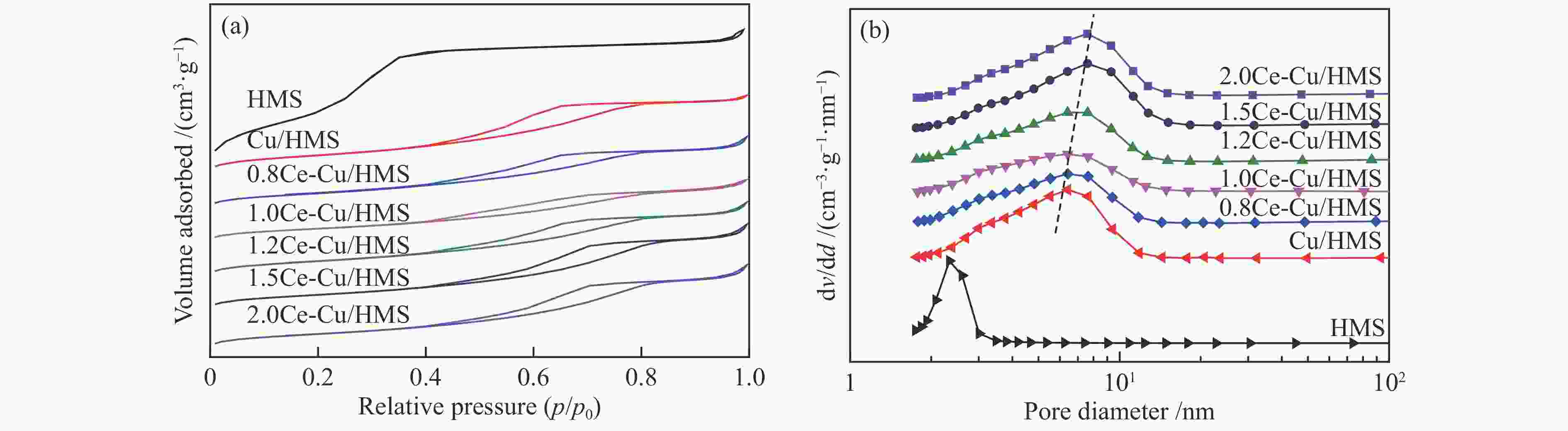
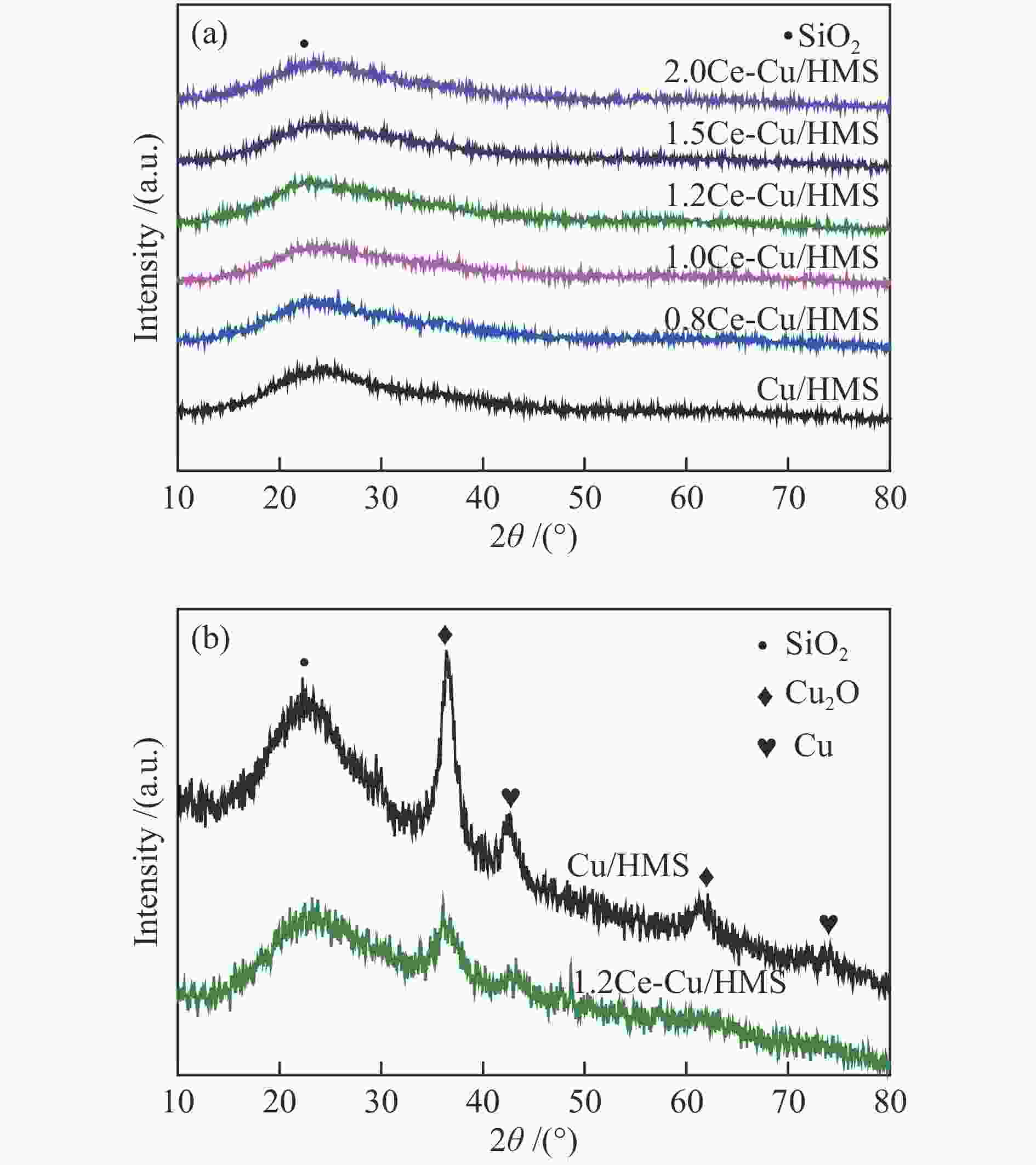
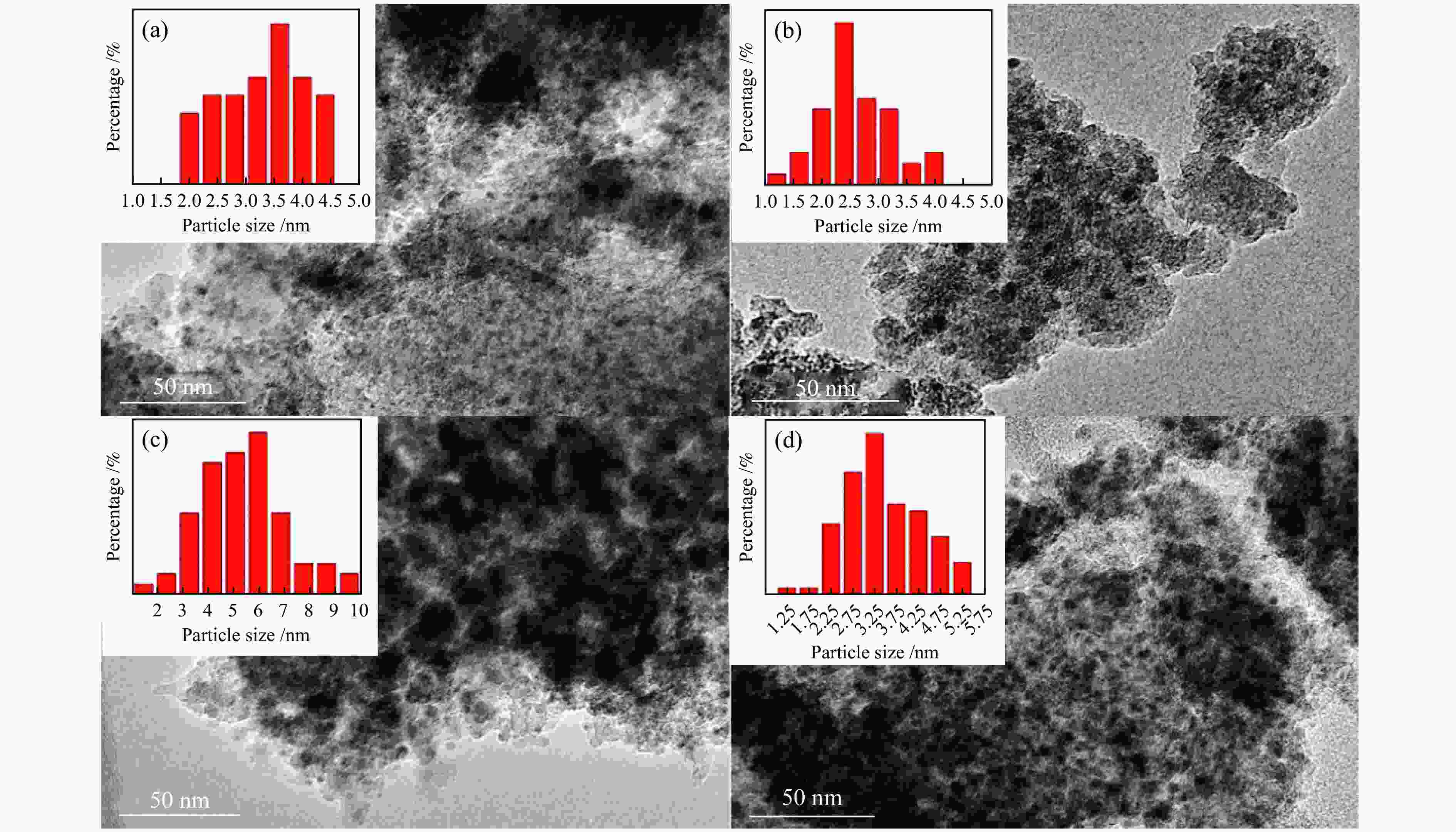
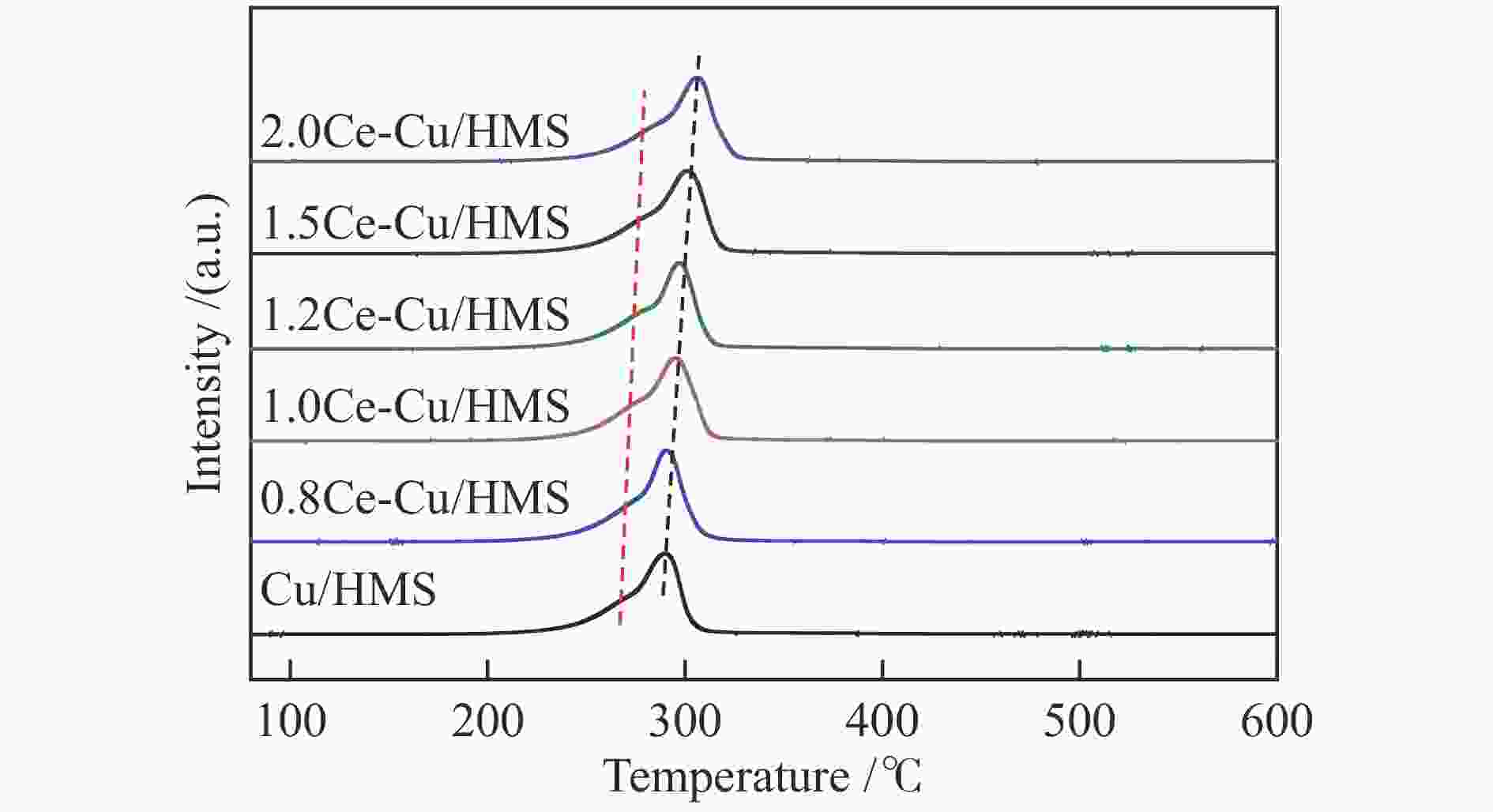
Table 1 Structural properties and chemical compositions of synthesized catalysts
Sample $S^{\rm{a}}_{{\rm{BET}}} $/(m2·g−1) $v_{\rm{p}}^{\rm{b}} $/(cm3·g−1) $D_{\rm{p}}^{\rm{c}} $/nm $D_{\rm{Cu}}^{\rm{d}} $/% $SA_{\rm{Cu}}^{\rm{e}} $/(m2·${\rm{g}}_{{\rm{cat}}}^{-1} $) $D_{\rm{vsCu}}^{\rm{f}} $/nm Ce loadingg/% HMS 1117 0.98 2.68 − − − − Cu/HMS 349 0.51 4.47 31.8 43.1 3.1 − 0.8Ce-Cu/HMS 310 0.47 4.95 39.0 52.8 2.6 0.83 1.0Ce-Cu/HMS 264 0.41 4.93 42.3 57.3 2.4 1.01 1.2Ce-Cu/HMS 308 0.48 5.08 45.5 61.5 2.2 1.44 1.5Ce-Cu/HMS 299 0.54 5.60 32.1 43.5 3.1 1.54 2.0Ce-Cu/HMS 299 0.53 5.58 21.1 28.5 4.7 1.96 a: BET specific surface area, b: Total pore volume obtained from N2 physisorption, c: Average pore size obtained from N2 physisorption, d: Cu dispersion determined by N2O titration, e: Cu surface area per gram of catalyst determined by N2O titration, f: Cu average volume-surface area diameter determined by N2O titration, g: Determined by ICP-AES Table 2 Catalytic performance of several catalysts under different reaction conditions
-
[1] YUE H, ZHAO Y, MA X, BGONG J. Ethylene glycol: Properties, synthesis, and applications[J]. Chem Soc Rev,2021,41(11):4218. [2] ZHU Y, KONG X, ZHENG H, ZHU Y. Strong metal-oxide interactions induce bifunctional and structural effects for Cu catalysts[J]. Mol Catal,2018,458:73−82. [3] WANG Z, SUN J, XU Z, GUO C. CO direct esterification to dimethyl oxalate and dimethyl carbonate: The key functional motifs for catalytic selectivity[J]. Nanoscale,2020,12(39):20131−20140. [4] WANG X, CHEN M, CHEN X, LIN R, ZHU H, HUANG C, YANG W, TAN Y, WANG S, DU Z, DING Y. Constructing copper-zinc interface for selective hydrogenation of dimethyl oxalate[J]. J Catal,2020,383:254−263. [5] TUREK T, TRIMM D L. The catalytic hydrogenolysis of esters to alcohols[J]. Catal Rev,1994,36(4):645−683. [6] VANDERGRIFT C J G, ELBERSE P A, MULDER A, GEUS J W. Prepapation of silica-supported copper-catalysts by means of depositon precipitation[J]. Appl Catal,1990,59(2):275−289. [7] YUAN P, LIU Z, ZHANG W, SUN H, LIU S. Cu-Zn/Al2O3 catalyst for the hydrogenation of esters to alcohols[J]. Chin J Catal,2010,31(7):769−775. [8] CUI G, MENG X, ZHANG X, WANG W, XU S, YE Y, TANG K, WANG W, ZHU J, WEI M, EVANS D G, DUAN X. Low-temperature hydrogenation of dimethyl oxalate to ethylene glycol via ternary synergistic catalysis of Cu and acid-base sites[J]. Appl Catal B: Environ,2019,248:394−404. [9] AI P, TAN M, REUBROYCHAROEN P, WANG Y, FENG X, LIU G, YANG G, TSUBAKI N. Probing the promotional roles of cerium in the structure and performance of Cu/SiO2 catalysts for ethanol production[J]. Catal Sci Technol,2018,8(24):6441−6451. [10] SITTHISA S, SOOKNOI T, MA Y, BALBUENA P B, RESASCO D E. Kinetics and mechanism of hydrogenation of furfural on Cu/SiO2 catalysts[J]. J Catal,2011,277(1):1−13. [11] BRANDS D S, POELS E K, BLIEK A. Ester hydrogenolysis over promoted Cu/SiO2 catalysts[J]. Appl Catal A: Gen,1999,184(2):279−289. [12] HUANG Z, CUI F, XUE J, ZUO J, CHEN J, XIA C. Synthesis and structural characterization of silica dispersed copper nanomaterials with unusual thermal stability prepared by precipitation-gel method[J]. J Phys Chem C,2010,114(39):16104−16113. [13] YIN A, WEN C, GUO X, DAI W, FAN K. Influence of Ni species on the structural evolution of Cu/SiO2 catalyst for the chemoselective hydrogenation of dimethyl oxalate[J]. J Catal,2011,280(1):77−88. [14] SHENG H, ZHANG H, MA H, QIAN W, YING W. An effective Cu-Ag/HMS bimetallic catalyst for hydrogenation of methyl acetate to ethanol[J]. Catal Today,2020,358:122−128. [15] ZHENG X, LIN H, ZHENG J, DUAN X, YUAN Y. Lanthanum oxide-modified Cu/SiO2 as a high-performance catalyst for chemoselective hydrogenation of dimethyl oxalate to ethylene glycol[J]. ACS Catal,2013,3(12):2738−2749. [16] CHEN L, GUO P, QIAO M, YAN S, LI H, SHEN W, XU H, FAN K. Cu/SiO2 catalysts prepared by the ammonia-evaporation method: Texture, structure, and catalytic performance in hydrogenation of dimethyl oxalate to ethylene glycol[J]. J Catal,2008,257(1):172−180. [17] DING J, POPA T, TANG J, GASEM K A M, FAN M, ZHONG Q. Highly selective and stable Cu/SiO2 catalysts prepared with a green method for hydrogenation of diethyl oxalate into ethylene glycol[J]. Appl Catal B: Environ,2017,209:530−542. [18] YIN A, GUO X, FAN K, DAI W. Ion-exchange temperature effect on Cu/HMS catalysts for the hydrogenation of dimethyl oxalate to ethylene glycol[J]. ChemCatChem,2010,2(2):206−213. [19] DONG X, MA X, XU H, GE Q. Comparative study of silica-supported copper catalysts prepared by different methods: Formation and transition of copper phyllosilicate[J]. Catal Sci Technol,2016,6(12):4154−4158. [20] CHEN C, LIN L, YE R, HUANG L, ZHU L, HUANG Y, QIN Y, YAO Y. Construction of Cu-Ce composite oxides by simultaneous ammonia evaporation method to enhance catalytic performance of Ce-Cu/SiO2 catalysts for dimethyl oxalate hydrogenation[J]. Fuel,2021,290:120083. [21] YE C, GUO C, ZHANG J. Highly active and stable CeO2-SiO2 supported Cu catalysts for the hydrogenation of methyl acetate to ethanol[J]. Fuel Process Technol,2016,143:219−224. [22] TANEV P T, PINNAVAIA T J. A neutral templating route to mesoporous molecular-sieves[J]. Science,1995,267(5199):865−867. [23] VINU A, SAWANT D P, ARIGA K, HOSSAIN K Z, HALLIGUDI S B, HARTMANN MNOMURA M. Direct synthesis of well-ordered and unusually reactive FeSBA-15 mesoporous molecular sieves[J]. Chem Mater,2005,17(21):5339−5345. [24] ZHAO Y, ZHANG Y, WANG Y, ZHANG J, XU Y, WANG S, MA X. Structure evolution of mesoporous silica supported copper catalyst for dimethyl oxalate hydrogenation[J]. Appl Catal A: Gen,2017,539:59−69. [25] ZHU S, GAO X, ZHU Y, FAN W, WANG J, LI Y. A highly efficient and robust Cu/SiO2 catalyst prepared by the ammonia evaporation hydrothermal method for glycerol hydrogenolysis to 1, 2-propanediol[J]. Catal Sci Technol,2015,5(2):1169−1180. [26] YIN A, GUO X, FAN K, DAI W. Influence of copper precursors on the structure evolution and catalytic performance of Cu/HMS catalysts in the hydrogenation of dimethyl oxalate to ethylene glycol[J]. Appl Catal A: Gen,2010,377(1/2):128−133. [27] DING J, WANG M, LIU H, GUO X, YU G, WANG Y. Effect of Cu content on Ce-doping CuO/ZrO2 catalysts for low-temperature hydrogenation of dimethyl oxalate to ethanol[J]. Asia-Pac J Chem Eng,2021,16(5):e2692. [28] BANCQUART S, VANHOVE C, POUILLOUX Y, BARRAULT J. Glycerol transesterification with methyl stearate over solid basic catalysts i. Relationship between activity and basicity[J]. Appl Catal A: Gen,2001,218(1-2):1−11. [29] TOUPANCE T, KERMAREC M, LAMBERT J F, LOUIS C. Conditions of formation of copper phyllosilicates in silica-supported copper catalysts prepared by selective adsorption[J]. J Phys Chem B,2002,106(9):2277−2286. [30] YUE H, ZHAO Y, ZHAO S, WANG B, MA X, GONG J. A copper-phyllosilicate core-sheath nanoreactor for carbon-oxygen hydrogenolysis reactions[J]. Nat Commun,2013,4:2339. [31] DI W, CHENG J, TIAN S, LI J, CHEN J, SUN Q. Synthesis and characterization of supported copper phyllosilicate catalysts for acetic ester hydrogenation to ethanol[J]. Appl Catal A: Gen,2016,510:244−259. [32] GERVASINI A, MANZOLI M, MARTRA G, PONTI A, RAVASIO N, SORDELLI L, ZACCHERIA F. Dependence of copper species on the nature of the support for dispersed CuO catalysts[J]. J Phys Chem B,2006,110(15):7851−7861. [33] RAIMONDI F, GEISSLER K, WAMBACH J, WOKAUN A. Hydrogen production by methanol reforming: Post-reaction characterisation of a Cu/ZnO/Al2O3 catalyst by XPS and TPD[J]. Appl Sur Sci,2002,189(1/2):59−71. [34] AI P, TAN M, ISHIKURO Y, HOSOI Y, YANG G, YONEYAMA Y, TSUBAKI N. Design of an autoreduced copper in carbon nanotube catalyst to realize the precisely selective hydrogenation of dimethyl oxalate[J]. ChemCatChem,2017,9(6):1067−1075. [35] YIN A, GUO X, DAI W, FAN K. High activity and selectivity of Ag/SiO2 catalyst for hydrogenation of dimethyl oxalate[J]. Chem Commun,2010,46(24):4348−4350. [36] ZHANG S, LIU Q, FAN G, LI F. Highly-dispersed copper-based catalysts from Cu-Zn-Al layered double hydroxide precursor for gas-phase hydrogenation of dimethyl oxalate to ethylene glycol[J]. Catal Lett,2012,142(9):1121−1127. [37] WEN C, CUI Y, YIN A, FAN K, DAI W. Remarkable improvement of catalytic performance for a new cobalt-decorated Cu/HMS catalyst in the hydrogenation of dimethyloxalate[J]. ChemCatChem,2013,5(1):138−141. [38] LI S, WANG Y, ZHANG J, WANG S, XU Y, ZHAO Y, MA X. Kinetics study of hydrogenation of dimethyl oxalate over Cu/SiO2 catalyst[J]. Ind Eng Chem Res,2015,54(4):1243−1250. -




 下载:
下载: