Performance of reversible solid oxide cells based on La0.6Ca0.4Fe0.7Sc0.1Ni0.2O3−δ oxygen electrode
-
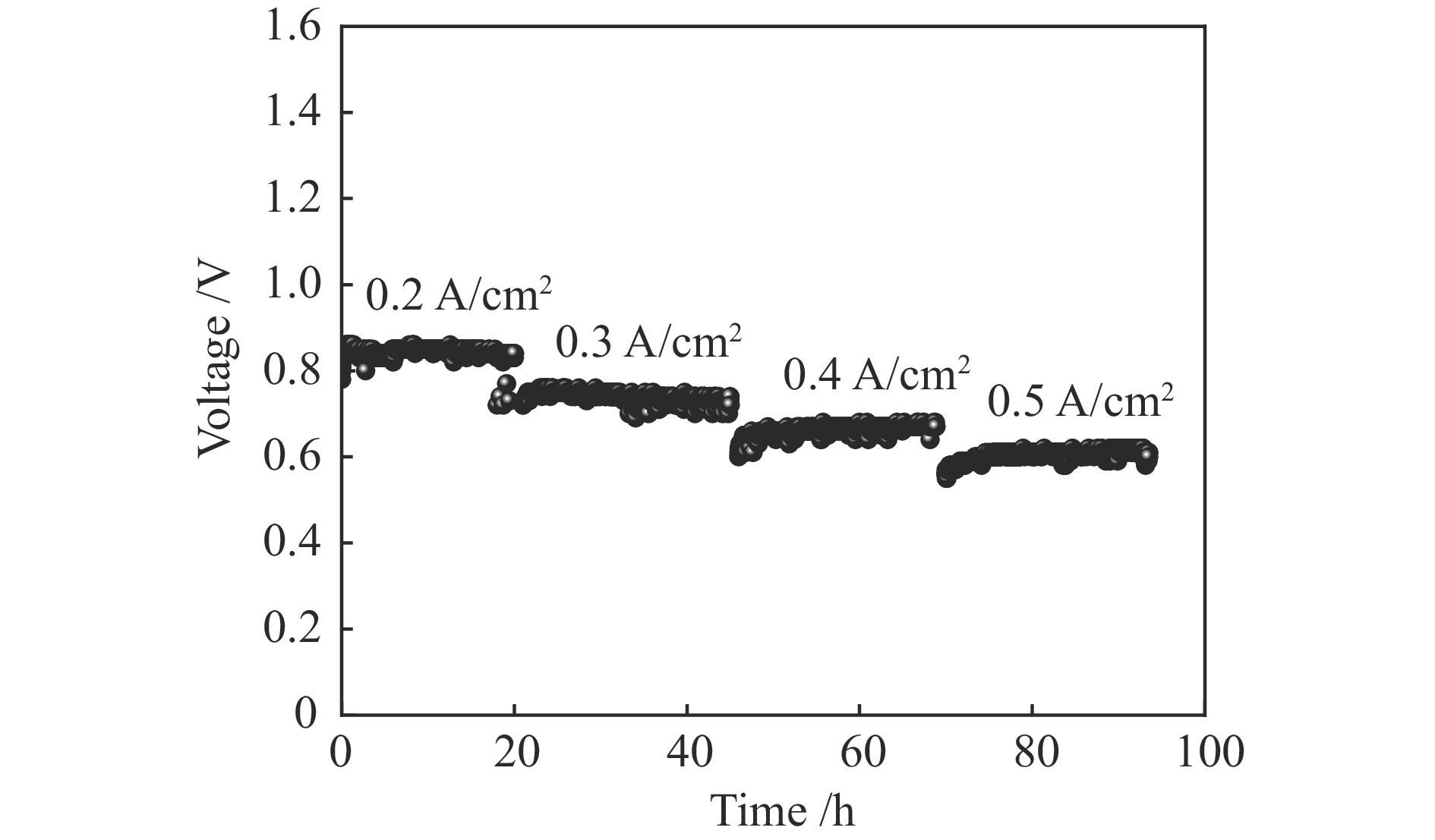
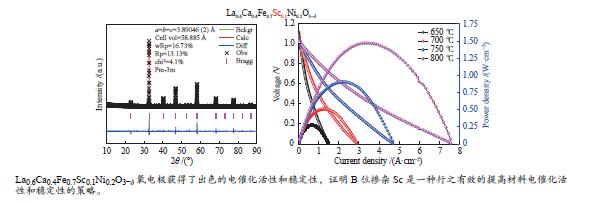
摘要: 本研究制备了10% Sc掺杂的La0.6Ca0.4Fe0.7Sc0.1Ni0.2O3−δ (LCFSN)材料,采用半电池研究了该材料的氧还原催化活性(ORR)和氧析出催化活性(OER),发现LCFSN的ORR催化活性优于OER催化活性。组装了Ni-YSZ((Y2O3)0.08(ZrO2)0.92)/YSZ/GDC(Gd0.1Ce0.9O2−δ)/LCFSN全电池并研究了其在燃料电池模式(SOFC)和电解池模式(SOEC)下的电化学性能,电池在800 ℃以H2为燃料时的最高功率密度可达1.471 W/cm2,在750 ℃,50%H2O和1.3 V热中性电解电压条件下其产氢速率高达627 mL/(cm2·h)。而且电池在100 h的稳定性测试中性能没有衰减,具有良好的稳定性。通过研究,证明了LCFSN是一个具有良好发展前景的氧电极材料。
-
关键词:
- 可逆固体氧化物电池 /
- 氧电极 /
- La0.6Ca0.4Fe0.7Sc0.1Ni0.2O3−δ /
- 电化学性能 /
- 稳定性
Abstract: Hydrogen energy is recognized as the most potential energy carrier in the 21st century. Reversible solid oxide cells (RSOCs) have attracted more and more attention due to their efficient use of hydrogen for power generation and efficient hydrogen production from water electrolysis. Numerous studies have shown the polarization loss and decay of oxygen electrodes are the technical bottlenecks hindering RSOCs development. In this work, 10% mol Sc-doped La0.6Ca0.4Fe0.7Sc0.1Ni0.2O3−δ (LCFSN) material was prepared, and the oxygen reduction reaction (ORR) and oxygen evolution reaction (OER) of this material were studied in detail by half cell. It is found that ORR catalytic activity of LCFSN is better than the OER. The Ni-YSZ/YSZ/GDC/LCFSN full cells were assembled and their electrochemical performances in fuel cell mode (SOFC) and electrolysis cell mode (SOEC) were investigated in detail. The maximum power density can reach 1.471 W/cm2 at 800 ℃ with H2 as fuel. And the hydrogen production rate is as high as 627 mL/(cm2·h) at 750 ℃, 50 %H2O and 1.3 V. In addition, the cell has no obvious degradation in the 100 h stability test and has good stability. These results prove that LCFSN is a promising oxygen electrode material. -
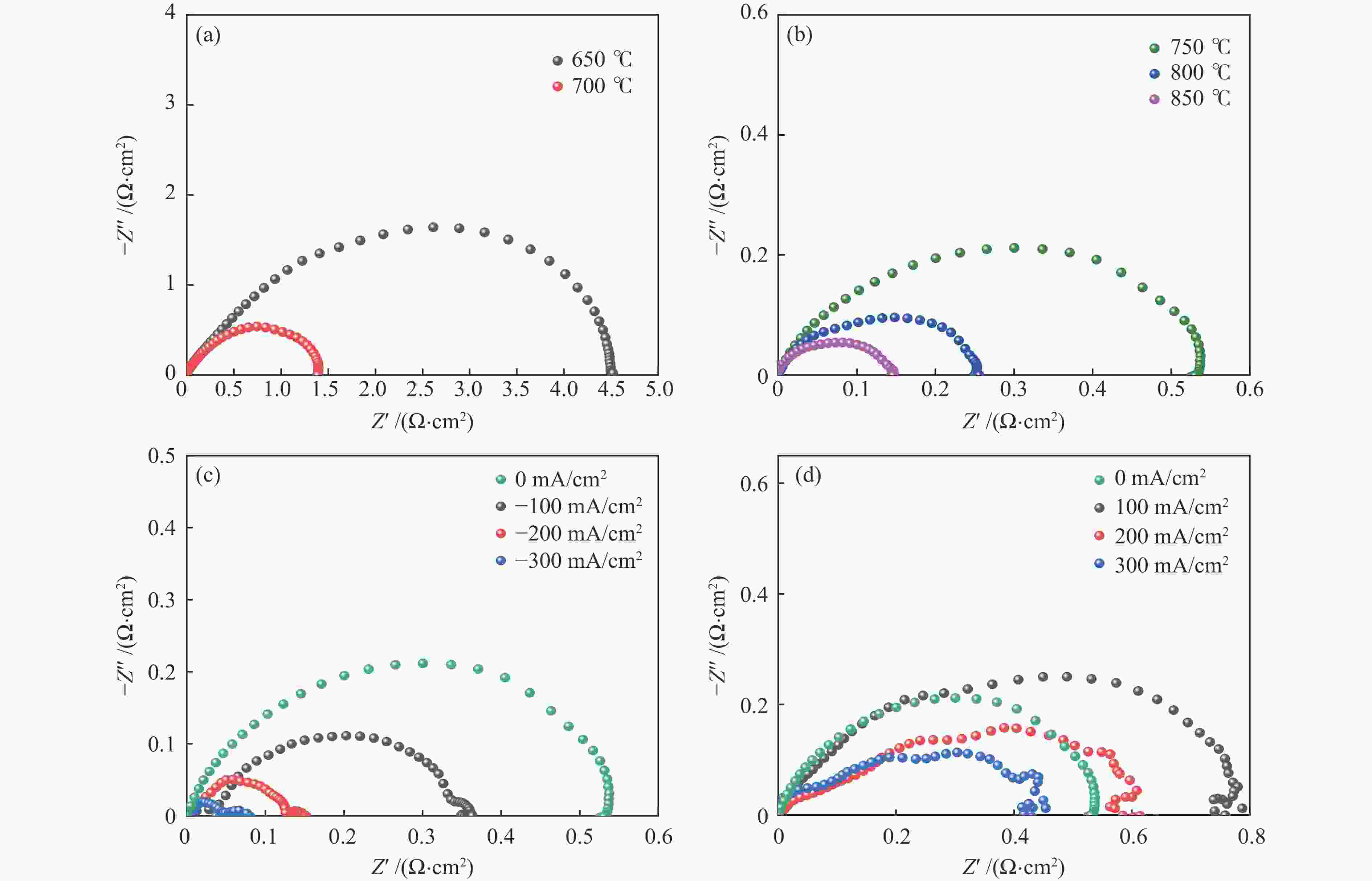
图 3 基于LCFSN材料的半电池性能研究,650−700 ℃(a)和750−850 ℃(b)开路状态下的交流阻抗谱,750 ℃时ORR模式(c)和OER模式(d)的不同电流密度下的交流阻抗谱
Figure 3 Performance of half cell based on LCFSN. AC impedance spectra under OCV condition at 650−700 ℃ (a), at 750−850 ℃ (b), AC impedance spectra at 750 ℃ with different current densities in ORR mode (c), in OER mode (d)
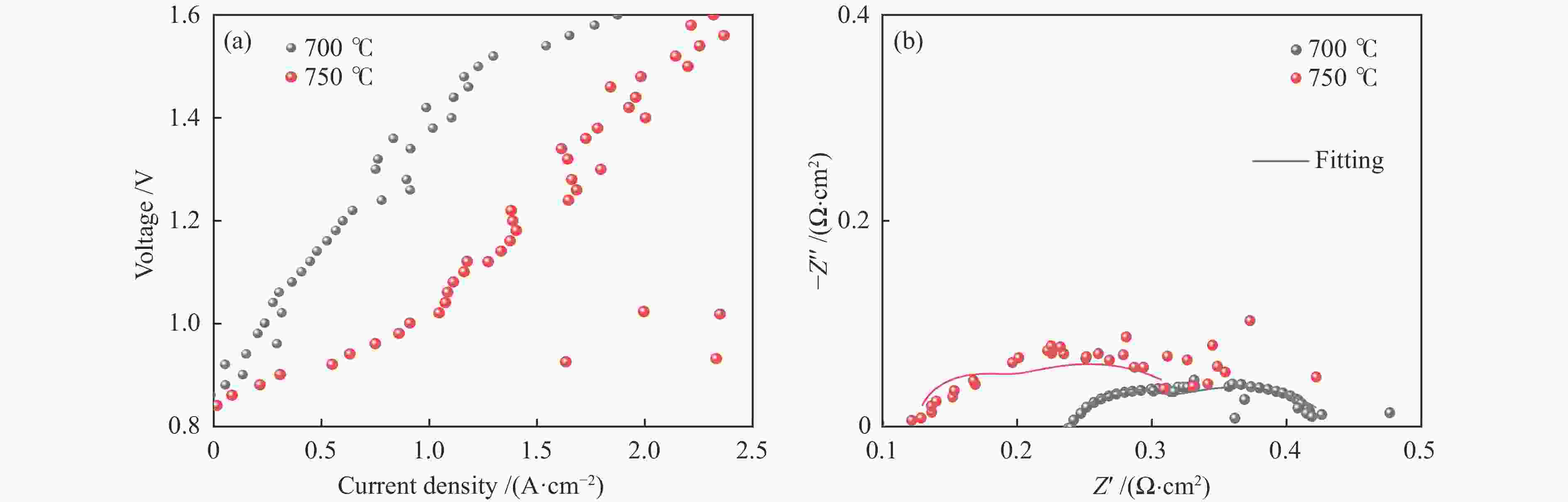
图 7 Ni-YSZ/YSZ/GDC/LCFSN无烧结全电池在H2下不同温度的I-V-P曲线(a)和开路状态下的交流阻抗谱(b),750 ℃@0.4 A/cm2的极化曲线(c)和12 h的稳定性测试曲线(d)
Figure 7 I-V-P curves (a) and EIS curves (b) at different temperatures, polarization curves at 750 ℃@0.4 A/cm2 (c), 12 h stability test (d) of Ni-YSZ/YSZ/GDC/LCFSN non-sintered single cell under H2
-
[1] KIM D, PARK J W, CHAE M S. An efficient and robust lanthanum strontium cobalt ferrite catalyst as a bifunctional oxygen electrode for reversible solid oxide cells[J]. J Mater Chem A,2021,9(9):5507−5521. doi: 10.1039/D0TA11233J [2] 朱冕, 赵加佩, 李欣珂, 梁超余, 袁金良. 可逆固体氧化物燃料电池(rSOFC)技术的研究进展[J]. 电源技术,2020,44(3):469−474.ZHU Mian, ZHAO Jia-pei, LI Xin-ke, LIANG Chao-yu, YUAN Jin-liang. Research status and prospects of reversible solid oxide fuel cell (rSOFC) technology[J]. Chin J Power Sources,2020,44(3):469−474. [3] SHIN J, LEE Y J, JAN A, CHOI S M, YOON K J. Highly active and thermally stable single-atom catalysts for high-temperature electrochemical devices[J]. Energy Environ Sci,2020,13:4903−4920. doi: 10.1039/D0EE01680B [4] 赵建军, 甄文龙, 吕功煊. 固体氧化物电解CO2技术现状与前景[J]. 燃料化学学报,2022,50(10):1237−1258. doi: 10.19906/j.cnki.jfct.2022028ZHAO Jian-jun, ZHEN Wen-long, LÜ Gong-xuan. Solid oxide electrolysis of carbon dioxide: Status and perspectives[J]. J Fuel Chem Technol,2022,50(10):1237−1258. doi: 10.19906/j.cnki.jfct.2022028 [5] HAUCH A, KUNGAS R, BLENNOW P, HANSEN A B, MOGENSEN M B. Recent advances in solid oxide cell technology for electrolysis[J]. Science,2020,370(6513):aba6118. [6] VIBHU V, VINKE I C, EICHEL R A. Cobalt substituted Pr2Ni1−xCoxO4 + δ (x= 0, 0.1, 0.2) oxygen electrodes: Impact on electrochemical performance and durability of solid oxide electrolysis cells[J]. J Power Sources,2021,482:228909. doi: 10.1016/j.jpowsour.2020.228909 [7] PARK B K, ZHANG Q, VOORHEES P W, BARNETT S A. Conditions for stable operation of solid oxide electrolysis cells: Oxygen electrode effects[J]. Energy Environ Sci,2019,12:3053−3062. doi: 10.1039/C9EE01664C [8] HOU Q, GUAN C, XIAO G, WANG J, ZHU Z. Effect of oxygen partial pressure on solid oxide electrolysis cells[J]. Acta Phys Chim Sin,2019,35(3):284−291. doi: 10.3866/PKU.WHXB201804171 [9] 梁明德, 于波, 文明芬, 陈靖, 徐景明, 翟玉春. 阴极支撑Ni-YSZ/YSZ/LSM-YSZ固体氧化物电解池制氢性能[J]. 中国稀土学报,2009,27(5):647−651.LIANG Ming-de, YU Bo, WEN Ming-fen, CHEN Jing, XU Jing-ming, ZHAI Yu-chun. Cathode-supported Ni-YSZ/YSZ/LSM-YSZ solid oxide electrolytic cell for hydrogen production[J]. J Rare Earths,2009,27(5):647−651. [10] YANG C, COFFIN A, CHEN F. High temperature solid oxide electrolysis cell employing porous structured (La0.75Sr0.25)0.95MnO3 with enhanced oxygen electrode performance[J]. Int J Hydrog Energy,2010,35(8):3221−3226. doi: 10.1016/j.ijhydene.2010.01.056 [11] SUBOTI V, FUTAMURA S, HARRINGTON G F, MASTSUDA J, SASAKI K. Towards understanding of oxygen electrode processes during solid oxide electrolysis operation to improve simultaneous fuel and oxygen generation[J]. J Power Sources,2021,492(1):229600. [12] PARK J, JUNG C, KIM K J, KIM D, SHIN H R, HONG J E, LEE K T. Enhancing bifunctional electrocatalytic activities of oxygen electrodes via incorporating highly conductive Sm3 + and Nd3 + double-doped ceria for reversible solid oxide cells[J]. ACS Appl Mater Interfaces,2021,13(2):2496−2506. doi: 10.1021/acsami.0c17238 [13] EBBESEN S D, SUN X, MOGENSEN M B. Understanding the processes governing performance and durability of solid oxide electrolysis cells[J]. Faraday Discuss,2015,182:393−422. doi: 10.1039/C5FD00032G [14] CHEN K, JIANG S P. Surface segregation in solid oxide cell oxygen electrodes: Phenomena, mitigation strategies and electrochemical properties[J]. Electrochem Energy Rev,2020,3(4):730−765. [15] MA Z, YIN Y, LEI J, ZHOU N. A robust high performance cobalt-free oxygen electrode La0.5Sr0.5Fe0.8Cu0.15Nb0.05O3-δ for reversible solid oxide electrochemical cell[J]. J Power Sources,2017,340(2):373−379. [16] MENG X, SHEN Y, XIE M, YIN Y, YANG N, MA Z, JOAO C, DIAIZ D, LIU S. Novel solid oxide cells with SrCo0.8Fe0.1Ga0.1O3−δ oxygen electrode for flexible power generation and hydrogen production[J]. J Power Sources,2016,306(29):226−232. [17] LIU X, HAN D, ZHOU Y, MENG X, WU H, LI J, ZENG F, ZHAN Z. Sc-substituted La0.6Sr0.4FeO3−δ mixed conducting oxides as promising electrodes for symmetrical solid oxide fuel cells[J]. J Power Sources,2014,246:457−463. doi: 10.1016/j.jpowsour.2013.07.111 [18] 赵晓虹, 王勇, 刘立敏, 李斌, 固体氧化物燃料电池新型钙钛矿. La0.9Ca0.1Fe0.9Nb0.1O3−δ阳极的制备及其性能研究[J]. 无机材料学报,2017,32(11):1188−1194.ZHAO Xiao-hong, WANG Yong, LIU Li-min, LI Bin. Preparation and properties of novel perovskite La0.9Ca0.1Fe0.9Nb0.1O3−δ anode for solid oxide fuel cells[J]. J Inorg Mater,2017,32(11):1188−1194. [19] LIU X, JIN F, LIU X, SUN N, LI J, SHEN Y, WANG F, YANG L, CHU X, XU M. Effect of calcium doping on Sm1−xCaxBaCo2O5 + δ cathode materials for intermediate-temperature solid oxide fuel cells[J]. Electrochim Acta,2021,390:138830. doi: 10.1016/j.electacta.2021.138830 [20] XIE X, XU Q, HUANG D, XIAO J, CHEN M, ZHAO K, CHEN D, ZHANG F. Insights into surface alkaline-earth segregation on the surface of La0.3Me0.7Fe0.7Cr0.3O3−δ (Me= Sr, Ca) porous electrodes for oxygen reduction reaction[J]. Appl Surf Sci,2021,566:150659. doi: 10.1016/j.apsusc.2021.150659 [21] TIAN Y, LIU Y, NADEN A, JIA L, XU M, CUI W, CHI B, PU J, IRVINE J, LI J. Boosting CO2 electrolysis performance via calcium-oxide-looping combined with in situ exsolved Ni-Fe nanoparticles in a symmetrical solid oxide electrolysis cell[J]. J Mater Chem A,2020,8:14895−14899. doi: 10.1039/D0TA05518B [22] 田云峰. 铁酸镧基固体氧化物电池电极材料电化学性能研究[D]. 武汉: 华中科技大学, 2020.TIAN Yun-feng. Study on the electrochemical performance of lanthanum ferrite-based electrode for solid oxide cells[D]. Wuhan: Huazhong University of Science and Technology, 2020. [23] WANG W, TIAN Y, LIU Y, ABHISHEK N, LI Y, CHI B, PU J. Tailored Sr-Co-free perovskite oxide as an air electrode for high-performance reversible solid oxide cells[J]. Sci China Mater,2021,64(7):1621−1631. doi: 10.1007/s40843-020-1567-2 [24] LIU Y, TIAN Y, WANG W, LI Y, PU J. Promoting electrocatalytic activity and stability via Er0.4Bi1.6O3−δ in situ decorated La0.8Sr0.2MnO3−δ oxygen electrode in reversible solid oxide cell[J]. ACS Appl Mater Interfaces,2020,12(52):57941−57949. doi: 10.1021/acsami.0c18583 [25] TIAN Y, WANG W, LIU Y, ZHANG L, LI J. Cobalt-free perovskite oxide La0.6Sr0.4Fe0.8Ni 0.2O3-δ as active and robust oxygen electrode for reversible solid oxide cells[J]. ACS Appl Energy Mater,2019,2(5):3297−3305. doi: 10.1021/acsaem.9b00115 [26] TIAN Y, YANG C, LI Y. A simple Sc doping strategy to enhance electrocatalytic activity and stability in symmetrical solid oxide cells[J]. J Electrochem Soc,2021,168(10):104515. doi: 10.1149/1945-7111/ac2e1e [27] LOPEZ-ROBLEDO M J, LAGUNA-BERCERO M A, LARREA A, ORERA V M. Reversible operation of microtubular solid oxide cells using La0.6Sr0.4Co0.2Fe0.8O3−δ-Ce0.9Gd0.1O2−δ oxygen electrodes[J]. J Power Sources,2018,378:184−189. doi: 10.1016/j.jpowsour.2017.12.035 [28] LAGUNA-BERCERO M A, MONZON H, LARREA A, ORERA V M. Improved stability of reversible solid oxide cells with a nickelate-based oxygen electrode[J]. J Mater Chem A,2016,4(4):1446−1453. doi: 10.1039/C5TA08531D [29] YANG E H, NOH Y S, RAMESH S, LIM S S, MOON D J. The effect of promoters in La0.9M0.1Ni0.5Fe0.5O3 (M= Sr, Ca) perovskite catalysts on dry reforming of methane[J]. Fuel Process Technol,2015,134:404−413. doi: 10.1016/j.fuproc.2015.02.023 [30] HUI F, KEANE M, SINGH P, HAN M. Electrochemical performance and stability of lanthanum strontium cobalt ferrite oxygen electrode with gadolinia doped ceria barrier layer for reversible solid oxide fuel cell[J]. J Power Sources,2014,268(5):634−639. [31] AI N, CHEN M, HE S, CHEN K, ZHANG T, JIANG S P. High performance nanostructured bismuth oxide-cobaltite as a durable oxygen electrode for reversible solid oxide cells[J]. J Mater Chem A,2018,6(15):6510−6520. doi: 10.1039/C8TA00370J [32] AI N, HE S, LI N, ZHANG Q, RICKARD W, CHEN K, ZHANG T, JIANG S P. Suppressed Sr segregation and performance of directly assembled La0.6Sr0.4Co0.2Fe0.8O3−δ oxygen electrode on Y2O3-ZrO2 electrolyte of solid oxide electrolysis cells[J]. J Power Sources,2018,384:125−135. doi: 10.1016/j.jpowsour.2018.02.082 -





 下载:
下载: