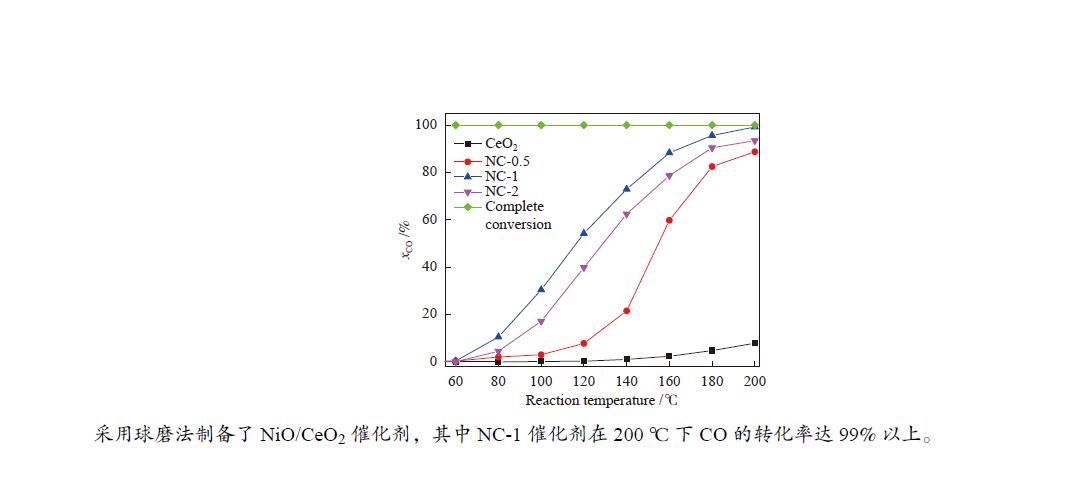
Effect of Ni content on catalytic oxidation of CO over NiO/CeO2 catalyst
-
摘要: 以CeO2为载体,Ni为活性组分,采用球磨法制备了低温条件下性能较好的CO氧化NiO/CeO2催化剂。通过XRD、BET、H2-TPR、XRF和XPS等技术对催化剂进行了表征,考察了镍含量对催化剂结构和CO低温氧化性能的影响。结果表明,不同Ni/Ce比主要影响催化剂表面晶格氧空位的数量以及活性组分和载体之间的相互作用。其中,Ni/Ce物质的量比为1∶9时,催化剂表面氧空位数量较多,因此,表现出优良的催化性能。在反应温度为200 ℃、氧过量系数为5、气体总空速为60000 mL/(gcat·h)的条件下,CO转化率为99.2%。此外,与传统液相制备催化剂技术相比,球磨法具有污染小、成本低、易操作等特点,有利于节约能源。Abstract: The NiO/CeO2 catalytic materials were prepared with ball milling method using CeO2 as carrier and Ni as active component with good CO oxidation performance at low temperature. The catalysts were characterized by XRD, BET, H2-TPR, XRF and XPS. The effects of nickel content on the structure and low temperature oxidation performance of CO were investigated. The results show that the Ni-Ce ratio mainly affects the number of lattice oxygen vacancies and the interaction between the active ingredient and the carrier. Among them, When the Ni/Ce mole ratios is 1∶9, there are more oxygen vacancies on the surface of the catalyst, so it shows excellent catalytic performance. When the reaction temperature is 200 ℃, oxygen excess coefficient is 5 and the total space velocity is 60000 mL/(gcat·h), the CO conversion reaches 99.2%. In addition, compared with the traditional liquid phase catalyst preparation technology, ball milling has the advantages of low pollution, low cost and easy operation, which is beneficial to save energy.
-
Key words:
- method of ball mill /
- CO oxidation /
- CeO2 /
- low temperature
-
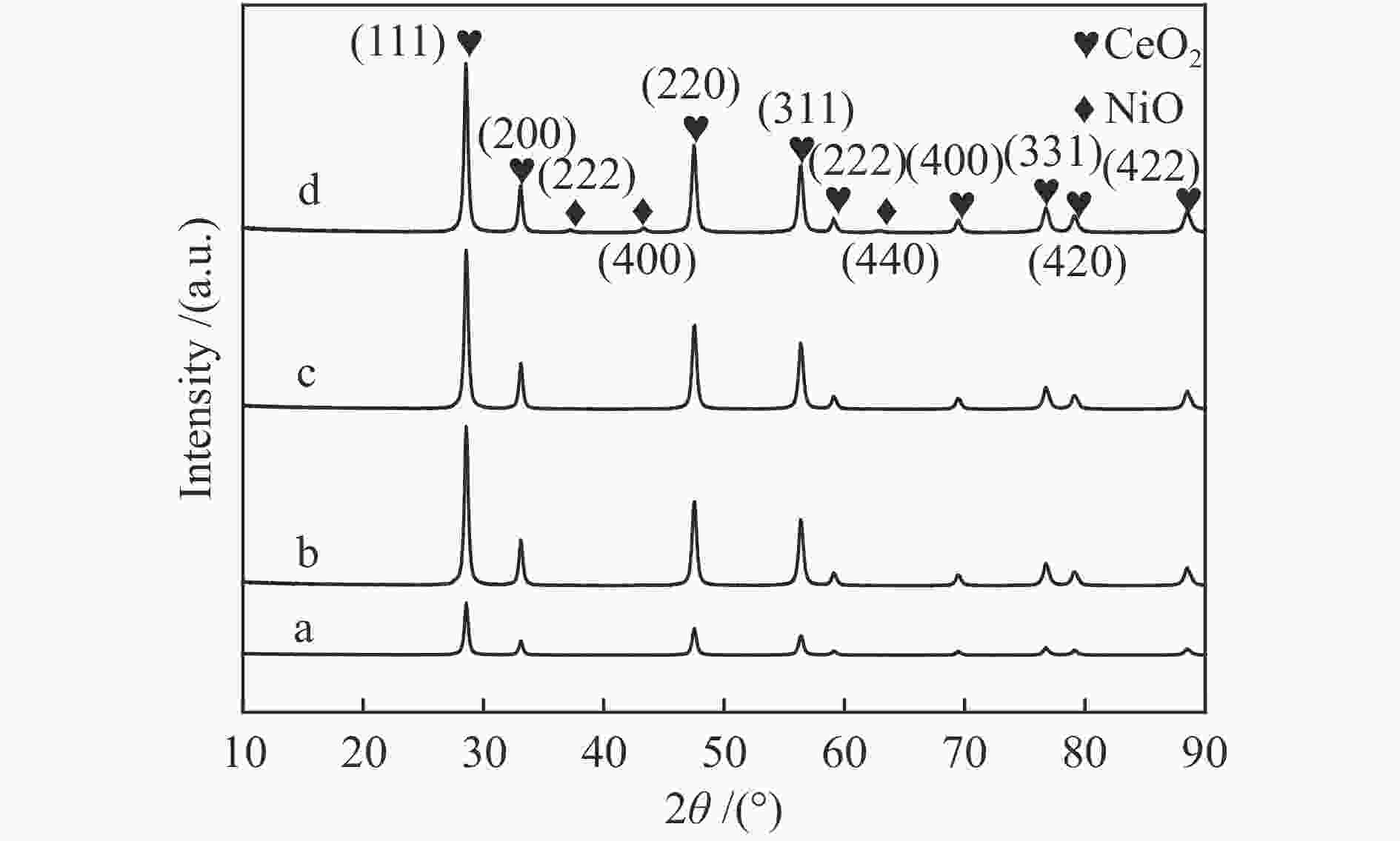
表 1 NiO/CeO2催化剂的XRD分析
Table 1 XRD data of NiO/CeO2 catalysts
Sample Cell parameter/nm Crystallite size of CeO2/nm CeO2 0.5410 22.0 NC-0.5 0.5408 21.9 NC-1 0.5399 20.9 NC-2 0.5409 20.8 表 2 NiO/CeO2催化剂的BET分析
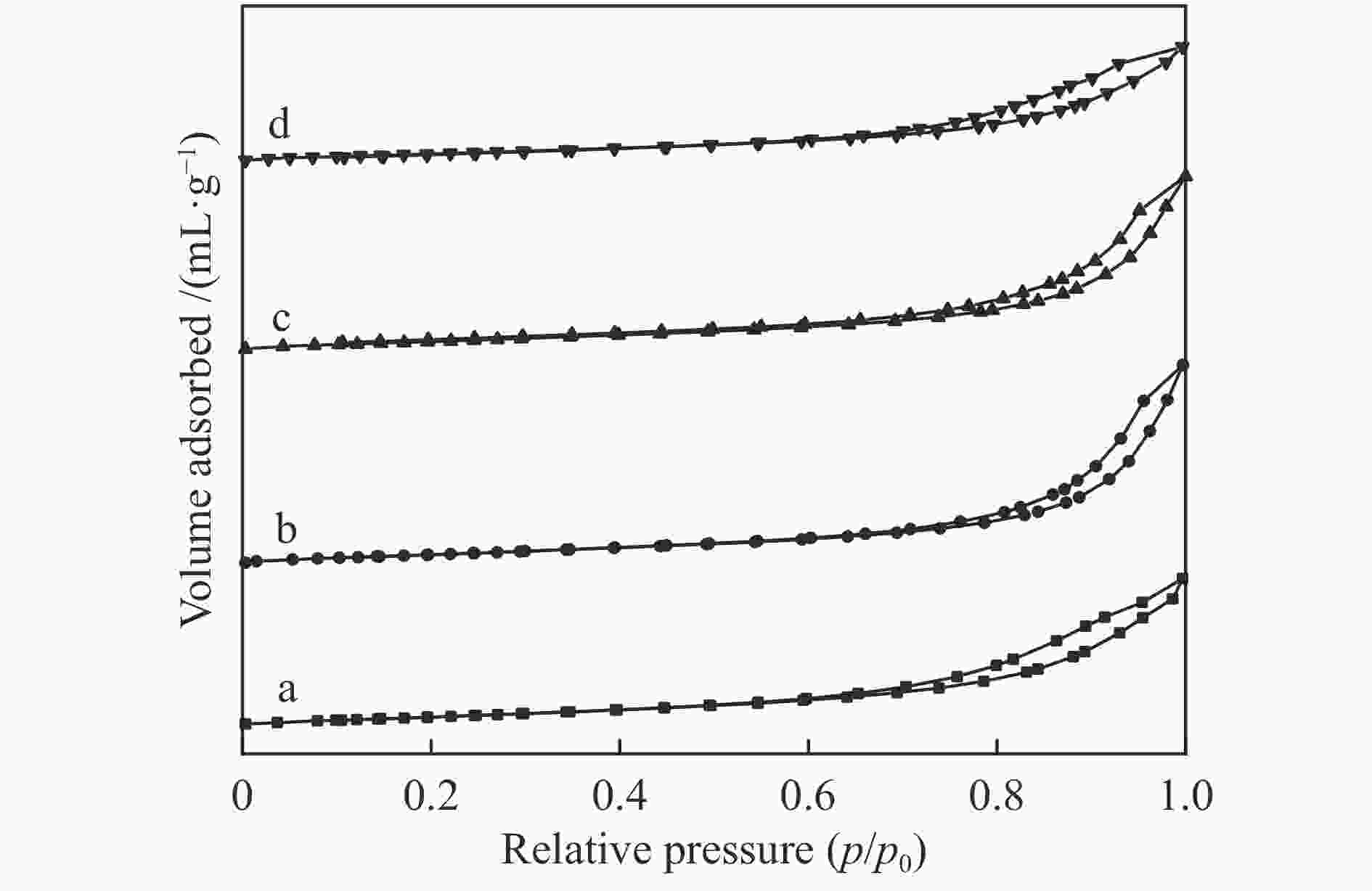
Table 2 BET analysis of NiO/CeO2 catalyst
Sample SBET/(m2·g−1) Pore size/nm CeO2 24.3 9.49 CeO2 34.6 9.46 NC-0.5 36.1 12.85 NC-1 34.5 11.90 NC-2 32.3 9.18 表 3 NiO/CeO2催化剂的Ni元素含量
Table 3 Ni element content of NiO/CeO2 catalyst
Sample Target content of Ni/% Ni content/%a CeO2 − − NC-0.5 2.20 2.08 NC-1 4.06 3.76 NC-2 9.80 9.85 a: determined by XRF experiment 表 4 NiO/CeO2催化剂还原峰位置
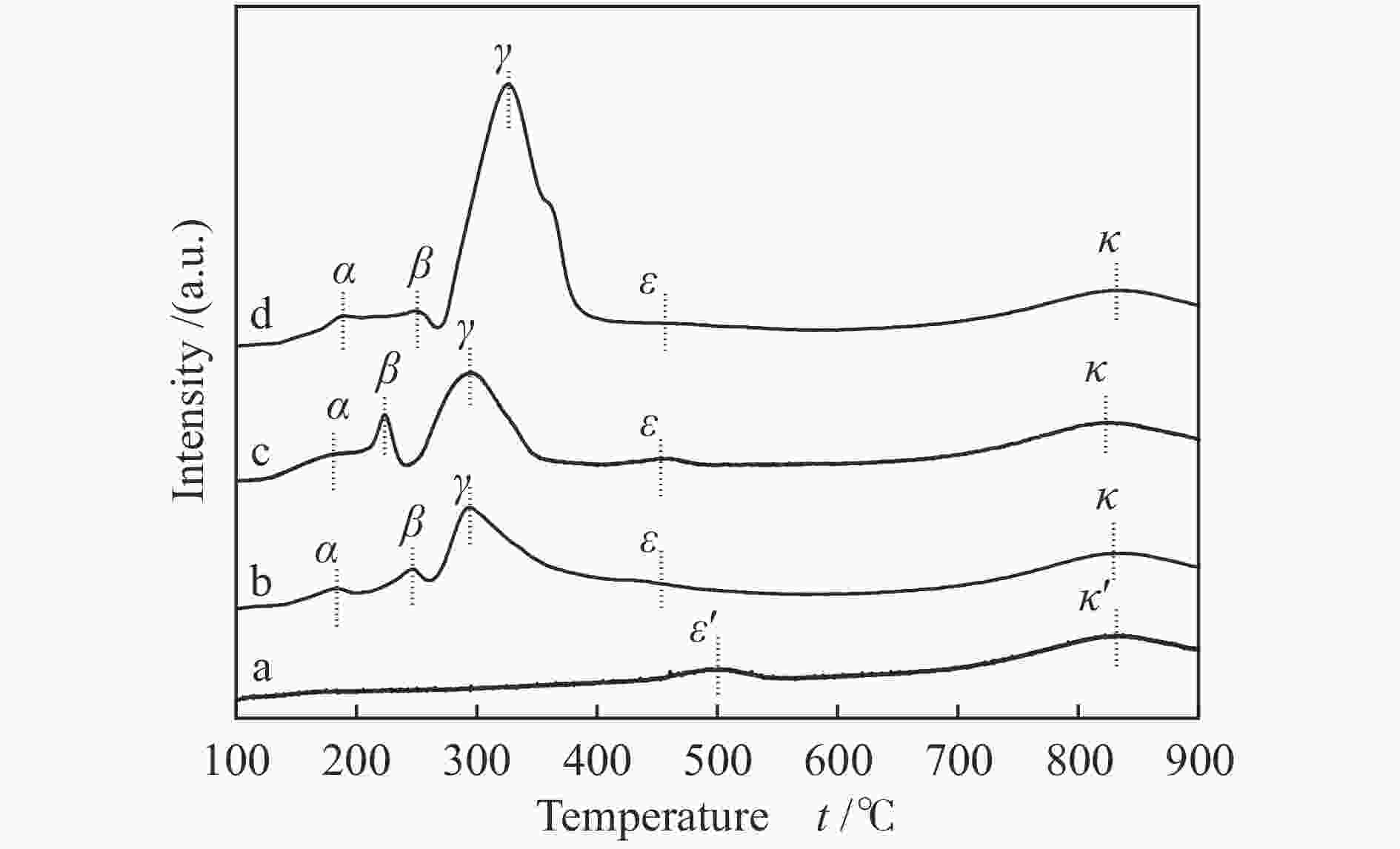
Table 4 Location of reduction peak of NiO/CeO2 catalyst
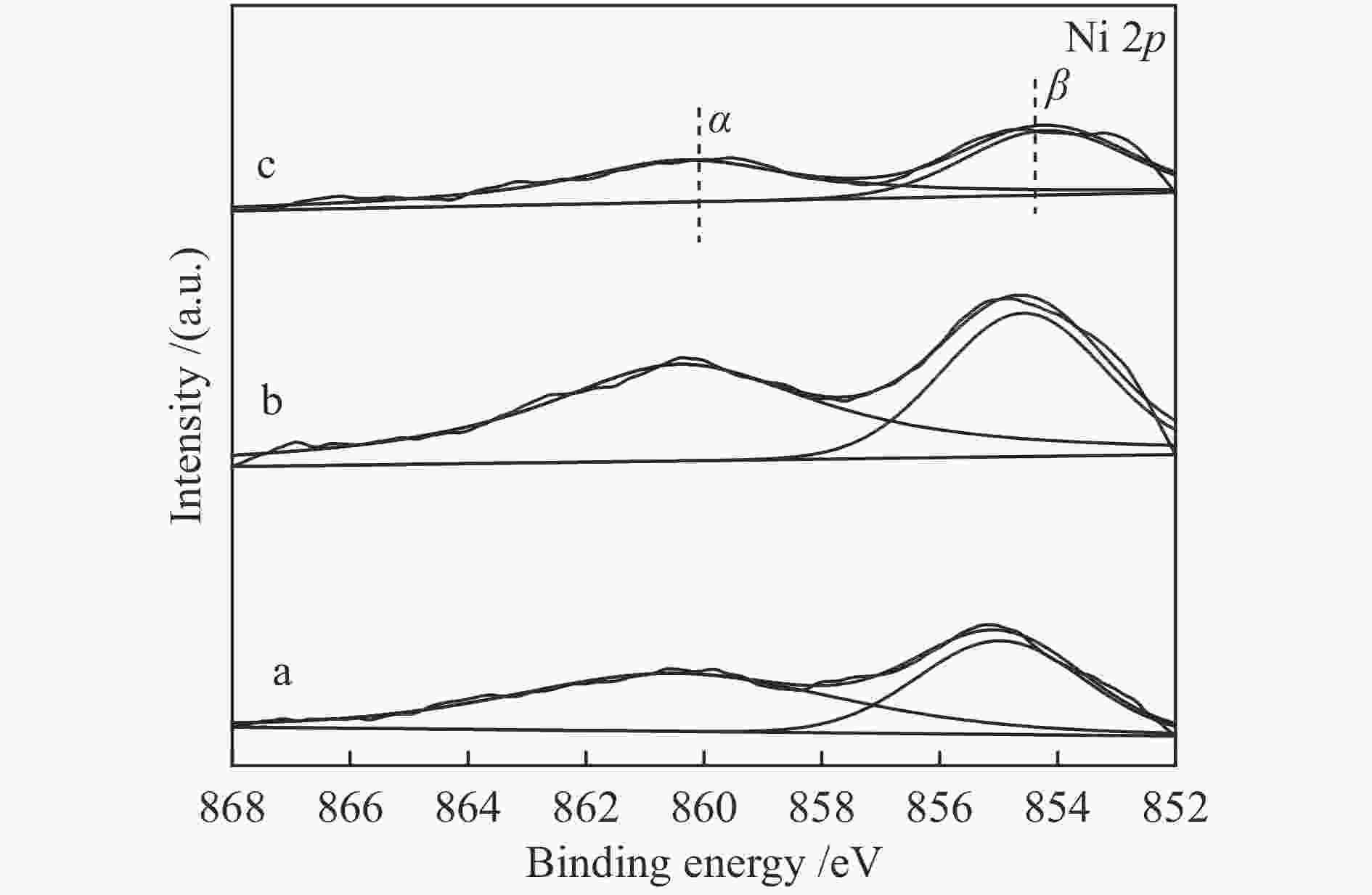
Sample Location of reduction peak /℃ α β γ ε/ε, k/k, CeO2 − − − 500 833 NC-0.5 182 246 294 453 831 NC-1 180 223 294 455 824 NC-2 187 249 327 456 830 表 5 NiO/CeO2催化剂中晶格氧含量
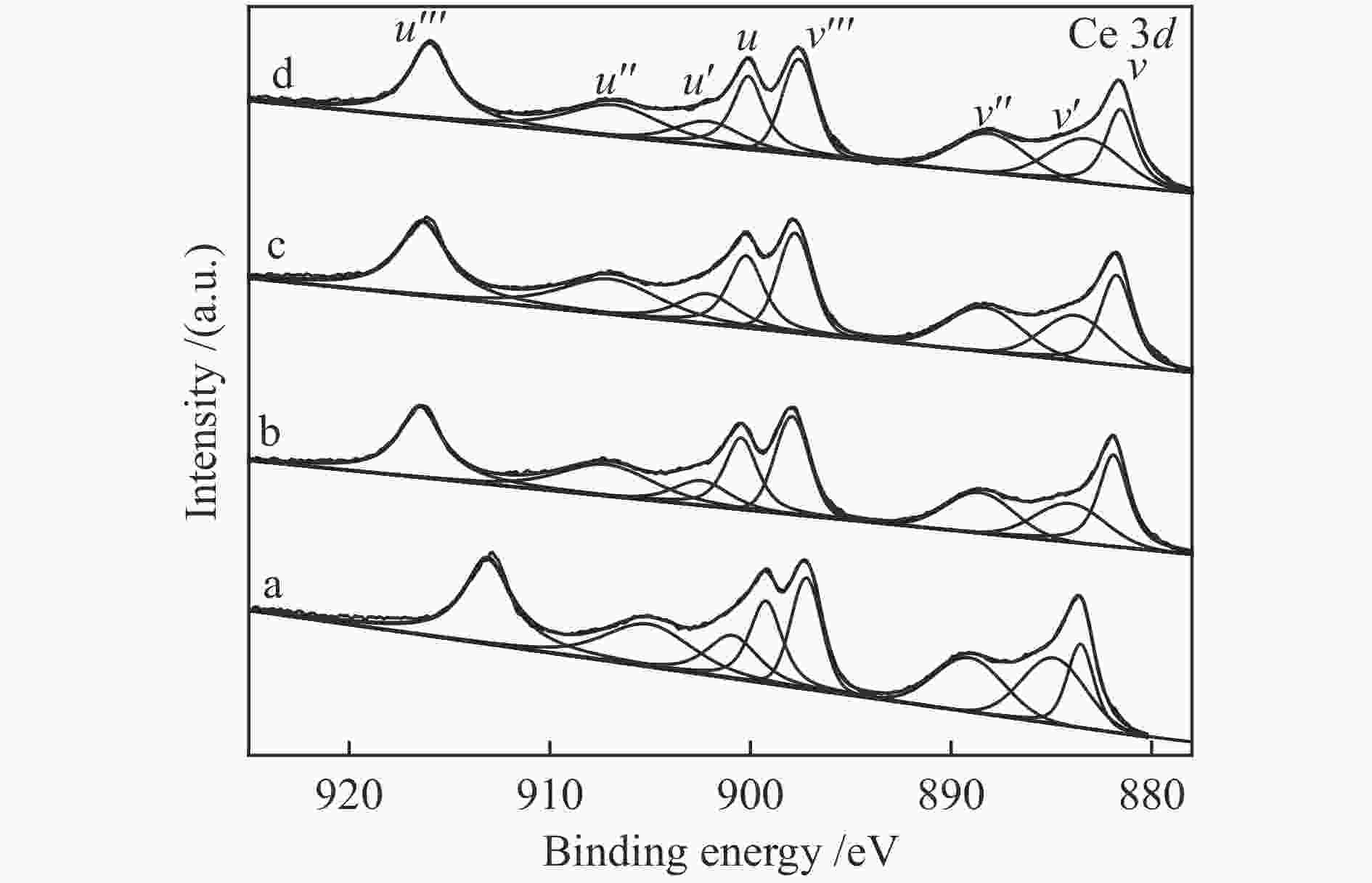
Table 5 Lattice oxygen content in NiO/CeO2 catalyst
Catalyst Olat/(Olat+Oads)/% 0.5-NiO/CeO2 60 1-NiO/CeO2 77 2-NiO/CeO2 55 -
[1] SETH D, NIU M. Hydrogen futures: Toward a sustainable energy system[J]. Int J Hydrogen Energy,2002,27(3):235−264. doi: 10.1016/S0360-3199(01)00131-8 [2] 王子良, 李瑞军, 解东来. 一种CO优先氧化装置的实验研究[J]. 化工进展,2012,31(3):523−527.WANG Zi-liang, LI Rui-jun, XIE Dong-lai. An experimental study on a CO preferential oxidation reactor[J]. J Energy Chem,2012,31(3):523−527. [3] PATRICK M, DAMON H. Hydrogen's role in an uncertain energy future[J]. Int J Hydrogen Energy,2009,34(1):31−39. doi: 10.1016/j.ijhydene.2008.10.060 [4] AGUILA G, GRACIS F, AMYA P. CuO and CeO2 catalysts supported on Al2O3, ZrO2 and SiO2 in the oxidation of CO at low temperature[J]. Appl Catal A: Gen,2008,343(1):16−24. [5] HAN S W, KIM D H, JEONG M G, PARK K J, KIM Y D. CO oxidation catalyzed by NiO supported on mesoporous Al2O3 at room temperature[J]. Chem Eng J,2016,283(1):992−998. [6] LU R, HE L, WANG Y, GAO X Q, LI W C. Promotion effects of nickel-doped Al2O3-nanosheet-supported Au catalysts for CO oxidation[J]. Chin J Catal,2020,41(2):350−356. doi: 10.1016/S1872-2067(19)63439-X [7] BANSMANN J, KUCEROVA G, ABDEL-MAGEED A M, EL-MOEMEN A A, BEHM R J. Influence of re-activation and ongoing CO oxidation reaction on the chemical and electronic properties of Au on Au/CeO2 catalyst: A XANES study at the Au Ledge[J]. J Electron Spectr,2017,220(34):86−90. [8] KAST P, FRIEDRICH M, GIRGSDIES F, KRÖHNERT J, TESCHNER D, LUNKENBEIN T, BEHRENS M, SCHLöGL R. Strong metal support interaction and alloying in Pd/ZnO catalysts for CO oxidation[J]. Catal Today,2015,260(62):21−31. [9] LEUNG E, SHIMIZU A, BARMAK K, FARRAUTO R. Copper oxide catalyst supported on niobium oxide for CO oxidation at low temperatures[J]. Catal Commun,2017,97(87):42−46. [10] WANG Y Z, ZHAO Y X, GAO C G, LIU D S. Preparation and catalytic performance of Co3O4 catalysts for low-temperature CO oxidation[J]. Catal Lett,2007,116(3/4):136−142. doi: 10.1007/s10562-007-9099-4 [11] LEE J, RYOU Y, KIM J, CHAN X J, KIM T, KIM D H. Influence of the defect concentration of ceria onthe Pt dispersion and the CO oxidation activity of Pt/CeO2[J]. J Phys Chem C,2018,122(9):4972−4983. doi: 10.1021/acs.jpcc.8b00254 [12] LU R, HE L, WANG Y, GAO X Q, LI W C. Promotion effects of nickel-doped Al2O3-nanosheet-supported Au catalysts for CO oxidation[J]. Chin J Catal,2019,02(12):350−358. [13] SUN J, ZHANG L, GE C, TANG C, DONG L. Comparative study on the catalytic CO oxidation properties of CuO/CeO2 catalysts prepared by solid state and wet impregnation[J]. Chin J Catal,2014,35(8):1347−1358. [14] 李树娜, 石奇, 李小军, 方振华, 孙平, 周跃花, 张杏梅, 杨晓慧. 金属掺杂Ce-M(M=Fe、Ni和Cu)催化剂的CO低温氧化性能研究[J]. 燃料化学学报,2017,45(6):707−713. doi: 10.3969/j.issn.0253-2409.2017.06.009LI Shu-na, SHI Qi, LL Xiao-jun, FANG Zhen-hua, SUN Ping, ZHOU Yue-hua, ZHANG Xing-mei, YANG Xiao-hui. Low temperature CO oxidation over the ceria oxide catalysts doped with Fe, Ni and Cu[J]. J Fuel Chem Technol,2017,45(6):707−713. doi: 10.3969/j.issn.0253-2409.2017.06.009 [15] DESHETTI J, VENKATASWAMY P, VICTORIA E C, REDDY B M, BHARGAVA S K. Low-temperature CO oxidation over manganese, cobalt, and nickel doped CeO2 nanorods[J]. RSC,2016,6(84):80541−80547. [16] TANGA C J, LI J C, YAO X J, SUN J F, CAO Y, ZHANG L, GAO F, DENG Y, DONG L. Mesoporous NiO-CeO2 catalysts for CO oxidation: Nickel content effect and mechanism aspect[J]. Appl Catal A: Gen,2015,494(1):77−86. [17] 肖卫明. 过渡金属催化剂的球磨法制备及其催化CO氧化性能研究[D]. 南昌: 南昌大学研究生院, 2018.XIAO Wei-ming. Transition metal catalyst prepared by ball milling and their activities for the CO oxidation[D]. Nanchang: Graduate School of Nanchang University, 2018. [18] ZHENG Y, LI K, WANG H, ZHU X, WEI Y G, ZHEBG M, WANG Y H. Enhanced activity of CeO2-ZrO2 solid solutions for chemical-looping reforming of methane via tuning the macroporous structure[J]. Energy Fuels,2016,30(1):638−647. doi: 10.1021/acs.energyfuels.5b02151 [19] ASHOK J, ANG M. L, KAWI S. Enhanced activity of CO2 methanation over Ni/CeO2-ZrO2 catalysts: Influence of preparation methods[J]. Catal Today,2017,281(2):304−311. [20] MAHAMMADUNNIS S, REDDY P M K, LINGAIAH N, SUBRAHMANYAM C. NiO/Ce1-xNixOy as an alternative to noble metal catalysts for CO oxidation[J]. Catal Sci Technol,2013,3(3):730−736. doi: 10.1039/C2CY20641B [21] AHMED S, KRUMPELT M. Hydrogen from hydrocarbon fuels for fuel cells[J]. Int J Hydrogen Energy,2001,26(4):291−301. doi: 10.1016/S0360-3199(00)00097-5 [22] SANGSEFIDI F S, SALAVATI-NIASARI M, SHABANI-NOOSHABADI M. Characterization of hydrogen storage behavior of the as-synthesized p-type NiO/n-type CeO2 nanocomposites by carbohydrates as a capping agent: The influence of morphology[J]. Int J Hydrogen Energy,2018,43(1):14557−14568. [23] LI J, ZHU P F, ZHOU R X. Effect of the preparation method on the performance of CuO-MnOx-CeO2 catalysts for selective oxidation of CO in H2-rich streams[J]. J Power Sources,2011,196(22):9590−9598. doi: 10.1016/j.jpowsour.2011.07.052 [24] LIU L J, YAO Z J, DENG Y, GAO F, LIU B, DONG L. Morphology and crystal‐plane effects of nanoscale ceria on the activity of CuO/CeO2 for NO reduction by CO[J]. ChemCatChem,2011,3(6):978−989. doi: 10.1002/cctc.201000320 [25] 闫宁, 周安宁, 张亚刚, 杨志远, 贺新福, 张亚婷. CeO2的形貌特征对Ni/CeO2催化剂CO甲烷化性能的影响[J]. 燃料化学学报,2020,48(4):466−475. doi: 10.3969/j.issn.0253-2409.2020.04.010YAN Ning, ZHOU An-ning, ZHANG Ya-gang, YANG Zhi-yuan, HE Xin-fu, ZHANG Ya-ting. Morphologic effect of CeO2 on the catalytic performance of Ni /CeO2 in CO methanation[J]. J Fuel Chem Technol,2020,48(4):466−475. doi: 10.3969/j.issn.0253-2409.2020.04.010 [26] LIN H K, CHIU H C, HSIN C T, CHIEN S H, WANG C B. Synthesis characterization and catalytic oxidation of carbon monoxide over cobalt oxide[J]. Catal Lett,2003,88(1):169−174. [27] LUSTEMBERG P G, FERIA L, GANDUGLIA-PIROVANO M V. Single Ni sites supported on CeO2(111) reveal cooperative effects in the water-gas shift reaction[J]. J Phys Chem,2018,3(1):2−28. -




 下载:
下载: