Preparation of iron carbide and its application in Fischer-Tropsch synthesis
-
摘要: 费托合成是合成气转化为清洁液体燃料和高附加值化学品的最佳途径和发展方向。铁基催化剂在费托合成反应中应用广泛。研究表明,碳化铁是Fe基费托合成催化剂的活性相,近年来,制备碳化铁直接用于费托合成已成为Fe基费托合成催化剂研究的热点。本工作概述了碳化铁的物相类型,综述了碳化铁的制备方法(主要包括湿化学合成法、热处理渗碳法、铁基金属有机骨架材料热解法、溶胶凝胶法)及其在费托合成中的应用,并对碳化铁在费托合成中的应用前景和研究方向进行了展望。Abstract: Fischer-Tropsch synthesis is the best way and development direction for the conversion of syngas into clean liquid fuels and high value-added chemicals. Iron-based catalysts are widely used in Fischer-Tropsch synthesis reactions. Studies have shown that iron carbide is the active phase of iron-based Fischer-Tropsch synthesis catalysts. In recent years, the preparation of iron carbide for Fischer-Tropsch synthesis directly has become a hot research topic of iron-based Fischer-Tropsch synthesis catalysts. In this paper, the phase types of iron carbide are summarized, the preparation methods of iron carbide (mainly including wet chemical synthesis, heat treatment carburizing, iron-based organic framework material pyrolysis, sol-gel method) and its application in Fischer-Tropsch synthesis are reviewed, and the application prospect and research direction of iron carbide in Fischer-Tropsch synthesis are prospected.
-
Key words:
- Fischer-Tropsch synthesis /
- syngas /
- iron-based catalyst /
- iron carbide /
- preparation method
-
图 3 (a)和(b)纳米级MIL-88 B-NH3和氯化萘的透射电镜照片;(c)氯化萘的HRTEM照片;(d)N2吸附氯化萘的解吸等温线,插图是相应的孔径分布;(e)氯化萘的XRD谱图;(f)氯化萘的拉曼光谱谱图[52]
Figure 3 (a) and (b) TEM images of nano-scale MIL-88 B-NH3 and chlorinated naphthalene; (c) HRTEM image of chlorinated naphthalene; (d) desorption isotherm of N2 adsorption of chlorinated naphthalene, inset is the corresponding pore size distribution; (e) XRD pattern of chlorinated naphthalene; (f) Raman spectrum of chlorinated naphthalene[52]
(with permission from Springer Publications)
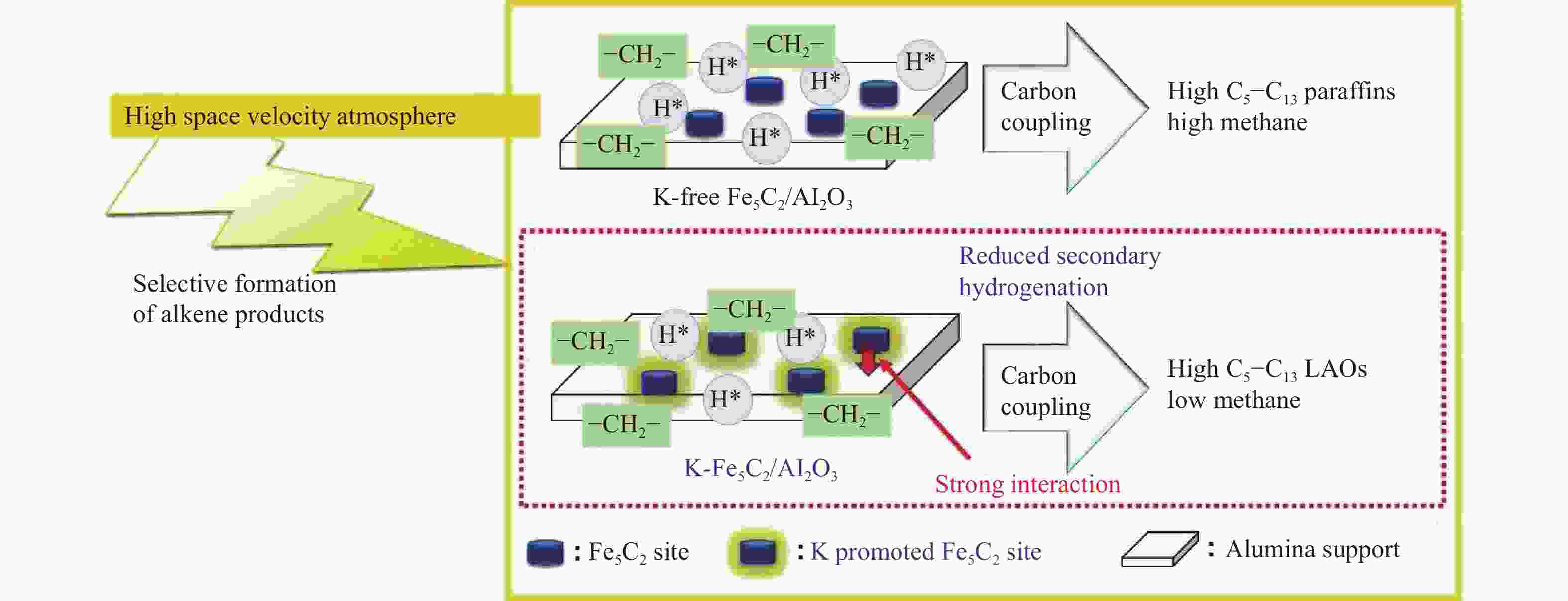
图 6 K-Fe5C2/Al2O3和无K的Fe5C2/Al2O3纳米催化剂上低碳烯烃(LAO)生成示意图[14]
Figure 6 Brief scheme for LAO formation over K-Fe5C2/Al2O3 and K-free Fe5C2/Al2O3 nanocatalysts: the combination of high space velocity condition and potassium as an alkali promoter on iron-carbide leads to decreased secondary hydrogenation of olefins and increased carbon coupling reaction[14]
(with permission from Elsevier Publications)
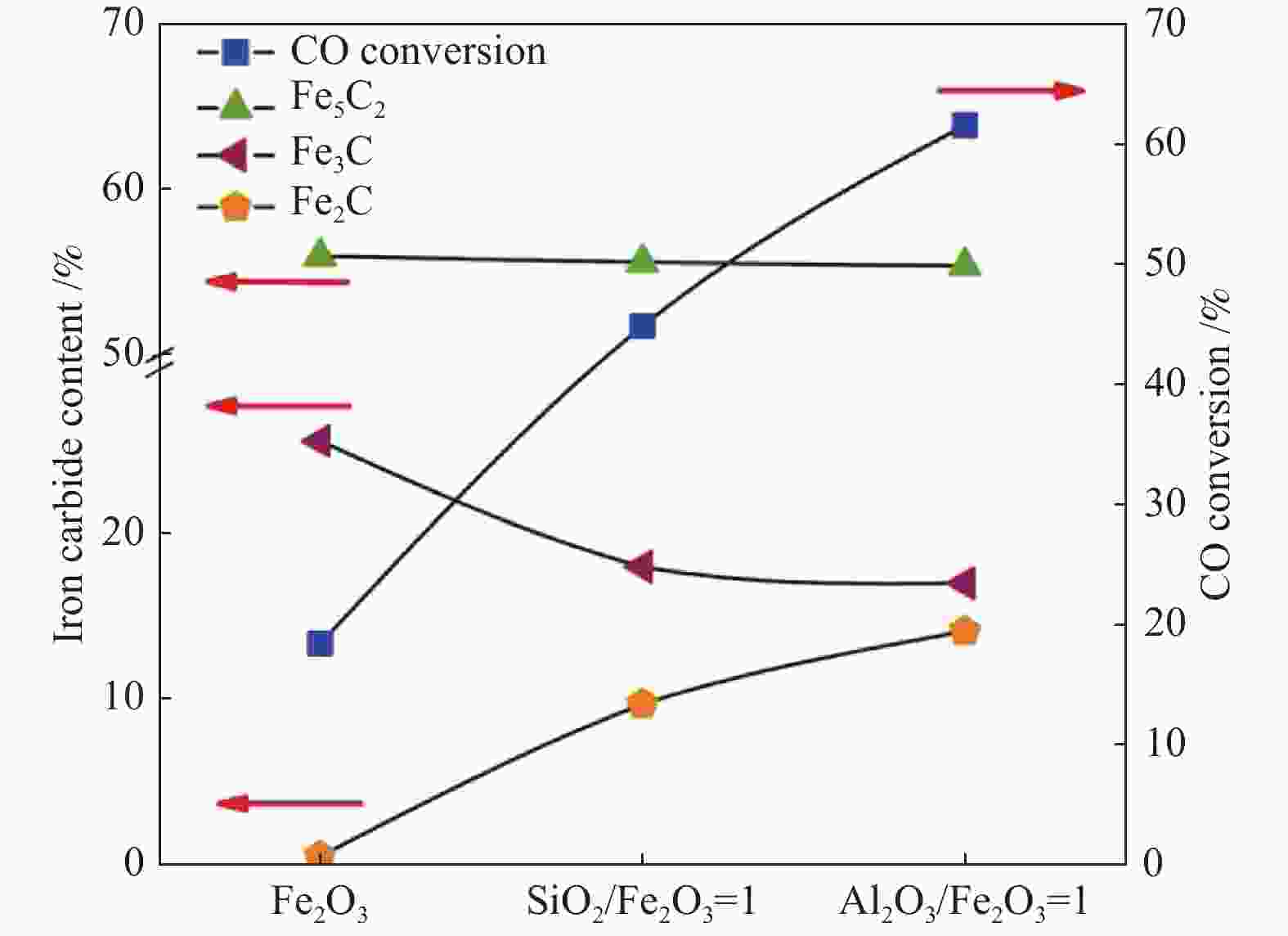
表 1 不同铁碳比制得的碳化铁的FTO反应性能[94]
Table 1 FTO reaction performance of iron carbide with different iron carbon ratios[94]
Catalyst CO conversion/% CO2 selectivity/% O/P Hydrocarbon distribution/% CH4 ${\rm{C} }^=_{2} -{\rm{C} }^=_{4}$ ${\rm{C} }^{0}_{2} -{\rm{C} }^{0}_{4}$ C5+ Fe/C=2∶6 94.79 38.75 1.45 26.09 34.76 24.00 15.30 Fe/C=2∶3 95.34 40.21 1.48 31.65 36.48 24.69 7.19 Fe/C=2∶1 95.94 39.97 1.56 29.29 37.17 23.84 9.70 H2/CO(volume ratio) = 1.5, 1 MPa, 320 ℃,GHSV = 12000 h−1 -
[1] LIN T, YU F, AN Y, QIN T, LI L, GONG K, ZHONG L, SUN Y. Cobalt carbide nanocatalysts for efficient syngas conversion to value-added chemicals with high selectivity[J]. Accounts Chem Res,2021,54(8):1961−1971. doi: 10.1021/acs.accounts.0c00883 [2] JIAO F, LI J, PAN X, XIAO J, LI H, MA H, WEI M, PAN Y, ZHOU Z, LI M, MIAO S, LI J, ZHU Y, XIAO D, HE T, YANG J, QI F, FU Q, BAO X. Selective conversion of syngas to light olefins[J]. Science,2016,351(6277):1065−1068. doi: 10.1126/science.aaf1835 [3] ZHONG L, YU F, AN Y, ZHAO Y, SUN Y, LI Z, LIN T, LIN Y, QI X, DAI Y, GU L, HU J, JIN S, SHEN Q, WANG H. Cobalt carbide nanoprisms for direct production of lower olefins from syngas[J]. Nature,2016,538(7623):84−87. doi: 10.1038/nature19786 [4] GALVIS H, BITTER J, KHARE C, RUITENBEEK M, DUGULAN A, JONG K. Supported iron nanoparticles as catalysts for sustainable production of lower olefins[J]. Science,2012,335(6070):835−838. doi: 10.1126/science.1215614 [5] SCHULZ H. Short history and present trends of Fischer-Tropsch synthesis[J]. Appl Catal A: Gen,1999,186(1):3−12. [6] KARRE A, DADYBURJOR D B. Review of iron-based catalysts with and without zeolite supports used in Fischer-Tropsch processes[J]. Chem Eng Commun,2021,1−21. [7] LIU Q, SHANG C, LIU Z. In situ active site for Fe-catalyzed Fischer-Tropsch synthesis: Recent progress and future challenges[J]. J Phys Chem Lett,2022,13(5):3342−3352. [8] GARONA H A, CAVALCANTI F M, DE A T F, SCHMAL M, ALVES R M B. Evaluation of Fischer-Tropsch synthesis to light olefins over Co- and Fe-based catalysts using artificial neural network[J]. J Clean Prod,2021,321(80):129003. [9] BROOS R J P, KLUMPERS B, ZIJLSTRA B, FILOT I A W, HENSEN E J M. A quantum-chemical study of the CO dissociation mechanism on low-index Miller planes of circle minus-Fe3C[J]. Catal Today,2020,342:152−160. doi: 10.1016/j.cattod.2019.02.015 [10] LI T, WEN X D, YANG Y, LI Y W, JIAO H J. Mechanistic aspects of CO activation and C-C bond formation on the Fe/C- and Fe-terminated Fe3C(010) surfaces[J]. ACS Catal,2020,10(1):877−890. doi: 10.1021/acscatal.9b04433 [11] REN J, AI N, YU Y. Insight into the Fischer-Tropsch mechanism on hcp-Fe7C3 (211) by density functional theory: the roles of surface carbon and vacancies[J]. RSC Adv,2021,11(55):34533−34543. doi: 10.1039/D1RA06396K [12] HERRANZ T, ROJAS S, PEREZ F J, OJEDA M, TERREROS P, FIERRO J L G. Genesis of iron carbides and their role in the synthesis of hydrocarbons from synthesis gas[J]. J Catal,2006,243(1):199−211. doi: 10.1016/j.jcat.2006.07.012 [13] 王玲玉, 韩文锋, 刘化章, 杨霞珍. 合成气制低碳烯烃铁催化剂活性相的研究进展[J]. 化工进展,2019,38(11):4949−4955. doi: 10.16085/j.issn.1000-6613.2019-0254WANG Ling-yu, HAN Wen-feng, LIU Hua-zhang, YANG Xia-zhen. Research progress on active phase of iron-based catalysts for light olefins synthesis from syngas[J]. Chem Ind Eng Prog,2019,38(11):4949−4955. doi: 10.16085/j.issn.1000-6613.2019-0254 [14] PARK J C, JANG S, RHIM G B, LEE J, CHOI H, JEONG H D, YOUN M, LEE D, KOO K Y, KANG S, YANG J, LEE H, JUNG H, KIM C S, CHUN D. A durable nanocatalyst of potassium-doped iron-carbide/alumina for significant production of linear alpha olefins via Fischer-Tropsch synthesis[J]. Appl Catal A: Gen,2018,564:190−198. doi: 10.1016/j.apcata.2018.07.037 [15] CHANG Q, ZHANG C H, LIU C W, WEI Y X, CHERUVATHUR A V, DUGULAN A I, NIEMANTSVERDRIET J W, LIU X W, HE Y R, QING M, ZHENG L R, YUN Y F, YANG Y, LI Y W. Relationship between iron carbide phases (epsilon-Fe2C, Fe7C3, and chi-Fe5C2) and catalytic performances of Fe/SiO2 Fischer-Tropsch catalysts[J]. ACS Catal,2018,8(4):3304−3316. doi: 10.1021/acscatal.7b04085 [16] WANG P, CHEN W, CHIANG F, DUGULAN A I, SONG Y, PESTMAN R, ZHANG K, YAO J, FENG B, MIAO P, XU W, HENSEN E J M. Synthesis of stable and low-CO2 selective ε-iron carbide Fischer-Tropsch catalysts[J]. Sci Adv,2018,4(10):eaau2947. doi: 10.1126/sciadv.aau2947 [17] CHIPMAN J. Thermodynamics and phase diagram of the Fe-C system[J]. Metall Mater Trans B,1972,3(1):55−64. doi: 10.1007/BF02680585 [18] NARAGHI R, SELLEBY M, AGREN J. Thermodynamics of stable and metastable structures in Fe-C system[J]. Calphad,2014,46:148−158. doi: 10.1016/j.calphad.2014.03.004 [19] LIBENSKA H, HANUS J, KOSUTOVA T, DOPITA M, KYLIAN O, CIESLAR M, CHOUKOUROV A, BIEDERMAN H. Plasma-based synthesis of iron carbide nanoparticles[J]. Plasma Process Polym,2020,17(11):2000105. doi: 10.1002/ppap.202000105 [20] LUO N, LI X, WANG X, YAN H, ZHANG C, WANG H. Synthesis and characterization of carbon-encapsulated iron/iron carbide nanoparticles by a detonation method[J]. Carbon,2010,48(13):3858−3863. doi: 10.1016/j.carbon.2010.06.051 [21] WANG D, CHEN B, DUAN X, CHEN D, ZHOU X. Iron-based Fischer-Tropsch synthesis of lower olefins: The nature of χ-Fe5C2 catalyst and why and how to introduce promoters[J]. J Energy Chem,2016,25(6):911−916. doi: 10.1016/j.jechem.2016.11.002 [22] LI S, YANG J, SONG C, ZHU Q, XIAO D, MA D. Iron carbides: Control synthesis and catalytic applications in COx hydrogenation and electrochemical HER[J]. Adv Mater,2019,31(50):1901796. doi: 10.1002/adma.201901796 [23] WANG X B, ZHU K, JU Y M, LI Y N, LI W, XU J J, HOU Y L. Iron carbides: Magic materials with magnetic and catalytic properties[J]. J Magn Magn Mater,2019,489(36):165432. [24] BROWNING L C, DEWITT T W, EMMETT P H. Equilibria in the systems Fe2C-Fe-CH4-H2 and Fe3C-Fe-CH4-H2[J]. J Am Chem Soc,1950,72(9):4211−4217. doi: 10.1021/ja01165a108 [25] NIEMANSVERDRIET J W, VAN K A M, VAN D W L, VAN B H S. Behavior of metallic iron catalysts during Fischer-Tropsch synthesis studied with Mössbauer spectroscopy, X-ray diffraction, carbon content determination, and reaction kinetic measurements[J]. J Phys Chem,1980,84(25):3363−3370. doi: 10.1021/j100462a011 [26] LV Z Q, SUN S H, JIANG P, WANG B Z, FU W T. First-principles study on the structural stability, electronic and magnetic properties of Fe2C[J]. Comp Mater Sci,2008,42(4):692−697. doi: 10.1016/j.commatsci.2007.10.007 [27] BERNAS H, CAMPBELL I A, FRUCHART R. Electronic exchange and the Mössbauer effect in iron-based interstitial compounds[J]. J Phys Chem Solids,1967,28(1):17−24. doi: 10.1016/0022-3697(67)90192-8 [28] ZHAO S, LIU X, HUO C, LI Y, WANG J, JIAO H. Determining surface structure and stability of ε-Fe2C, χ-Fe5C2, θ-Fe3C and Fe4C phases under carburization environment from combined DFT and atomistic thermodynamic studies[J]. Catal Struct React,2015,1(1):44−60. [29] YANG W, REHMAN S, CHU X, HOU Y, GAO S. Transition metal (Fe, Co and Ni) carbide and nitride nanomaterials: Structure, chemical synthesis and applications[J]. ChemNanoMat,2015,1(6):376−398. doi: 10.1002/cnma.201500073 [30] JACK K H, WILD S. Nature of χ-carbide and its possible occurrence in steels[J]. Nature,1966,212(5059):248−250. [31] HAMMERL C, KÖNIGER A, RAUSCHENBACH B. Electronmicroscopical study of the formation of iron carbide phases after high-fluence carbon ion implantation into iron at low temperatures[J]. J Mater Res,1998,13(9):2614−2622. doi: 10.1557/JMR.1998.0365 [32] HERBSTEIN F H, SNYMAN J A. Identification of Eckstrom-Adcock iron carbide as Fe7C3[J]. Inorg Chem,1964,3(6):894−896. doi: 10.1021/ic50016a026 [33] SICHEN D, SEETHARAMAN S, STAFFANSSON L I. Some phase-diagram aspects of the manganese-carbon system[J]. Metall Trans B,1989,20(5):747−754. doi: 10.1007/BF02655933 [34] BAUER E, FRANTZ C, LE CAER G, HEIMAN N. Formation of Fe7C3 and Fe5C2 type metastable carbides during the crystallization of an amorphous Fe75C25 alloy[J]. J Non-cryst Solids,1981,44(2):277−286. [35] HASAN M, YANG W, JU Y, CHU X, WANG Y, DENG Y, MAHMOOD N, HOU Y. Biocompatibility of iron carbide and detection of metals ions signaling proteomic analysis via HPLC/ESI-Orbitrap[J]. Nano Res,2017,10(6):1912−1923. doi: 10.1007/s12274-016-1375-4 [36] PRESCHER C, DUBROVINSKY L, BYKOVA E, KUPENKO I, GLAZYRIN K, KANTOR A, MCCAMMON C, MOOKHERJEE M, NAKAJIMA Y, MIYAJIMA N, SINMYO R, CERANTOLA V, DUBROVINSKAIA N, PRAKAPENKA V, RÜFFER R, CHUMAKOV A, HANFLAND M. High poisson's ratio of earth's inner core explained by carbon alloying[J]. Nat Geosci,2015,8(3):220−223. doi: 10.1038/ngeo2370 [37] CHIHI T, BOUHEMADOU A, REFFAS M, KHENATA R, GHEBOULI M A, GHEBOULI B, LOUAIL L. Structural, elastic and thermodynamic properties of iron carbide Fe7C3 phases: An ab initio study[J]. Chin J Phys,2017,55(3):977−988. doi: 10.1016/j.cjph.2016.12.014 [38] HOFER L J E, COHN E M, PEEBLES W C. The modifications of the carbide, Fe2C: Their properties and identification[J]. J Am Chem Soc,1949,71(1):189−195. doi: 10.1021/ja01169a048 [39] STENCEL J M, EKLUND P C, BI X X, DERBYSHIRE F J. Iron ultrafine particle catalysts formed by laser pyrolysis: synthesis, characterization and coal liquefactionactivity[J]. Catal Today,1992,15(2):285−306. doi: 10.1016/0920-5861(92)80180-U [40] YANG C, ZHAO H, HOU Y, MA D. Fe5C2 nanoparticles: A facile bromide-induced synthesis and as an active phase for Fischer-Tropsch synthesis[J]. J Am Chem Soc,2012,134(38):15814−15821. doi: 10.1021/ja305048p [41] YAO S, YANG C, ZHAO H, LI S, LIN L, WEN W, LIU J, HU G, LI W, HOU Y, MA D. Reconstruction of the wet chemical synthesis process: The case of Fe5C2 nanoparticles[J]. J Phys Chem C,2017,121(9):5154−5160. doi: 10.1021/acs.jpcc.7b00198 [42] YU G, SUN B, PEI Y, XIE S, YAN S, QIAO M, FAN K, ZHANG X, ZONG B. FexOy@C spheres as an excellent catalyst for Fischer-Tropsch synthesis[J]. J Am Chem Soc,2010,132(3):935−937. doi: 10.1021/ja906370b [43] HIRANO S I, TAJIMA S. Synthesis and magnetic properties of Fe5C2 by reaction of iron oxide and carbon monoxide[J]. J Mater Sci,1990,25(10):4457−4461. doi: 10.1007/BF00581108 [44] TAJIMA S, HIRANO S. Synthesis and magnetic properties of Fe7C3 particles with high saturation magnetization[J]. Jpn J Appl Phys,1990,29(4):662−668. [45] WANG R, WU B, LI Y. Synthesis of single-phase iron carbides and their adsorption performance[J]. Chin J Catal,2012,33(5):863−869. [46] MALINA O, JAKUBEC P, KAŠLÍK J, TUČEK J, ZBOŘIL R. A simple high-yield synthesis of high-purity Hägg carbide (χ-Fe5C2) nanoparticles with extraordinary electrochemical properties[J]. Nanoscale,2017,9(29):10440−10446. doi: 10.1039/C7NR02383A [47] TONG A, DAN D, YI C. Effect of reduction and carburization pretreatment on iron catalyst for synthesis of light olefins from CO hydrogenation[J]. Chem Res Chin Univ,2017,33(4):672−677. doi: 10.1007/s40242-017-6316-6 [48] NARKIEWICZ U, GUSKOS N, ARABCZYK W, TYPEK J, BODZIONY T, KONICKI W, GĄSIOREK G, KUCHAREWICZ I, ANAGNOSTAKIS E A. XRD, TEM and magnetic resonance studies of iron carbide nanoparticle agglomerates in a carbon matrix[J]. Carbon,2004,42(5):1127−1132. [49] WEZENDONK T A, SANTOS V P, NASALEVICH M A, WARRINGA Q S E, DUGULAN A I, CHOJECKI A, KOEKEN A C J, RUITENBEEK M, MEIMA G, ISLAM H-U, SANKAR G, MAKKEE M, KAPTEIJN F, GASCON J. Elucidating the nature of Fe species during pyrolysis of the Fe-BTC MOF into highly active and stable Fischer-Tropsch catalysts[J]. ACS Catal,2016,6(5):3236−3247. doi: 10.1021/acscatal.6b00426 [50] AN B, CHENG K, WANG C, WANG Y, LIN W. Pyrolysis of metal-organic frameworks to Fe3O4@Fe5C2 core-shell nanoparticles for Fischer-Tropsch synthesis[J]. ACS Catal,2016,6(6):3610−3618. doi: 10.1021/acscatal.6b00464 [51] SANTOS V P, WEZENDONK T A, JAÉN J J D, DUGULAN A I, NASALEVICH M A, ISLAM H, CHOJECKI A, SARTIPI S, SUN X, HAKEEM A A, KOEKEN A C J, RUITENBEEK M, DAVIDIAN T, MEIMA G R, SANKAR G, KAPTEIJN F, MAKKEE M, GASCON J. Metal organic framework-mediated synthesis of highly active and stable Fischer-Tropsch catalysts[J]. Nat Commun,2015,6(1):6451. doi: 10.1038/ncomms7451 [52] ZHAO S, YIN H, DU L, HE L, ZHAO K, CHANG L, YIN G, ZHAO H, LIU S, TANG Z. Carbonized nanoscale metal-organic frameworks as high performance electrocatalyst for oxygen reduction reaction[J]. ACS Nano,2014,8(12):12660−12668. doi: 10.1021/nn505582e [53] LI J, LI S, TANG Y, HAN M, DAI Z, BAO J, LAN Y. Nitrogen-doped Fe/Fe3C@graphitic layer/carbon nanotube hybrids derived from MOFs: efficient bifunctional electrocatalysts for ORR and OER[J]. Chem Commun,2015,51(13):2710−2713. doi: 10.1039/C4CC09062D [54] HOU Y, HUANG T, WEN Z, MAO S, CUI S, CHEN J. Metal-organic framework-derived nitrogen-doped core-shell-structured porous Fe/Fe3C@C nanoboxes supported on graphene sheets for efficient oxygen reduction reactions[J]. Adv Energy Mater,2014,4(11):1400337. doi: 10.1002/aenm.201400337 [55] GIORDANO C, ANTONIETTI M. Synthesis of crystalline metal nitride and metal carbide nanostructures by sol-gel chemistry[J]. Nano Today,2011,6(4):366−380. doi: 10.1016/j.nantod.2011.06.002 [56] GAO J, WANG X, PAN X, REN X, HAN Y, YANG X, YANG H. Facile synthesis of nanocrystalline Fe/Fe3C induced by bromide[J]. J Mater Sci: Mater Electron,2016,27(1):64−69. doi: 10.1007/s10854-015-3717-5 [57] SCHNEPP Z, WIMBUSH S C, ANTONIETTI M, GIORDANO C. Synthesis of highly magnetic iron carbide nanoparticles via a biopolymer route[J]. Chem Mater,2010,22(18):5340−5344. doi: 10.1021/cm101746z [58] NIKITENKO S I, KOLTYPIN Y, FELNER I, YESHURUN I, SHAMES A I, JIANG J Z, MARKOVICH V, GORODETSKY G, GEDANKEN A. Tailoring the properties of Fe-Fe3C nanocrystalline particles prepared by sonochemistry[J]. J Phys Chem B,2004,108(23):7620−7626. doi: 10.1021/jp036376r [59] DONG X L, ZHANG Z D, XIAO Q F, ZHAO X G, CHUANG Y C, JIN S R, SUN W M, LI Z J, ZHENG Z X, YANG H. Characterization of ultrafine γ-Fe(C), α-Fe(C) and Fe3C particles synthesized by arc-discharge in methane[J]. J Mater Sci,1998,33(7):1915−1919. doi: 10.1023/A:1004369708540 [60] LOBO GUERRERO A, REBOLLO B, GALLEGOS J H G, URIBE D B, ALTAMIRANO M A G, CABAL J G. Study of bamboo-type carbon nanotubes with magnetic iron carbide nanoparticles fabricated by a modified CVD method[J]. J Nanopart Res,2021,23(4):94. doi: 10.1007/s11051-021-05207-3 [61] SERGIIENKO R, SHIBATA E, AKASE Z, SUWA H, NAKAMURA T, SHINDO D. Carbon encapsulated iron carbide nanoparticles synthesized in ethanol by an electric plasma discharge in an ultrasonic cavitation field[J]. Mater Chem Phys,2006,98(1):34−38. doi: 10.1016/j.matchemphys.2005.08.064 [62] SUN X, GUTIERREZ A, YACAMAN M J, DONG X, JIN S. Investigations on magnetic properties and structure for carbon encapsulated nanoparticles of Fe, Co, Ni[J]. Mater Sci Eng A,2000,286(1):157−160. doi: 10.1016/S0921-5093(00)00628-6 [63] 高天鹏, 张恒, 李朝龙, 史浩飞, 杜春雷. CVD法原位制备Fe3C填充的碳纳米管及其静态磁性能[C]// 第七届全国青年表面工程学术会议暨重庆市第二届汽车摩托车摩擦学材料先进技术与应用推进会论文集, 重庆, 2015: 89−94.GAO Tian-peng, ZHANG Heng, LI Chao-long, SHI Hao-fei, DU Chun-lei. In-situ preparation of Fe3C-filled carbon nanotubes by CVD method and its static magnetic properties[C]//The 7th National Youth Surface Engineering Conference and the 2nd Chongqing Automobile and Motorcycle Tribological Materials Advanced Technology and Application Promotion Conference Papers, Chongqin, 2015: 89−94. [64] DAVYDOV V, RAKHMANINA A, KIREEV I, ALIEVA I, ZHIRONKINA O, STRELKOVA O, DIANOVA V, SAMANI T D, MIRELES K, YAHIA L H, UZBEKOV R, AGAFONOV V, KHABASHESKU V. Solid state synthesis of carbon-encapsulated iron carbide nanoparticles and their interaction with living cells[J]. J Mater Chem B,2014,2(27):4250−4261. doi: 10.1039/C3TB21599G [65] DRY M E. Fischer-Tropsch reactions and the environment[J]. Appl Catal A: Gen,1999,189(2):185−190. doi: 10.1016/S0926-860X(99)00275-6 [66] AMELSE J A, SCHWARTZ L H, BUTT J B. Iron alloy Fischer-Tropsch catalysts: III. Conversion dependence of selectivity and water-gas shift[J]. J Catal,1981,72(1):95−110. doi: 10.1016/0021-9517(81)90081-6 [67] SCHLINGER W G. Coal gasification development and commercialization of the texaco coal gasification process[J]. Int J Energ Res,1980,4(2):127−136. doi: 10.1002/er.4440040205 [68] 李宇萍, 王铁军, 马隆龙, 吴创之, 定明月. Hβ改性Co/SiO2对费托合成航空燃油类烃的影响[J]. 无机材料学报,2014,29(6):599−604.LI Yu-ping, WANG Tie-jun, MA Long-long, WU Chuang-zhi, DING Ming-yue. Hβ modified Co/SiO2 catalysts for Fischer-Tropsch synthesis of jet fuel-range hydrocarbons[J]. J Inorg Mater,2014,29(6):599−604. [69] KHODAKOV A Y, CHU W, FONGARLAND P. Advances in the development of novel cobalt Fischer-Tropsch catalysts for synthesis of long-chain hydrocarbons and clean fuels[J]. Chem Rev,2007,107(5):1692−1744. doi: 10.1021/cr050972v [70] RAHIMPOUR M R, ELEKAEI H. A comparative study of combination of Fischer-Tropsch synthesis reactors with hydrogen-permselective membrane in GTL technology[J]. Fuel Process Technol,2009,90(6):747−761. doi: 10.1016/j.fuproc.2009.02.011 [71] DAVIS B H. Overview of reactors for liquid phase Fischer-Tropsch synthesis[J]. Catal Today,2002,71(3):249−300. [72] 王玲玉. 合成气制低碳烯烃催化剂的研究[D]. 杭州: 浙江工业大学, 2019.WANG Ling-yu. The research of catalysts for syngas to lower olefins[D]. Hangzhou: Zhejiang University of Technology, 2019. [73] DE S E, WECKHUYSEN B M. The renaissance of iron-based Fischer-Tropsch synthesis: on the multifaceted catalyst deactivation behaviour[J]. Chem Soc Rev,2008,37(12):2758−2781. doi: 10.1039/b805427d [74] DE S E, BEALE A M, NIKITENKO S, WECKHUYSEN B M. Local and long range order in promoted iron-based Fischer-Tropsch catalysts: A combined in situ X-ray absorption spectroscopy/wide angle X-ray scattering study[J]. J Catal,2009,262(2):244−256. doi: 10.1016/j.jcat.2008.12.021 [75] LI S, DING W, MEITZNER G D, IGLESIA E. Spectroscopic and transient kinetic studies of site requirements in iron-catalyzed Fischer-Tropsch synthesis[J]. J Phys Chem B,2002,106(1):85−91. doi: 10.1021/jp0118827 [76] LIU Y, CHEN J, BAO J, ZHANG Y. Manganese-modified Fe3O4 microsphere catalyst with effective active phase of forming light olefins from syngas[J]. ACS Catal,2015,5(6):3905−3909. doi: 10.1021/acscatal.5b00492 [77] 王祎凡. θ-Fe3C催化剂上费托合成机理的密度泛函理论计算[D]. 天津: 天津大学, 2017.WANG Yi-fan. Density functional theory study on the mechanism of the Fischer-Tropsch synthes over θ-Fe3C catalyst[D]. Tianjin: Tianjin University, 2017. [78] GAO W, GAO R, ZHAO Y, PENG M, SONG C, LI M, LI S, LIU J, LI W, DENG Y, ZHANG M, XIE J, HU G, ZHANG Z, LONG R, WEN X, MA D. Photo-driven syngas conversion to lower olefins over oxygen-decorated Fe5C2 catalyst[J]. Chem,2018,4(12):2917−2928. doi: 10.1016/j.chempr.2018.09.017 [79] 周立进, 王磊, 黄慧慧, 管国锋. 费托合成工艺研究进展[J]. 石油化工,2012,41(12):1429−1436. doi: 10.3969/j.issn.1000-8144.2012.12.017ZHOU Li-jin, WANG Lei, HUANG Hui-hui, GUAN Guo-feng. Research progress in Fischer-Tropsch synthesis process[J]. Petrochem Technol,2012,41(12):1429−1436. doi: 10.3969/j.issn.1000-8144.2012.12.017 [80] XU K, SUN B, LIN J, WEN W, PEI Y, YAN S, QIAO M, ZHANG X, ZONG B. ε-Iron carbide as a low-temperature Fischer-Tropsch synthesis catalyst[J]. Nat Commun,2014,5(1):5783. doi: 10.1038/ncomms6783 [81] FU X, YU W, MA C, LIN J, SUN S, LI S, REN P, JIA F, LI M, WANG W, WANG X, JIA C, WU K, SI R, YAN C. Supported Fe2C catalysts originated from Fe2N phase and active for Fischer-Tropsch synthesis[J]. Appl Catal B: Environ,2021,284:119702. doi: 10.1016/j.apcatb.2020.119702 [82] ZHAO H, LIU J, YANG C, YAO S, SU H, GAO Z, DONG M, WANG J, RYKOV A I, WANG J, HOU Y, LI W, MA D. Synthesis of iron-carbide nanoparticles: Identification of the active phase and mechanism of Fe-based Fischer-Tropsch synthesis[J]. CCS Chemistry,2021,3(11):2712−2724. doi: 10.31635/ccschem.020.202000555 [83] LU F, CHEN X, LEI Z, WEN L, ZHANG Y. Revealing the activity of different iron carbides for Fischer-Tropsch synthesis[J]. Appl Catal B: Environ,2021,281:119521. doi: 10.1016/j.apcatb.2020.119521 [84] 吴彦丽. 高温与低温费托合成联产系统过程分析及产品设计[D]. 太原: 太原理工大学, 2017.WU Yan-li. System process intensification of high temperature/low temperature Fischer-Tropsch synthesis and product design[D]. Taiyuan: Taiyuan University of Technology, 2017. [85] TENG X, HUANG S, WANG J, WANG H, ZHAO Q, YUAN Y, MA X. Fabrication of Fe2C embedded in hollow carbon spheres: A high-performance and stable catalyst for Fischer-Tropsch synthesis[J]. ChemCatChem,2018,10(17):3883−3891. doi: 10.1002/cctc.201800488 [86] KANG S W, KIM K, CHUN D H, YANG J, LEE H, JUNG H, LIM J T, JANG S, KIM C S, LEE C, JOO S H, HAN J W, PARK J C. High-performance Fe5C2@CMK-3 nanocatalyst for selective and high-yield production of gasoline-range hydrocarbons[J]. J Catal,2017,349:66−74. doi: 10.1016/j.jcat.2017.03.004 [87] JANG S, KANG S W, CHUN D H, LEE H-T, YANG J-I, JUNG H, JEONG H-D, NAM K M, PARK J C. Robust iron-carbide nanoparticles supported on alumina for sustainable production of gasoline-range hydrocarbons[J]. New J Chem,2017,41(7):2756−2763. doi: 10.1039/C7NJ00437K [88] MA Z Z, MA H F, ZHANG H T, WU X, QIAN W X, SUN Q W, YING W Y. Direct conversion of syngas to light olefins through Fischer-Tropsch synthesis over Fe-Zr catalysts modified with sodium[J]. ACS Omega,2021,6(7):4968−4976. doi: 10.1021/acsomega.0c06008 [89] YANG X, ZHANG H, LIU Y, NING W, HAN W, LIU H, HUO C. Preparation of iron carbides formed by iron oxalate carburization for Fischer-Tropsch synthesis[J]. Catalysts,2019,9(4):347. doi: 10.3390/catal9040347 [90] LU Y, YAN Q, HAN J, CAO B, STREET J, YU F. Fischer-Tropsch synthesis of olefin-rich liquid hydrocarbons from biomass-derived syngas over carbon-encapsulated iron carbide/iron nanoparticles catalyst[J]. Fuel,2017,193:369−384. doi: 10.1016/j.fuel.2016.12.061 [91] ZHAO X, LV S, WANG L, LI L, WANG G, ZHANG Y, LI J. Comparison of preparation methods of iron-based catalysts for enhancing Fischer-Tropsch synthesis performance[J]. Mol Catal,2018,449:99−105. doi: 10.1016/j.mcat.2018.02.022 [92] TANG L, HE L, WANG Y, CHEN B, XU W, DUAN X, LU A. Selective fabrication of χ-Fe5C2 by interfering surface reactions as a highly efficient and stable Fischer-Tropsch synthesis catalyst[J]. Appl Catal B: Environ,2021,284:119753. doi: 10.1016/j.apcatb.2020.119753 [93] LU F, CHEN X, WEN L, WU Q, ZHANG Y. The synergic effects of iron carbides on conversion of Syngas to alkenes[J]. Catal Lett,2021,151(7):2132−2143. doi: 10.1007/s10562-020-03505-4 [94] 张红. 碳化铁的制备及其费托合成制低碳烯烃反应性能的研究[D]. 杭州: 浙江工业大学, 2019.ZHANG Hong. Preparation of iron carbides and its performance of Fischer-Tropsch synthesis to light olefins[D]. Hangzhou: Zhejiang University of Technology, 2019. [95] ZHAI P, XU C, GAO R, LIU X, LI M, LI W, FU X, JIA C, XIE J, ZHAO M, WANG X, LI Y W, ZHANG Q, WEN X D, MA D. Highly tunable selectivity for syngas-derived alkenes over zinc and sodium-modulated Fe5C2 catalyst[J]. Angew Chem Int Ed,2016,55(34):9902−9907. doi: 10.1002/anie.201603556 [96] TIAN Z, WANG C, YUE J, ZHANG X, MA L. Effect of a potassium promoter on the Fischer-Tropsch synthesis of light olefins over iron carbide catalysts encapsulated in graphene-like carbon[J]. Catal Sci Technol,2019,9(11):2728−2741. doi: 10.1039/C9CY00403C -




 下载:
下载: