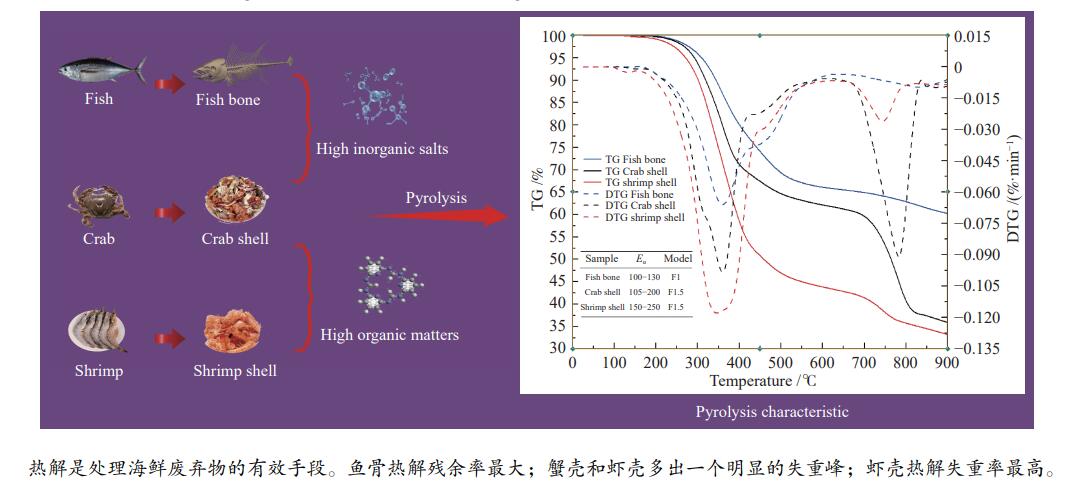
Pyrolysis characteristics and kinetics of typical seafood wastes
-
摘要: 本研究采用热重分析手段,对鱼骨、蟹壳、虾壳三种典型海鲜废弃物的热解特性进行分析,研究不同升温速率(20、40、60 ℃/min)条件下热解过程的特征参数,分析原料成分组成对于海鲜废弃物热解特性的影响。基于热解特性参数对热解过程进行动力学分析,结合表观动力学参数并在对比多种机理模型的拟合效果基础上,确定了较为适宜的海鲜废弃物热解过程机理模型。结果表明,海鲜废弃物热解过程与其成分组成具有密切关联,TG-DTG曲线对比分析发现,有机质和无机盐含量是影响热解过程的重要因素。随着升温速率的提高, 三种海鲜废弃物的热解特性参数具有一致的增长趋势。动力学研究发现,鱼骨主要热解过程符合一级化学反应机理,而蟹壳和虾壳在有机物分解阶段可由一段1.5级化学反应过程描述,分析认为,反应级数的差别与与海鲜废弃物中几丁质含量有关。表观活化能Ea随着升温速率的提高而增大,而活化能增量∆Eα逐渐变小,可以推测采用不低于40 ℃/min的升温速率不会导致三种海鲜废弃物热解过程难度的增大并更具经济性。研究结果为海鲜废弃物热解处理技术的开发提供了基础过程特性数据。Abstract: Pyrolysis is an important technology for the harmless reduction of food waste. In this paper, thermogravimetric analysis was used to analyze the pyrolysis characteristics of three typical seafood wastes, namely fish bones, crab shells, and shrimp shells, and to study the characteristic parameters of the pyrolysis process at different heating rates (20, 40, and 60 ℃/min), to analyze the effect of different components on the pyrolysis characteristics of seafood waste. The kinetic analysis of the pyrolysis process was carried out based on the pyrolysis characteristic parameters, combined with the apparent kinetic parameters and the fitting effects of various mechanism models were compared, and a more suitable mechanism model for the pyrolysis process of seafood waste was determined. The results showed that the pyrolysis processes of the three seafood wastes were closely related to their components, and the comparative analysis of TG-DTG curves found that the content of organic matter and inorganic salts were important factors affecting the pyrolysis process. The pyrolysis characteristic parameters of the three seafood wastes showed a consistent increasing trend with the increase of the heating rate. The main pyrolysis process of fish bones conforms to the first-order chemical reaction mechanism, and the organic matter decomposition stage of crab shells and shrimp shells can be described by a 1.5-order chemical reaction process. The large amount of chitin in shrimp shells and crab shells is the main reason for the difference in the order of chemical reactions. The activation energy increases with the increase of the heating rate, while the increment of the activation energy gradually decreases. It could be presumed that the adoption of larger heating rates over 40 ℃/min would probably not arise more severe pyrolysis condition, which would be more economical from technical view. The results achieved in this paper may provide some fundamentals for competitive technology development for seafood wastes pyrolysis and carbonization.
-
Key words:
- seafood waste /
- pyrolysis /
- heating rate /
- kinetic analysis /
- activation energy
-
表 1 海鲜废弃物原料工业分析及元素分析
Table 1 Proximate and ultimate analysis of samples
Sample Proximate analysis wdry/% Elemental analysis wdry/% QHHV/(MJ·kg−1) M A V FC C H N S O* Fish bone 1.88 54.54 41.70 1.88 23.94 3.74 27.23 0.34 44.75 8.63 Crab shell 1.73 30.96 64.50 2.81 25.80 4.04 31.43 0.27 38.46 12.46 Shrimp shell 2.52 20.54 70.95 5.99 37.74 4.87 43.05 0.32 14.02 15.70 *: by difference 表 2 海鲜废弃物原料化学组成分析
Table 2 Chemical composition of samples
Project/% Fish bone Crab shell Shrimp shell Crude protein 28.17 19.28 40.29 Crude fat 13.26 3.67 8.12 Chitin 2.14 26.59 24.20 Calcium carbonate − 49.66 26.41 Else 56.43 0.80 0.98 Else: by difference 表 3 不同反应机理、模型及f (α)和g(α)
Table 3 Different reaction mechanisms, models, f (α) and g(α)
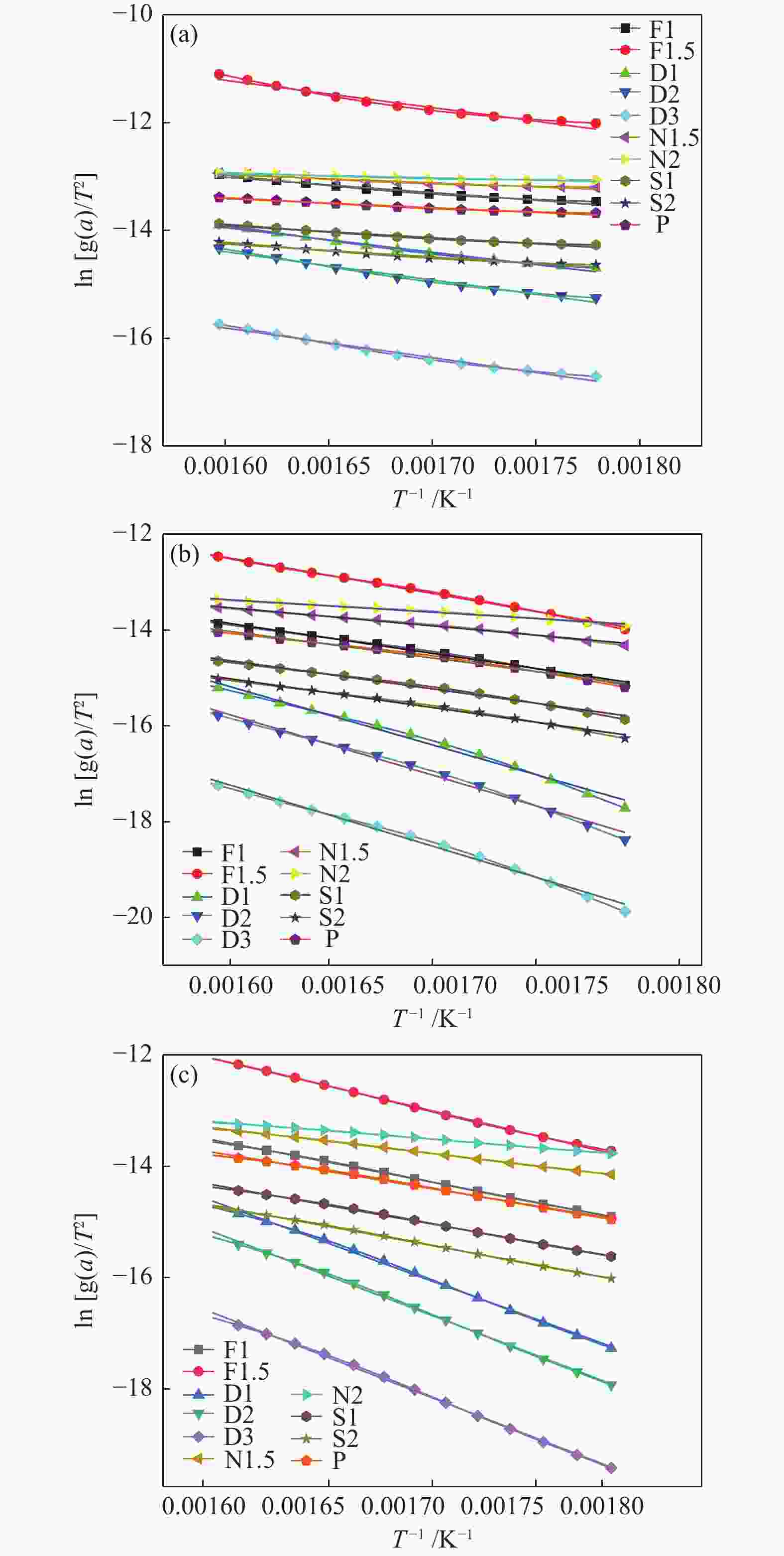
Reaction mechanism Model name ${f}\left(\mathrm{\alpha }\right)$ $ \mathrm{g}\left(\mathrm{\alpha }\right) $ Chemical reaction order Chemical Reaction Order 1(F1) $ 1-\mathrm{\alpha } $ $ -\mathrm{l}\mathrm{n}\left(1-\mathrm{\alpha }\right) $ Chemical Reaction Order 1.5(F1.5) $ {\left(1-\mathrm{\alpha }\right)}^{1.5} $ $ 2\left[\mathrm{l}\mathrm{n}{\left(1-\mathrm{\alpha }\right)}^{-1.5}-1\right] $ Diffusion Parabolic law (D1) $\dfrac{1}{2}\mathrm{\alpha }$ $ {\alpha }^{2} $ Valensi equation(D2) ${-\left[\ln\left(1-\alpha \right)\right]}^{-1}$ $ \mathrm{\alpha } + \left(1-\mathrm{\alpha }\right)\mathrm{l}\mathrm{n}\left(1-\mathrm{\alpha }\right) $ Ginstling-Broushtein equation(D3) $\dfrac{3}{2}{\left[{\left(1-\mathrm{\alpha }\right)}^{\frac{1}{3} }-1\right]}^{-1}$ $\left(1-\dfrac{2}{3}\alpha \right)-{\left(1-\alpha \right)}^{\frac{2}{3} }$ Nucleation and growth Avrami-Erofeev equation Nucleation and growth(N1.5) $ 3{\left(1-\mathrm{\alpha }\right)\left[-\mathrm{l}\mathrm{n}\left(1-\mathrm{\alpha }\right)\right]}^{\frac{2}{3}} $ ${\left[-\ln\left(1-\alpha \right)\right]}^{\frac{2}{3} }$ Avrami-Erofeev equation Nucleation and growth(N2) $ 2{\left(1-\mathrm{\alpha }\right)\left[-\mathrm{l}\mathrm{n}\left(1-\mathrm{\alpha }\right)\right]}^{\frac{1}{2}} $ ${\left[-\ln\left(1-\alpha \right)\right]}^{\frac{1}{2} }$ Phase interfacial reaction Shrinkage geometrical column(S1) $ 2{\left(1-\mathrm{\alpha }\right)}^{\frac{1}{2}} $ $ {1-\left(1-\alpha \right)}^{\frac{1}{2}} $ Shrinkage geometrical Spherical(S2) $ 3{\left(1-\mathrm{\alpha }\right)}^{\frac{2}{3}} $ $ {1-\left(1-\alpha \right)}^{\frac{1}{3}} $ Power law Power law(P) $ 1 $ $ \mathrm{\alpha } $ 表 4 热解特性参数
Table 4 Pyrolysis characteristic parameters
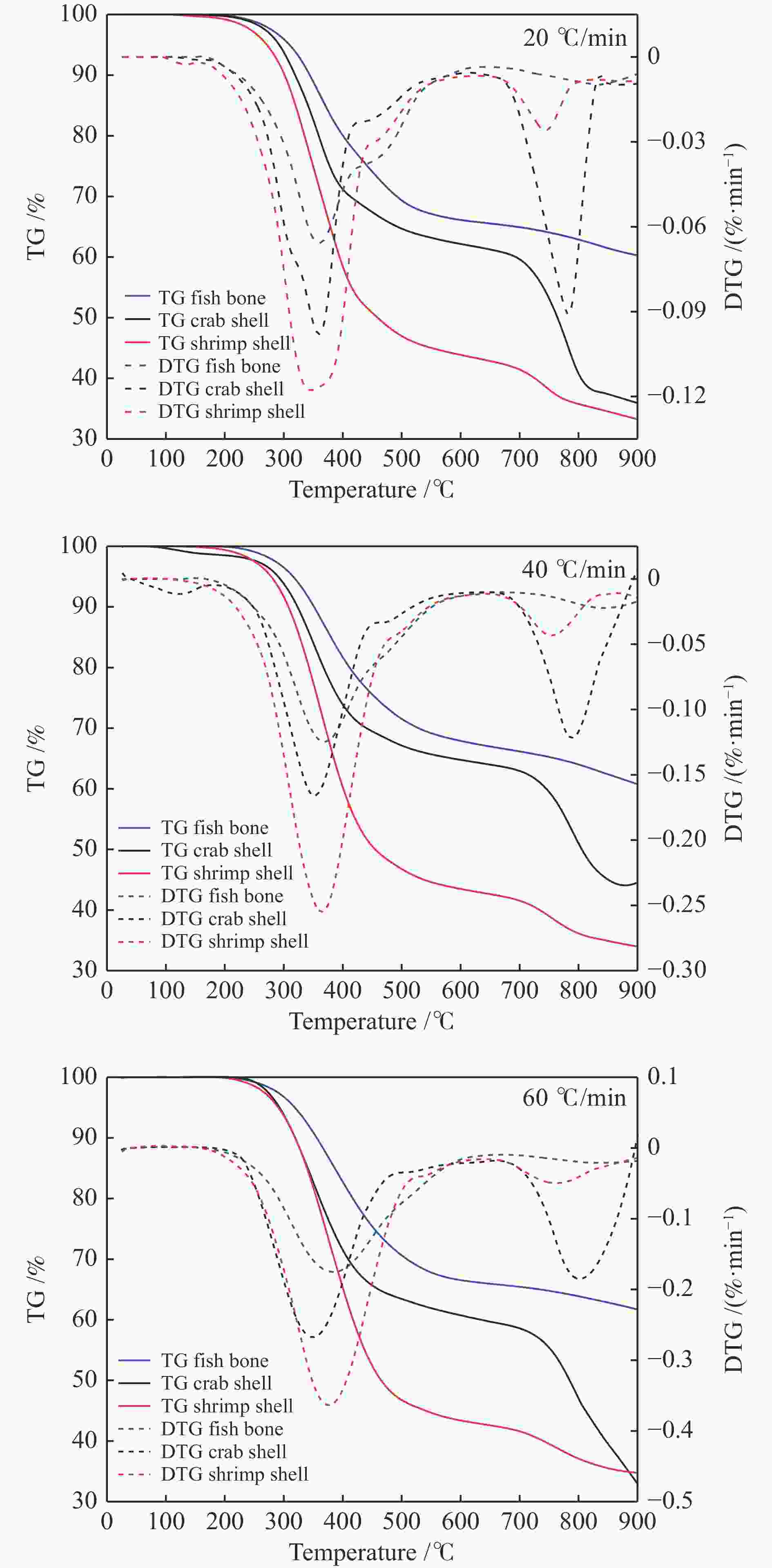
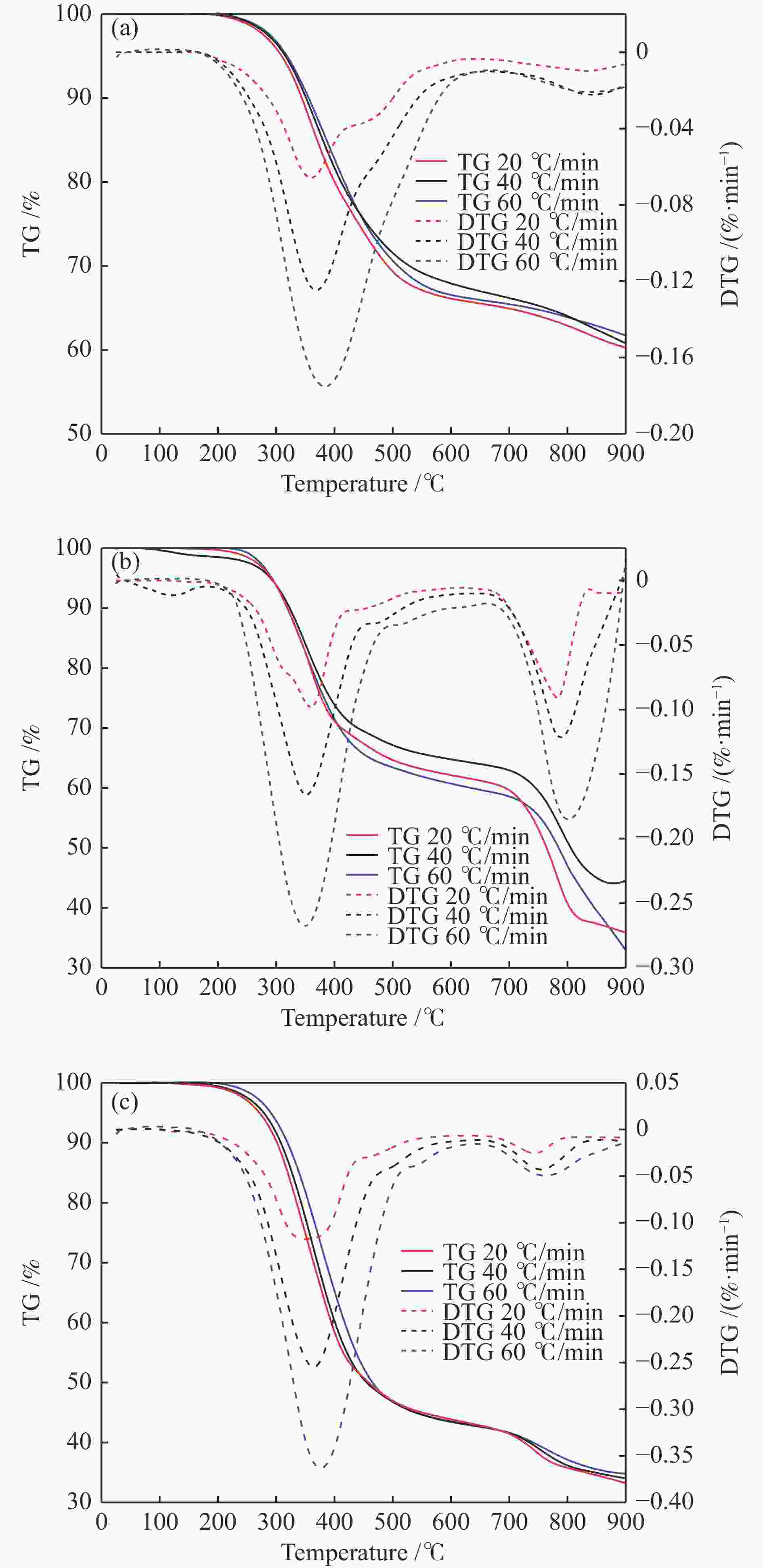
Sample $ {\beta } $/(℃·min−1) ti/℃ te/℃ tmax/℃ $\Delta {t}_{\frac{1}{2} }/$℃ ${\left(\dfrac{ { {\rm{d} } }{w} }{ { {\rm{d} } }{t} }\right)}_{\max}/\left(\% \cdot {\min}^{-1}\right)$ ${\left(\dfrac{ { {\rm{d} } }{w} }{ { {\rm{d} } }{t} }\right)}_{{\rm{mean}}}/\left(\% \cdot {\min}^{-1}\right)$ $ \mathrm{\phi } $/% $ D /{10}^{-5} $ Fish bone 20 289.1 462.2 353.4 132 4.08 0.86 62.21 0.97 40 290.8 465.1 354.9 154 8.25 1.74 62.39 3.42 60 295.2 475.9 370.1 170 12.40 2.27 62.02 5.76 Crab shell 20 290.1 422.3 355.6 165 6.22 1.42 37.69 3.23 40 291.5 424.8 361.5 167 10.17 2.61 43.10 8.58 60 293.9 428.6 363.9 178 20.25 3.58 36.66 24.12 Shrimp shell 20 281.3 440.4 350.3 160 7.13 1.53 33.33 4.61 40 288.6 444.5 359.1 164 15.21 2.90 36.67 16.43 60 297.4 446.7 370.2 172 24.22 4.11 34.17 34.60 表 5 20 ℃/ min 升温速率条件下各种机理模型拟合情况及动力学参数
Table 5 Fitting precision and pyrolysis kinetic parameters with various mechanistic model under 20 ℃/min heating rate
Sample Model ${E}_{\text{a} }$/
(kJ·mol−1)$ A $/
min−1$ {R}^{2} $ Fish bone F1 24.55 15.18 0.99373 F1.5 64.12 580897.02 0.97987 D1 27.97 12.06 0.94494 D2 36.07 48.42 0.97086 D3 39.74 26.65 0.97909 N1.5 12.80 0.85 0.98876 N2 6.92 0.15 0.97631 S1 15.55 0.67 0.96583 S2 18.30 0.98 0.98019 P 8.63 0.16 0.85496 Crab shell F1 41.80 258.20 0.94215 F1.5 51.71 8377.52 0.99889 D1 78.14 117446.92 0.93472 D2 83.08 181575.48 0.94190 D3 84.88 60732.26 0.94437 N1.5 24.40 7.71 0.92395 N2 15.69 1.12 0.89692 S1 37.70 48.08 0.92887 S2 39.03 44.28 0.93359 P 33.87 37.76 0.91278 Shrimp shell F1 43.75 565.68 0.97009 F1.5 68.08 470422.78 0.99625 D1 73.15 71963.72 0.94220 D2 80.45 191870.25 0.95528 D3 83.29 81155.45 0.95985 N1.5 25.68 13.24 0.96013 N2 16.65 1.71 0.94519 S1 37.17 58.53 0.94942 S2 39.30 64.90 0.95718 P 31.35 28.16 0.92024 表 6 不同升温速率条件下海鲜废弃物的热解动力学参数
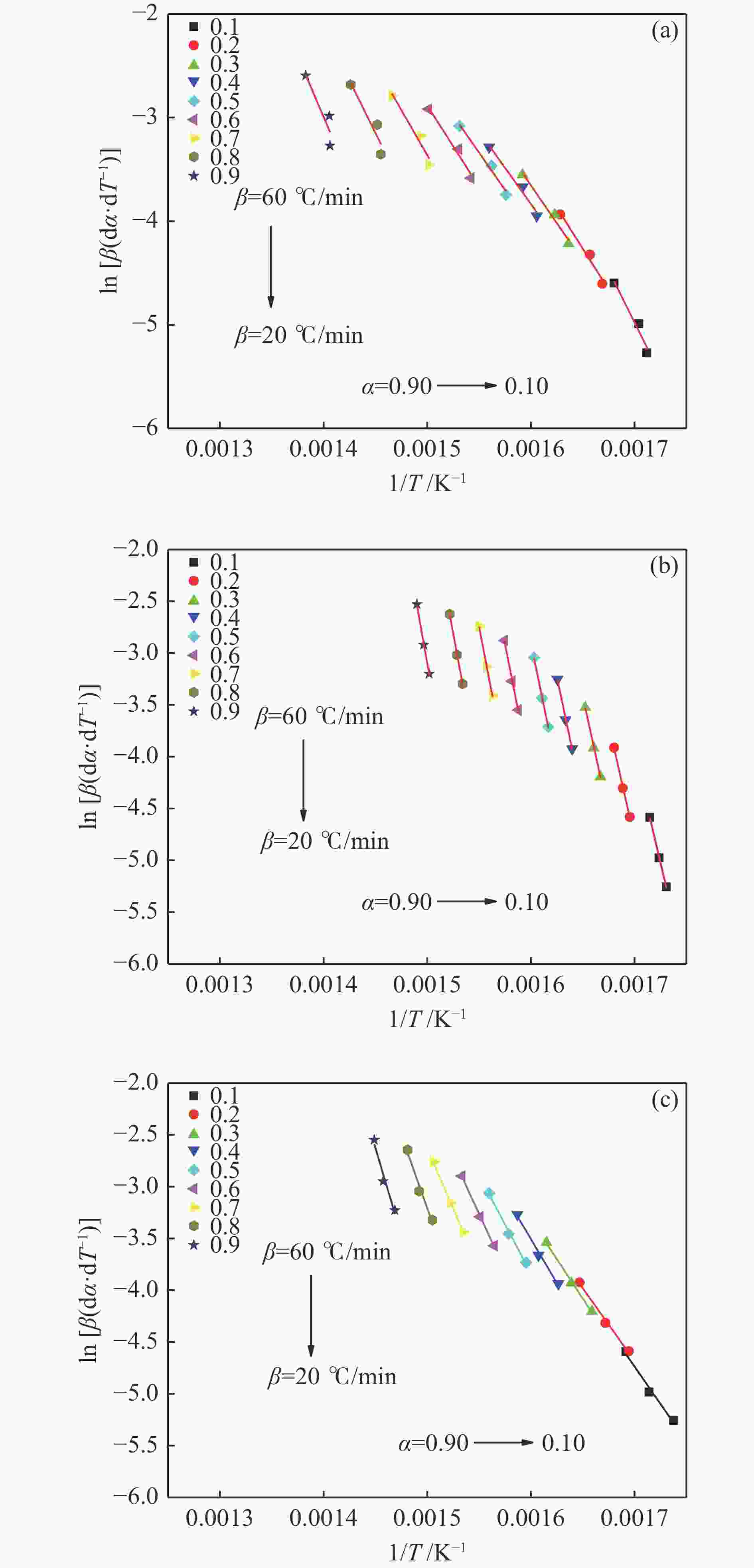
Table 6 Pyrolysis kinetic parameters of seafood wastes under varied heating rates condition
Sample β/(℃·min−1) t/℃ Ea/(kJ·mol−1) A/min−1 R2 Model Fish bone 20 289.1−462.2 24.55 15.18 0.99373 F1 40 290.8−465.1 41.30 502.37 0.99284 F1 60 295.2−475.9 45.01 1370.20 0.99737 F1 Crab shell 20 290.1−422.3 51.71 8377.52 0.99798 F1.5 40 291.5−424.8 56.29 42154.83 0.99622 F1.5 60 293.9−428.6 57.97 126006.41 0.99617 F1.5 Shrimp shell 20 281.3−440.4 68.08 470422.78 0.99625 F1.5 40 288.6−444.5 78.06 7323013.54 0.99780 F1.5 60 297.4−446.7 80.43 13216157.66 0.99755 F1.5 -
[1] ISLAM MD S, KHAN S, TANAKA M. Waste loading in shrimp and fish processing effluents: Potential source of hazards to the coastal and nearshore environments[J]. Mar Pollut Bull,2004,49:103−110. doi: 10.1016/j.marpolbul.2004.01.018 [2] GHALY A E, RAMAKRISHNAN V V, BROOKS M S, BUDGE S M, DAVE D. Fish processing wastes as a potential source of proteins, amino acidsand oils: A critical review[J]. J Micro Biochem Technol,2013,5(4):107−129. doi: 10.4172/1948-5948.1000110 [3] YAN N, CHEN X. Sustainability: Don’t waste seafood waste. Nature, 2015, 524: 155–157. [4] NDINELAGO E V, TIMOTEUS K, NAFTAL G N, LEE T K, SEM I, TIAGO M. Assessment and quantification of Namibian seafood waste production[J]. Ocean Coast Manage,2021,199:105402. [5] ELKHALIFA S, AL-ANSARI T, MACKEY HR, MCKAY G. Food waste to biochars through pyrolysis: A review[J]. Res Conserv Recy,2019,144:310−320. [6] JOGEIR T, SISSEL A, BRITT H, ANDERS A. Chemical composition, mineral content and amino acid and lipid profiles in bones from various fish species[J]. Comp Biochem Physiol B Biochem Mol Biol,2007,146(3):395−401. [7] REN X, WANG J, YU J, SONG B, FENG H, SHEN M, ZHANG H, ZOU J, ZENG G, TANG L, WANG J. Waste valorization: Transforming the fishbone biowaste into biochar as an efficient persulfate catalyst for degradation of organic pollutant[J]. J Clean Prod,2021,291:125225. [8] SHI G, HE S, CHEN G, RUAN C, MA Y, CHEN Q, JIN X, LIU X, HE C, DU C, DAI H, YANG X. Crayfish shell-based micro-mesoporous activated carbon: Insight into preparation and gaseous benzene adsorption mechanism[J]. Chem Eng J,2022,428:131148. [9] XUE X, WENG Y, JIANG Z, YANG S, WU Y, MENG S, ZHANG C, SUN Q, ZHANG Y. Naturally nitrogen-doped porous carbon derived from waste crab shell as anode material for high performance sodium-ion battery[J]. J Anal Appl Pyrol,2021,157(2):105215. [10] NORISHAM S F, MUNAHAT N F. Torrefaction of fish waste (Sardinella fimbriata) in a fixed bed reactor: Yields and biochar characterization[C]. IOP Conf Ser: Earth Environ. Sci. , Malaysia: IOP Publishing, 2021, 765: 012022. [11] DOU S, KE X, SHAO Z, ZHONG L, ZHAO Q, ZHENG Y. Fish scale-based biochar with defined pore size and ultrahigh specific surface area for highly efficient adsorption of ciprofloxacin[J]. Chemosphere,2021,287:131962. [12] DAI L, TAN F, LI H, ZHU N, HE M, ZHU Q, HU G, WANG L, ZHAO J. Calcium-rich biochar from the pyrolysis of crab shell for phosphorus removal[J]. J Environ Manage,2017,198:70−74. [13] 代敏怡, 郭占斌, 赵立欣, 孟海波, 姚宗路, 盛晨绪, 李丽洁, 吴雨浓, 张迎, 丛宏斌. 玉米秸秆与市政污泥混合热解特性及动力学分析[J]. 农业工程学报,2021,37(2):242−250. doi: 10.11975/j.issn.1002-6819.2021.2.028DAI Min-yi, GUO Zhan-bin, ZHAO Li-xin, MENG Hai-bo, YAO Zong-lu, SHENG Chen-xu, LI Li-jie, WU Yu-nong, ZHANG Ying, CONG Hong-bin. Pyrolysis characteristics and kinetic analysis of mixed pyrolysis of corn stover and municipal sludge[J]. Trans CSAE,2021,37(2):242−250. doi: 10.11975/j.issn.1002-6819.2021.2.028 [14] VYAZOVKIN S, BURNHAM A K, FAVERGEON L, KOGA N, MOUKHINA E, PÉREZ-MAQUEDA L A, SBIRRAZZUOLI N. ICTAC Kinetics Committee recommendations for analysis of multi-step kinetics[J]. Thermochim Acta,2020,689:178597. [15] NAQVI S R, TARIQ R, HAMEED Z, ALI I, NAQVI M, CHEN W H, CEYLAN S, RASHID H, AHMAD J, TAQVI S A, SHAHBAZ M. Pyrolysis of high ash sewage sludge: Kinetics and thermodynamic analysis using Coats-Redfern method[J]. Renewable Energy,2019,131:854−860. [16] WANG W, LIU Y, SONG S, CAI W. Facile pyrolysis of fishbone charcoal with remarkable adsorption performance towards aqueous Pb (II)[J]. J Environ Chem Eng,2017,5(5):4621−4629. [17] PHAM V N, VAN H N, TRUNG T S, Properties of hydroxyapatites prepared from different fish bones: A comparative study. Ceram Int, 2019, 45: 20141-20147. [18] MUHAMAD N, TRIANNISA R, FITRI D. Synthesis and characterization of biochar from crab shell by pyrolysis[C]. IOP Conf Ser: Mater Sci Eng, Indonesia: IOP Publishing, 2019, 553: 012031. [19] SUWANNASINGHA N, KANTAWONG A, TUNKIJJANUKIJ S, AENGLONG C, LIU H-B, KLAYPRADIT W. Effect of calcination temperature on structure and characteristics of calcium oxide powder derived from marine shell waste[J]. J Saudi Chem Soc,2022,26:101441. [20] HARYATI E, DAHLAN K, TOGIBASA O, DAHLAN K. The influence of calcination temperature to calcium content in the mangrove crab shells (Scylla serrata) from merauke[J]. JPCS,2021,1940(1):012024. [21] GBENEBOR O P, ADEOSUN S O, LAWAL G I, JUN S. Role of CaCO3 in the physicochemical properties of crustacean-sourced structural polysaccharides[J]. Mater Chem Phys,2016,184:203−209. [22] LI X, LV Y, MA B, WANG W, JIAN S. Decomposition kinetic characteristics of calcium carbonate containing organic acids by TGA[J]. Arab J Chem,2017,10(S2):S2534−S2538. [23] GLASNER N, HANSEN H C B, YONGFENG H, BEKIARIS G, BRUUN S. Low crystalline apatite in bone char produced at low temperature ameliorates phosphorus-deficient soils[J]. Chemosphere,2019,223:723−730. [24] BOUTINGUIZA M, POU J, COMESAÑA R, LUSQUIÑOS F, CARLOS AD, LEÓN B. Biological hydroxyapatite obtained from fish bones[J]. Mat Sci Eng C,2012,32(3):478−486. [25] WANG G, DAI G, DING S, WU J, WANG S. A new insight into pyrolysis mechanism of three typical actual biomass: The influence of structural differences on pyrolysis process[J]. J Analyt Appl Pyrolysis,2021,156:105184. [26] 杜瑞岭, 吴铿, 刘起航, 员晓, 张黎. 分段法研究兴隆庄煤热解反应过程动力学[J]. 哈尔滨工业大学学报,2016,48(4):172−176. doi: 10.11918/j.issn.0367-6234.2016.04.029DU Rui-ling, WU Keng, LIU Qi-hang, YUAN Xiao, ZHANG Li. Study on kinetics of Xinglongzhuang coal pyrolysis process by segmentation method[J]. J Harbin Inst Technol,2016,48(4):172−176. doi: 10.11918/j.issn.0367-6234.2016.04.029 [27] MOHSIN R, BASIM A J, AL-MARZUOQI A H, ABRAR I. Kinetic and thermodynamic analyses of date palm surface fibers pyrolysis using Coats-Redfern method[J]. Renewable Energy,2022,183:67−77. [28] 陈志文. 基于热质传递过程的桉木屑热解模型研究[D]. 广州: 华南农业大学, 2018.CHEN Zhi-wen. Study on the pyrolysis model of eucalyptus wood chips based on heat and mass transfer process[D]. Guangzhou: South China Agriculrual University, 2018 [29] ZENG L, HU X, GU N, FU B, QIN C. Investigation of volatile chemicals and their distributions from pyrolysis of chitin by FT-IR and GC-MS[J]. J Analyt Appl Pyrolysis,2015,112:357−362. [30] STOLAREK P, LEDAKOWICZ S. Pyrolysis kinetics of chitin by non-isothermal thermogravimetry[J]. Thermochim Acta,2005,433:200−208. [31] 张晓东, 许敏, 孙荣峰, 孙立. 玉米秸热解动力学研究[J]. 燃料化学学报,2006,34(1):123−125.ZHANG Xiao-dong, XU Min, SUN Rong-feng, SUN Li. Study on the kinetics of pyrolysis of corn stover[J]. J Fuel Chem Technol,2006,34(1):123−125. [32] CHENG Q, JIANG M, CHEN Z, WANG X, XIAO B. Pyrolysis and kinetic behavior of banana stem using thermogravimetric analysis[J]. Energy Source Part A,2016,38(22):3383−3390. doi: 10.1080/15567036.2016.1153754 -




 下载:
下载: