Synthesis of γ-Mo2N/C catalysts and its performance on formic acid dehydrogenation
-
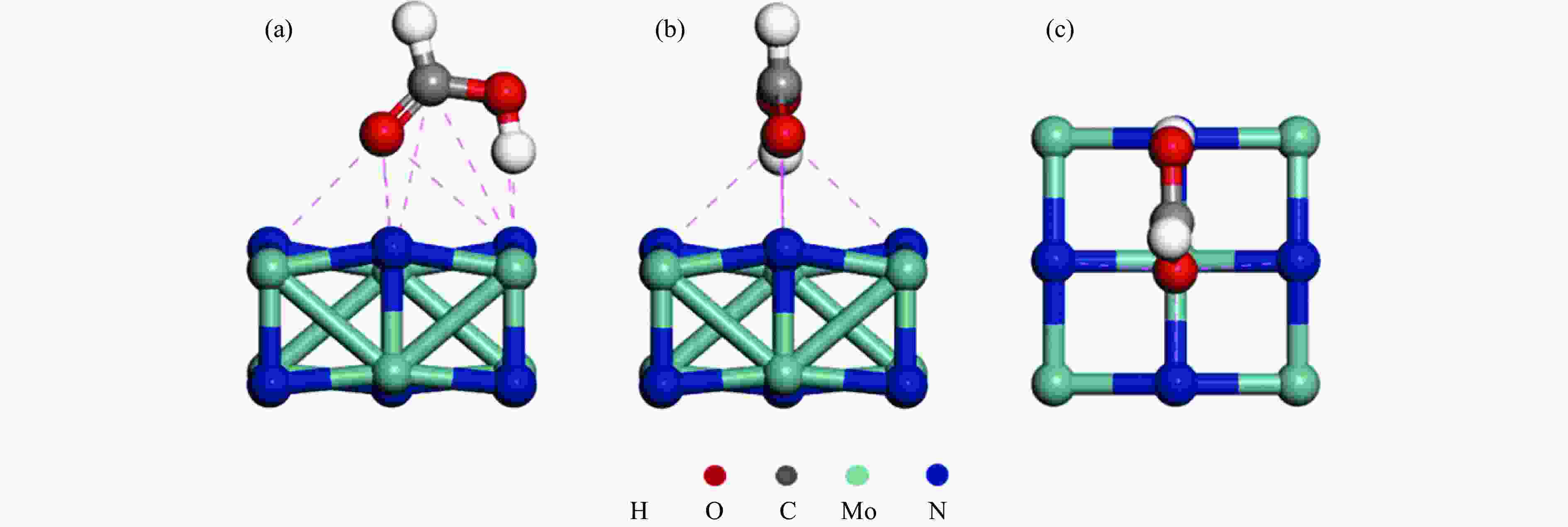
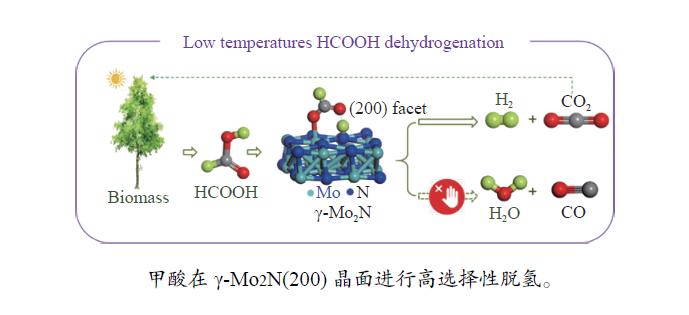
摘要: 甲酸 (FA) 因其H含量较高 (4.4%)、易产H2、可经小平台化合物合成等优势受到广泛关注,而γ-Mo2N/C对FA沿H2和CO2路径分解具有非常高的选择性,产生CO极少,显示出较高的应用价值。基于此,本研究采用对苯二胺和钼酸铵水溶液经前驱体制备γ-Mo2N/C催化剂,并对其FA分解性能进行了原位评价,采用热重分析 (TG)、X射线衍射 (XRD)、傅里叶变换红外光谱 (FT-IR)、扫描电镜 (SEM)、透射电镜 (TEM) 等表征手段对催化剂的结构和表面官能团进行了分析,利用DFT对FA在γ-Mo2N (200) 晶面的吸附构型进行了计算,在此基础上,对催化剂性能及FA在其表面的分解机理进行了研究。结果表明,γ-Mo2N/C在较低温度下即可表现出极高的催化活性,提高γ-Mo2N在C载体上的分散性能有效改善FA转化率。对苯二胺与钼酸铵的物质的量比为4∶1时,催化性能最佳,在160 ℃、100 h的FA分解实验中,催化剂性能稳定、H2选择性高(N2 40 mL/min, CO<5.0×10−5)。而DFT计算表明,FA中O−H键的H原子与γ-Mo2N/C (200) 晶面上N原子结合的可能性更大,而C=O键的O原子更有可能与γ-Mo2N/C (200) 晶面上Mo原子结合。上述结果有助于明确FA在γ-Mo2N/C作用下的分解机理,也显示出非贵金属催化剂γ-Mo2N/C在FA分解制H2方面潜在的应用前景。Abstract: Formic acid (FA) has received much attention due to its high hydrogen content (4.4%), easy H2 production and synthesis from small platform compounds. γ-Mo2N/C is very selective for the decomposition of FA along the H2 and CO2 pathways, generating very little CO and showing high application value. In this study, γ-Mo2N/C catalysts were prepared using aqueous p-phenylenediamine and ammonium molybdate solutions as precursors, and their FA decomposition performance was evaluated in-situ. The adsorption conformation of FA on the crystalline surface of γ-Mo2N (200) was calculated by DFT, and on this basis, the catalyst performance and the decomposition mechanism of FA on its surface were investigated. The results showed that γ-Mo2N/C exhibited very high catalytic activity at low temperatures and that improving the dispersion of γ-Mo2N on the C carrier was effective in improving the FA conversion. The best catalytic performance was achieved at a molar ratio of 4∶1 between p-phenylenediamine and ammonium molybdate, and the catalyst showed stable performance and high H2 selectivity (N2 40 mL/min, CO<5.0×10−5) in the FA decomposition experiments at 160 ℃ and 100 h. DFT calculations showed that the H atom of the O−H bond in FA was more likely to bind to the N atom on the crystalline surface of γ-Mo2N/C (200), while the O atom of the C=O bond are more likely to bind to Mo atoms on the γ-Mo2N/C (200) crystal plane. The above results help to clarify the mechanism of FA decomposition under the action of γ-Mo2N/C and show the potential application of the non-precious metal catalyst γ-Mo2N/C in the decomposition of FA for H2 production.
-
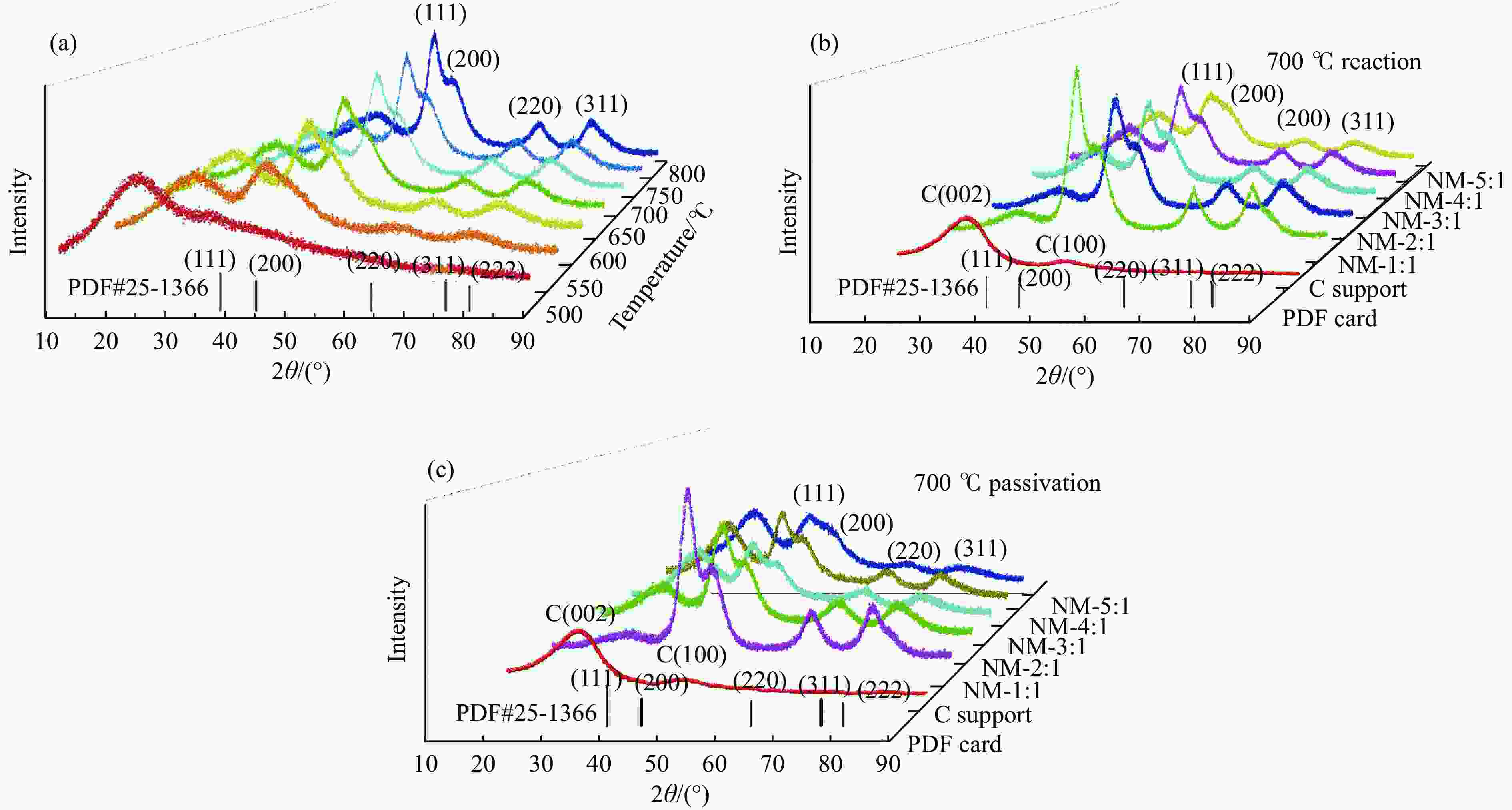
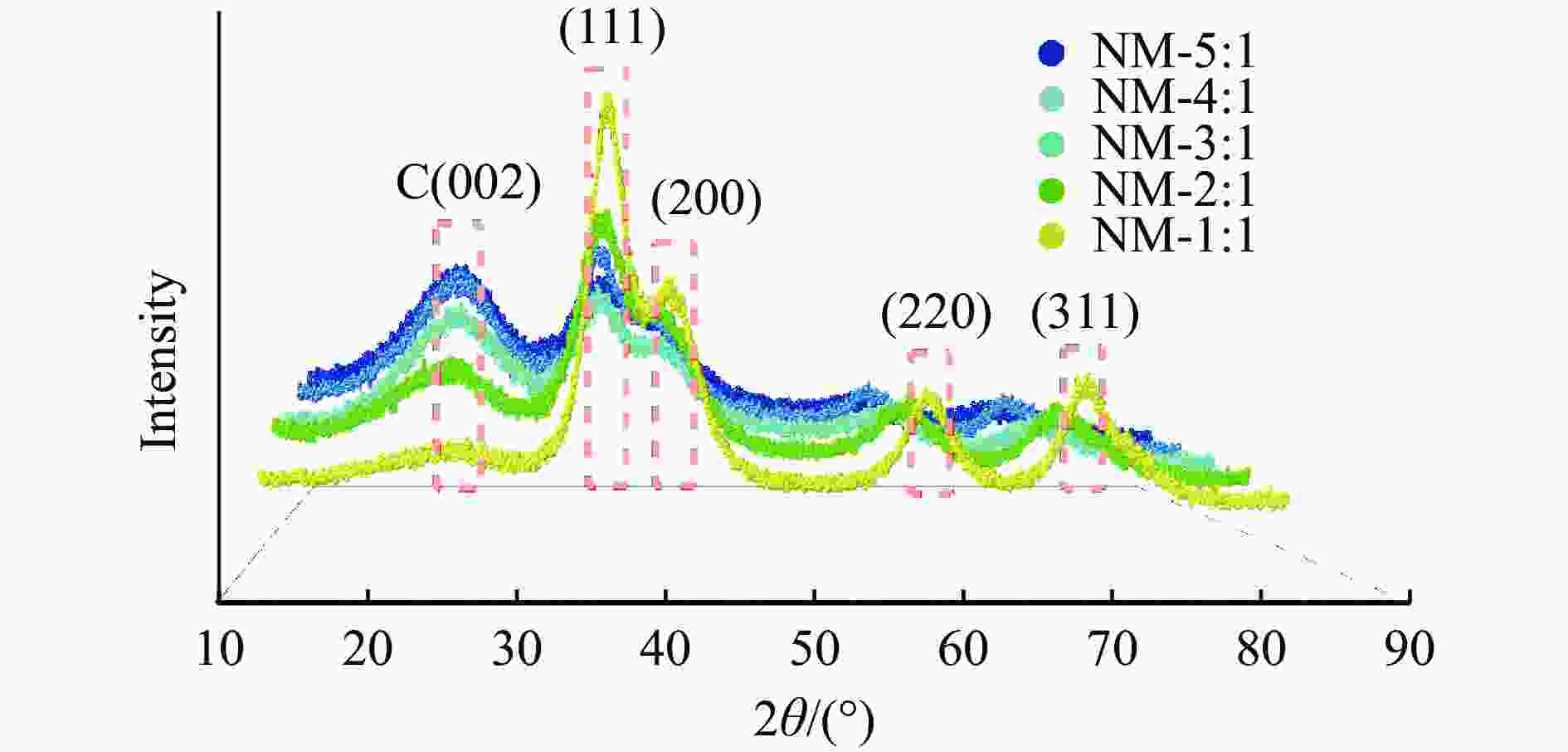
图 5 不同γ-Mo2N/C的XRD谱图
Figure 5 The XRD results of different γ-Mo2N/C (a): XRD patterns of γ-Mo2N/C synthesised by pyrolysis at different temperatures; (b): XRD patterns of C carriers synthesized at 700 ℃ and different ratios of γ-Mo2N/C after reaction with formic acid; (c): XRD patterns of C carriers and different ratios of γ-Mo2N/C at 700 ℃ after passivation with oxygen.
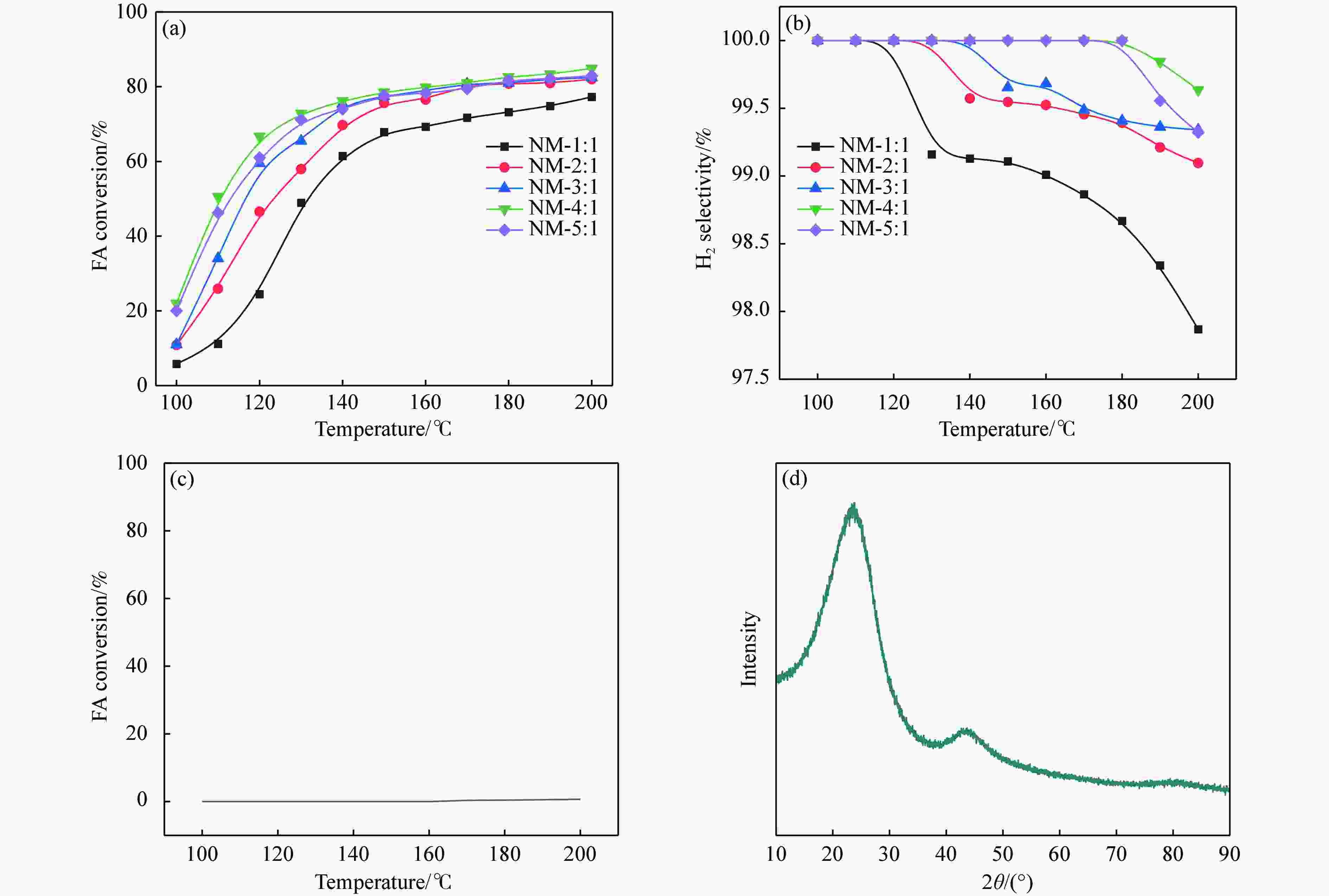
图 13 700 ℃下不同合成比例的γ-Mo2N/C在不同温度下分解FA的H2选择性和FA转化率及C载体的FA转化率与其XRD谱图
Figure 13 H2 selectivity and FA conversion of different synthetic ratios on γ-Mo2N/C at 700 ℃ for the decomposition of FA at different temperatures and FA conversion of C carriers with their XRD patterns (a): FA conversion on γ-Mo2N/C; (b): H2 selectivity; (c): FA conversion over C carriers; (d): XRD spectra of C carriers.
表 1 不同温度下获得的γ-Mo2N/C的N和H含量
Table 1 N and H contents of γ-Mo2N/C obtained at different temperatures
Pyrolysis temperature/℃ N w/% H w/% 500 14.92 1.80 550 12.45 1.50 600 10.07 1.35 650 7.52 0.92 700 5.75 0.65 750 2.76 0.58 800 2.54 0.44 表 2 700 ℃下不同比例催化剂Mo质量分数
Table 2 Mo content of different ratios of catalysts at 700 ℃
Sample Mo
(SEM-EDS, %)Mo
(ICP-MS, %)Moδ+
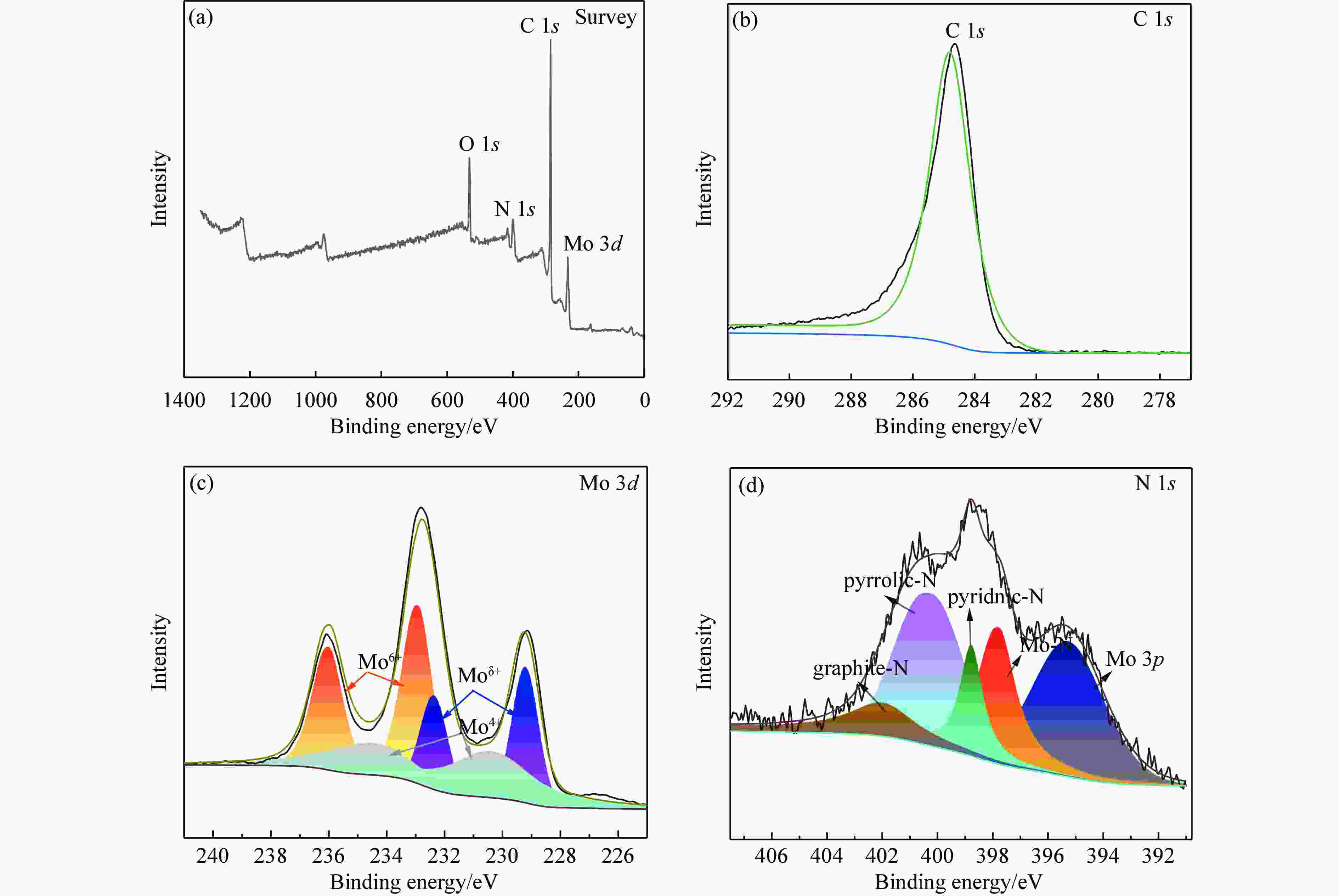
(XPS, atomic %)NM-1∶1 47.52 68.16 0.64 NM-2∶1 39.02 59.21 0.38 NM-3∶1 21.44 46.48 0.22 NM-4∶1 15.56 35.80 0.15 NM-5∶1 14.36 33.20 0.07 表 3 不同合成比例下FA评价后与钝化后获得的γ-Mo2N/C的N和C质量分数
Table 3 N and C contents of γ-Mo2N/C obtained after formic acid reaction and passivation at different synthesis ratios
Composite ration N w/% C w/% NM-1∶1re 1.25 27.94 NM-1∶1pa 1.28 28.27 NM-2∶1 re 1.86 42.42 NM-2∶1 pa 2.14 40.48 NM-3∶1 re 2.28 46.37 NM-3∶1 pa 2.34 46.16 NM-4∶1re 2.53 54.38 NM-4∶1pa 2.77 52.86 NM-5∶1re 2.98 55.52 NM-5∶1pa 3.01 53.70 re: evaluation of the catalyst after testing; pa: passivated catalyst. 表 4 700 ℃合成不同比例催化剂的物理性质
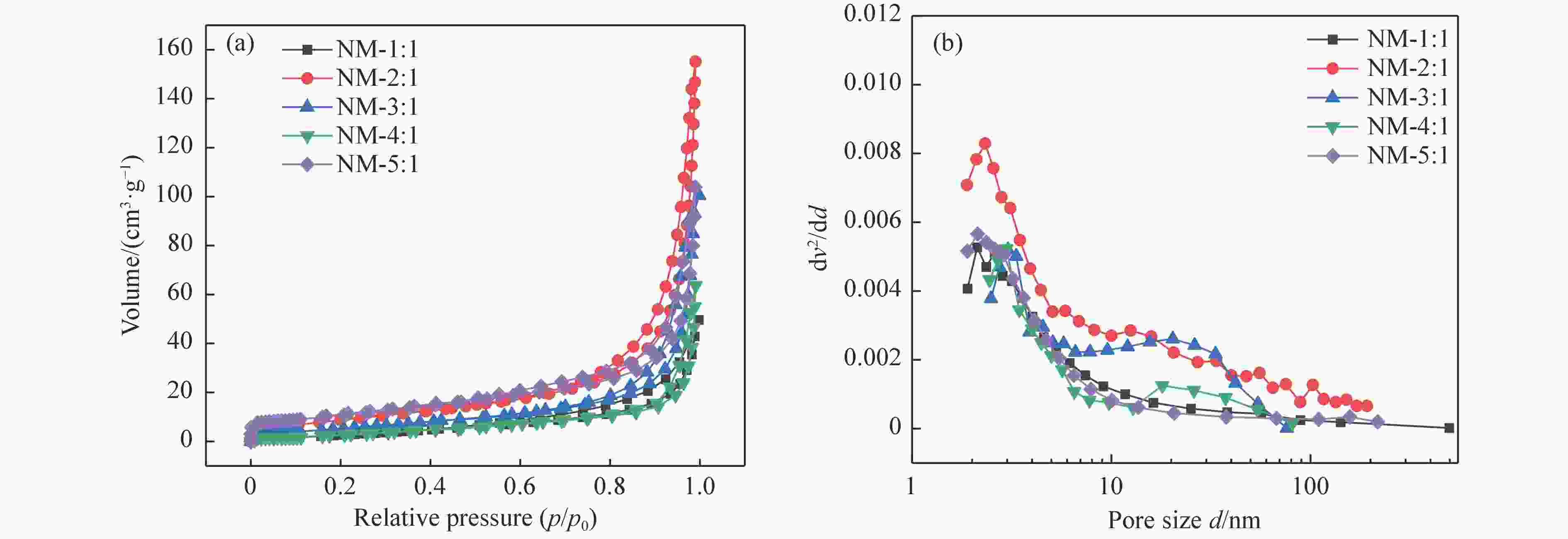
Table 4 Physical properties of catalysts for the synthesis of different ratios of catalysts at 700 ℃
Sample Surface area/(m2·g−1) Pore volume/(cm3·g−1) Macropore ratio/
%NM-1∶1 6.52 0.07 43.28 NM-2∶1 13.44 0.13 52.44 NM-3∶1 13.97 0.15 53.08 NM-4∶1 17.06 0.20 59.20 NM-5∶1 13.88 0.16 46.86 表 5 钝化和评价后的γ-Mo2N/C的元素分析
Table 5 Elemental content of γ-Mo2N/C after passivation and evaluation
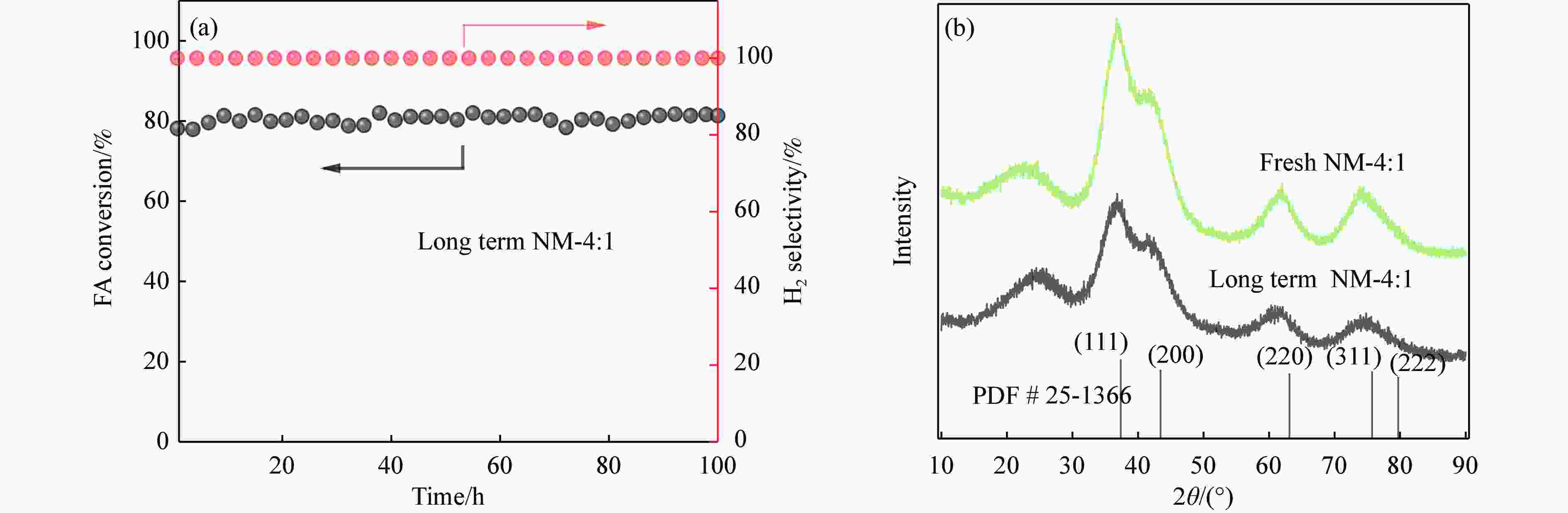
Sample Element abundance w /% N C H Fresh 2.54 53.70 0.77 Long term 2.78 55.55 0.76 表 6 钝化和评价后的γ-Mo2N/C的物理特性
Table 6 Physical properties of γ-Mo2N/C after passivation and evaluation
Sample Surface area/(m2·g−1) Pore volume/(cm3·g−1) Macropore
ratio/%Fresh 17.06 0.20 59.20 Long term 16.24 0.11 58.00 -
[1] STAFFELL I, SCAMMAN D, ABAD A V, et al. The role of hydrogen and fuel cells in the global energy system[J]. Energy Environ Sci,2019,12(2):463−491. doi: 10.1039/C8EE01157E [2] LEBROUHI B E, DJOUPO J J, LAMRANI B, et al. Global hydrogen development a technological and geopolitical overview[J]. Int J Hydrog Energy,2022,47(11):7016−7048. doi: 10.1016/j.ijhydene.2021.12.076 [3] ZHANG F, ZHAO P C, NIU M, et al. The survey of key technologies in hydrogen energy storage[J]. Int J Hydrog Energy,2016,41(33):14535−14552. doi: 10.1016/j.ijhydene.2016.05.293 [4] PREUSTER P, PAPP C, WASSERSCHEID P. Liquid organic hydrogen carriers (LOHCs): Toward a hydrogen free hydrogen economy[J]. Acc Chem Res,2017,50(1):74−85. doi: 10.1021/acs.accounts.6b00474 [5] SINGH A K, SINGH S, KUMAR A. Hydrogen energy future with formic acid: A renewable chemical hydrogen storage system[J]. Catal Sci Technol,2016,6(1):12−40. doi: 10.1039/C5CY01276G [6] PREUSTER P, ALBERT J. Biogenic formic acid as a green hydrogen carrier[J]. Energy Technol,2018,6(3):501−509. doi: 10.1002/ente.201700572 [7] LANG C G, JIA Y, YAO X D. Recent advances in liquid-phase chemical hydrogen storage[J]. Energy Storage Mater,2020,26:290−312. doi: 10.1016/j.ensm.2020.01.010 [8] BULUSHEV D A, ROSS J R H. Heterogeneous catalysts for hydrogenation of CO2 and bicarbonates to formic acid and formates[J]. Catal Rev-Sci Eng,2018,60(4):566−593. doi: 10.1080/01614940.2018.1476806 [9] BULUSHEV D A, ROSS J R H. Towards sustainable production of formic acid[J]. ChemSusChem,2018,11(5):821−836. doi: 10.1002/cssc.201702075 [10] NEMATOLLAHI P, MA H Y, SCHNEIDER W F, et al. DFT and microkinetic comparison of Ru-doped porphyrin-like graphene and nanotubes toward catalytic formic acid decomposition and formation[J]. J Phys Chem C,2021,125(34):18673−18683. doi: 10.1021/acs.jpcc.1c03914 [11] LI Y S, HE Y L, YANG W W. A high-performance direct formate-peroxide fuel cell with palladium gold alloy coated foam electrodes[J]. J Power Sources,2015,278:569−573. doi: 10.1016/j.jpowsour.2014.12.064 [12] YU X W, MANTHIRAM A. Catalyst selective, scalable membraneless alkaline direct formate fuel cells[J]. Appl Catal B: Environ,2015,165:63−67. doi: 10.1016/j.apcatb.2014.09.069 [13] COPENHAVER T S, PUROHIT K H, DOMALAON K, et al. A microfluidic direct formate fuel cell on paper[J]. Electrophoresis,2015,36(16):1825−1829. doi: 10.1002/elps.201400554 [14] VO T, PUROHIT K, NGUYEN C, et al. Formate: An energy storage and transport bridge between carbon dioxide and a formate fuel cell in a single device[J]. ChemSusChem,2015,8(22):3853−3858. doi: 10.1002/cssc.201500958 [15] LI J J, CHEN W, ZHAO H, et al. Size-dependent catalytic activity over carbon-supported palladium nanoparticles in dehydrogenation of formic acid[J]. J Catal,2017,352:371−381. doi: 10.1016/j.jcat.2017.06.007 [16] MONTANDON-CLERC M, DALEBROOK A F, LAURENCZY G. Quantitative aqueous phase formic acid dehydrogenation using iron(II) based catalysts[J]. J Catal,2016,343:62−67. doi: 10.1016/j.jcat.2015.11.012 [17] CHEN H L, JIANG D C, SUN Z J, et al. Cobalt nitride as an efficient cocatalyst on CdS nanorods for enhanced photocatalytic hydrogen production in water[J]. Catal Sci Technol,2017,7(7):1515−1522. doi: 10.1039/C7CY00046D [18] SUN Z, CHEN H, ZHANG L, et al. Enhanced photocatalytic H2 production on cadmium sulfide photocatalysts using nickel nitride as a novel cocatalyst[J]. J Mater Chem A,2016,4(34):13289−13295. doi: 10.1039/C6TA04696G [19] DHANASEKARAN P, SALUNKE H G, GUPTA N M. Visible-light-induced photosplitting of water over γ′-Fe4N and γ′-Fe4N/α-Fe2O3 Nanocatalysts[J]. J Phys Chem C,2012,116(22):12156−12164. doi: 10.1021/jp303255f [20] MENG X J, QI W L, KUANG W D, et al. Chromium-titanium nitride as an efficient co-catalyst for photocatalytic hydrogen production[J]. J Mater Chem A,2020,8(31):15774−15781. doi: 10.1039/D0TA00488J [21] TOMINAGA H, NAGAI M. Reaction mechanism for hydrodenitrogenation of carbazole on molybdenum nitride based on DFT study[J]. Appl Catal A: Gen,2010,389(1/2):195−204. doi: 10.1016/j.apcata.2010.09.020 [22] GHAMPSON I T, SEPULVEDA C, GARCIA R, et al. Hydrodeoxygenation of guaiacol over carbon-supported molybdenum nitride catalysts: Effects of nitriding methods and support properties[J]. Appl Catal A: Gen,2012,439−440:111−124. doi: 10.1016/j.apcata.2012.06.047 [23] SONG Y J, YUAN Z Y. One-pot synthesis of Mo2N/NC catalysts with enhanced electrocatalytic activity for hydrogen evolution reaction[J]. Electrochim Acta,2017,246:536−543. doi: 10.1016/j.electacta.2017.06.086 [24] YU Z L, AN X W, KURNIA I, et al. Full spectrum decomposition of formic acid over γ-Mo2N-based catalysts: From dehydration to dehydrogenation[J]. ACS Catal,2020,10(9):5353−5361. doi: 10.1021/acscatal.0c00752 [25] KANAZAWA S, YAMADA Y, SATO S. Infrared spectroscopy of graphene nanoribbons and aromatic compounds with sp3C−H (methyl or methylene groups)[J]. J Mater Sci,2021,56(21):12285−12314. doi: 10.1007/s10853-021-06001-1 [26] LIN H, LONG J, GU Q, et al. In situ IR study of surface hydroxyl species of dehydrated TiO2: Towards understanding pivotal surface processes of TiO2 photocatalytic oxidation of toluene[J]. Phys Chem Chem Phys,2012,14(26):9468−9474. doi: 10.1039/c2cp40893g [27] CHEN T, WU G P, FENG Z C, et al. In situ FT-IR study of photocatalytic decomposition of formic acid to hydrogen on Pt/TiO2 catalyst[J]. Chin J Catal,2008,29(2):105−107. doi: 10.1016/S1872-2067(08)60019-4 [28] LV Z, TAHIR M, LANG X W, et al. Well dispersed molybdenum nitrides on a nitrogen-doped carbon matrix for highly efficient hydrogen evolution in alkaline media[J]. J Mater Chem A,2017,5(39):20932−20937. doi: 10.1039/C7TA06981B [29] GANIYU S A, ALHOOSHANI K, ALI S A. Single pot synthesis of Ti-SBA-15-NiMo hydrodesulfurization catalysts: Role of calcination temperature on dispersion and activity[J]. Appl Catal B: Environ,2017,203:428−441. doi: 10.1016/j.apcatb.2016.10.052 [30] TRILLO J M, MUNUERA G, CRIADO J M. Catalytic decomposition of formic acid on metal oxides[J]. Catal Rev-Sci Eng,2006,7(1):51−86. [31] POPOVA G Y, CHESALOV Y A, SADOVSKAYA E M, et al. Effect of water on decomposition of formic acid over V-Ti oxide catalyst: Kinetic and in situ FTIR study[J]. J Mol Catal A: Chem,2012,357:148−153. doi: 10.1016/j.molcata.2012.02.005 [32] SADOVSKAYA E M, CHESALOV Y A, GONCHAROV V B, et al. Formic acid decomposition over V-Ti oxide catalyst: Mechanism and kinetics[J]. Mol Catal,2017,430:54−62. doi: 10.1016/j.molcata.2016.12.010 -




 下载:
下载: